Advanced Catalytic Route for N-Ethyl-D-Glucosamine: Scaling High-Purity Pharmaceutical Intermediates
Advanced Catalytic Route for N-Ethyl-D-Glucosamine: Scaling High-Purity Pharmaceutical Intermediates
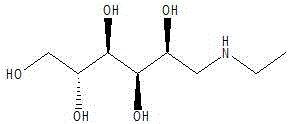
The pharmaceutical industry continuously demands intermediates that combine high stereochemical purity with cost-effective manufacturability. A significant breakthrough in this domain is detailed in patent CN111100018A, which discloses a novel preparation method for N-ethyl-D-glucosamine. This compound serves as a critical chiral building block and drug synthesis intermediate, widely utilized to enhance the lipid solubility and pharmacological efficacy of various active pharmaceutical ingredients. The patented process distinguishes itself by replacing conventional, less efficient catalytic systems with a specialized nickel-containing ternary powder catalyst. This technological shift addresses long-standing challenges regarding product purity and process safety. By optimizing the reductive amination of glucose with monoethylamine, the method achieves a titration content exceeding 99.6% without the need for laborious repeated recrystallization steps. For procurement and R&D teams seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic advantages of this route is essential for securing a stable supply of high-quality materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-ethyl-D-glucosamine has relied heavily on methods such as those disclosed in US patent application US4246193A and US2016962A. These traditional processes typically employ aqueous solutions of glucose and ethylamine, utilizing methanol as a solvent and Raney Nickel as the catalyst. While functional, these legacy methods suffer from significant drawbacks that impact both quality and cost. The primary issue lies in the difficulty of separating the catalyst from the reaction mixture, often leading to high impurity content in the crude product. Consequently, manufacturers are forced to perform repeated recrystallization cycles to achieve acceptable purity levels, which drastically reduces the overall yield and increases solvent consumption. Furthermore, the reaction conditions in these older patents often require rigorous control to prevent side reactions, and the resulting product frequently exhibits a melting point range of only 132-134°C with suboptimal optical rotation, indicating the presence of isomeric impurities that complicate downstream drug formulation.
The Novel Approach
In stark contrast, the method described in CN111100018A introduces a paradigm shift by utilizing a nickel-containing ternary powder catalyst, such as aluminum-nickel-iron (Al-Ni-Fe), aluminum-nickel-molybdenum (Al-Ni-Mo), or aluminum-nickel-cobalt (Al-Ni-Co). This innovative approach begins by dissolving glucose in an alcohol solvent and introducing monoethylamine gas under controlled stirring at moderate temperatures (35-55°C). The subsequent hydrogenation step is conducted under mild pressure (0.5-3.0 MPa) and temperature (50-100°C), which significantly reduces energy consumption compared to high-pressure alternatives. The key advantage of this novel route is the exceptional selectivity of the ternary catalyst, which minimizes side reactions and allows for the direct isolation of the product with a purity greater than 99.6%. This eliminates the need for multiple recrystallization steps, thereby streamlining the workflow and enhancing the overall economic viability of cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Ternary Catalyst Reductive Amination
The core of this technological advancement lies in the sophisticated interaction between the glucose substrate and the nickel-containing ternary catalyst during the reductive amination process. Mechanistically, the reaction proceeds through the initial formation of an imine intermediate when monoethylamine reacts with the aldehyde group of glucose in the alcoholic medium. The ternary catalyst, composed primarily of nickel with specific proportions of aluminum and a third metal component (such as iron, molybdenum, or cobalt), provides a highly active surface for hydrogen activation. Unlike traditional Raney Nickel, which can be pyrophoric and difficult to handle, the ternary powder catalyst offers enhanced structural stability. The specific mass ratios—typically around 47% aluminum, 50% nickel, and 3% third component—create a synergistic effect that optimizes the adsorption of hydrogen and the imine intermediate. This precise catalytic environment ensures that the hydrogenation proceeds rapidly and selectively to form the secondary amine bond without over-reduction or degradation of the sensitive sugar backbone, resulting in yields consistently above 90%.
From an impurity control perspective, the mechanism inherently suppresses the formation of by-products that plague conventional syntheses. In traditional aqueous systems, the presence of water can lead to hydrolysis side reactions or the formation of polymeric impurities that are difficult to remove. By shifting to an alcohol solvent system (methanol or ethanol) and employing the robust ternary catalyst, the reaction environment becomes much more conducive to clean conversion. The catalyst's surface properties prevent the accumulation of tarry residues, allowing for simple filtration post-reaction. This clarity in the reaction profile means that the filtrate contains predominantly the desired N-ethyl-D-glucosamine, which can be crystallized directly upon cooling to 0-10°C. This mechanistic cleanliness is vital for high-purity pharmaceutical intermediates, as it ensures that the final material meets stringent regulatory specifications for heavy metals and organic impurities without requiring extensive purification protocols.
