Scalable Synthesis of Deuterated Difluoromethyl Thioaryl Sulfonate for Advanced Drug Discovery
The pharmaceutical industry is currently witnessing a paradigm shift towards deuterated drugs, driven by the compelling metabolic advantages offered by the kinetic isotope effect. Patent CN114149354A introduces a groundbreaking methodology for the synthesis of deuterated difluoromethyl substituted thioaryl sulfonate, specifically the reagent PhSO2SCF2D. This innovation addresses a critical bottleneck in modern medicinal chemistry: the difficulty of introducing the lipophilic and metabolically stable SCF2D moiety into complex drug scaffolds with high isotopic purity. By leveraging a novel two-step synthetic route that avoids common pitfalls of hydrogen-deuterium exchange, this technology provides a robust platform for developing next-generation therapeutics with improved pharmacokinetic profiles. For research and development teams, access to such high-fidelity deuterated building blocks is essential for optimizing lead compounds and extending patent life cycles through novel isotopic variations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of difluoromethylthio-containing compounds has relied heavily on reagents such as TMSCF2Br or various sulfinates that often suffer from significant drawbacks regarding isotopic integrity. A major challenge in prior art methods is the inadvertent generation of protic sources, such as water, during the reaction process. When synthesizing deuterated analogues, the presence of even trace amounts of H2O can lead to rapid H/D exchange, resulting in a final product with diluted deuterium content that fails to meet the rigorous standards required for metabolic studies. Furthermore, many existing protocols require harsh conditions or unstable intermediates that must be generated in situ, complicating the scale-up process and introducing safety hazards related to the handling of volatile fluorinated gases or reactive halogenated species. These limitations have historically restricted the widespread adoption of SCF2D groups in drug discovery pipelines.
The Novel Approach
The methodology disclosed in CN114149354A offers a transformative solution by utilizing a carefully controlled sequence involving sodium hydride, alkaline heavy water, and diethyl bromodifluoromethyl phosphate. This approach effectively isolates the deuterium source from potential proton contaminants, ensuring that the intermediate benzyl(difluoromethyl-d)sulfane (BnSCF2D) is formed with exceptional isotopic purity. The subsequent conversion to the final sulfonate reagent proceeds under mild conditions, avoiding the extreme temperatures often required by alternative radical pathways. This not only enhances the safety profile of the manufacturing process but also significantly improves the overall yield and reproducibility. By providing a shelf-stable, easily handled reagent, this invention removes the barrier of in-situ generation, allowing chemists to introduce the SCF2D motif into diverse substrates with unprecedented ease and reliability.
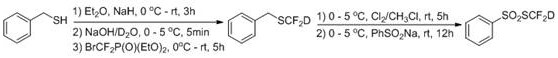
Mechanistic Insights into Deuterated Difluoromethylthiolation
The core of this synthetic strategy lies in the nucleophilic substitution mechanism facilitated by the strong base sodium hydride in an anhydrous ether environment. Initially, benzyl mercaptan is deprotonated to form a reactive thiolate anion, which then attacks the difluorocarbene source derived from the phosphonate reagent. The critical innovation occurs in the quenching and workup phase, where the use of a concentrated alkaline heavy water solution (10% NaOH in D2O) ensures that any protonation events utilize deuterium rather than