How to Synthesize N-Ethyl-D-Glucosamine Efficiently
Implementing this synthesis route requires careful attention to the sequential addition of reagents and the maintenance of specific reaction parameters to maximize the benefits of the ternary catalyst system. The process is designed to be operationally simple yet chemically precise, starting with the dissolution of glucose in the chosen alcohol solvent followed by the controlled introduction of monoethylamine gas. This initial step is critical for establishing the correct stoichiometry for imine formation before the hydrogenation phase begins. Once the catalyst is introduced and the system is purged with nitrogen to ensure safety, hydrogen is introduced to drive the reduction. The detailed standardized synthetic steps, including exact mass ratios and specific temperature ramps for optimal catalyst performance, are outlined below to guide process engineers in replicating this high-yield pathway.
- Dissolve glucose in an alcohol solvent (methanol or ethanol) and introduce monoethylamine gas, maintaining the temperature between 35-55°C for imine formation.
- Add the nickel-containing ternary powder catalyst (Al-Ni-Fe/Mo/Co) to the system, replace with nitrogen, and introduce hydrogen at 0.5-3.0 MPa pressure.
- React at 50-100°C for approximately 12 hours, then filter to recover the catalyst and crystallize the filtrate to obtain the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic advantages beyond mere chemical yield. The shift from traditional Raney Nickel to a recoverable ternary powder catalyst fundamentally alters the cost structure of production. Traditional catalysts often require complex disposal procedures or result in significant metal loss, whereas the new catalyst can be recovered by simple filtration and potentially reused, leading to substantial cost savings in raw material expenditure. Additionally, the elimination of repeated recrystallization steps reduces solvent usage and waste generation, aligning with modern green chemistry principles and lowering environmental compliance costs. This efficiency gain is crucial for maintaining competitive pricing in the global market for commercial scale-up of complex pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The implementation of the nickel-containing ternary catalyst significantly lowers the overall production cost by enhancing catalyst longevity and recovery rates. Unlike pyrophoric catalysts that pose safety risks and require specialized handling equipment, this stable powder catalyst simplifies the operational workflow. The high selectivity of the reaction minimizes the formation of waste by-products, meaning that less raw material is lost to purification processes. Furthermore, the ability to achieve high purity in a single crystallization step drastically reduces energy consumption associated with heating and cooling cycles, contributing to a leaner and more cost-effective manufacturing profile.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the volatility of raw material availability and the complexity of synthesis routes. This method utilizes readily available starting materials such as glucose and monoethylamine, which are commodity chemicals with stable global supply chains. The robustness of the reaction conditions (moderate temperature and pressure) reduces the likelihood of batch failures due to equipment sensitivity or operator error. By reducing lead time for high-purity pharmaceutical intermediates through a streamlined process, manufacturers can respond more agilely to market demand fluctuations, ensuring that downstream drug production schedules are not disrupted by intermediate shortages.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to industrial reactors often introduces unforeseen challenges, particularly regarding heat transfer and safety. The described method operates within a safe pressure range of 0.5-3.0 MPa and temperatures below 100°C, making it highly scalable using standard hydrogenation reactors. The use of alcohol solvents instead of aqueous mixtures simplifies solvent recovery and recycling, significantly reducing the volume of wastewater generated. This alignment with environmental regulations minimizes the risk of production halts due to compliance issues, ensuring a sustainable and uninterrupted supply of N-ethyl-D-glucosamine for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of N-ethyl-D-glucosamine using this advanced catalytic method. These insights are derived directly from the patent data and practical considerations for industrial application, providing clarity on how this technology outperforms legacy methods. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their supply chains.
Q: What are the advantages of the ternary catalyst over traditional Raney Nickel?
A: The nickel-containing ternary powder catalyst offers superior structural stability and selectivity compared to traditional Raney Nickel. It allows for easier filtration and recovery, eliminates the need for repeated recrystallization, and achieves titration content exceeding 99.6%, significantly reducing impurity profiles.
Q: How does this process improve supply chain safety?
A: By utilizing milder reaction conditions (50-100°C) and a stable heterogeneous catalyst, the process reduces the risks associated with pyrophoric materials often found in traditional methods. This enhances operational safety and ensures more consistent batch-to-batch reliability for large-scale manufacturing.
Q: Can this method be scaled for commercial production?
A: Yes, the method is designed for scalability. The use of standard alcohol solvents and robust hydrogenation parameters (0.5-3.0 MPa) makes it highly adaptable for industrial reactors, facilitating the transition from laboratory synthesis to multi-ton commercial output without compromising yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Ethyl-D-Glucosamine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent to production requires deep technical expertise and robust infrastructure. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the CN111100018A process are fully realized in a commercial setting. Our facilities are equipped with state-of-the-art hydrogenation reactors and rigorous QC labs capable of meeting stringent purity specifications required by global pharmaceutical regulators. We are committed to delivering N-ethyl-D-glucosamine that not only meets but exceeds the quality benchmarks set by this innovative synthesis route, providing our partners with a secure and high-quality source of this critical intermediate.
We invite potential partners to engage with our technical team to explore how this optimized synthesis route can benefit your specific drug development projects. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages tailored to your volume requirements. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
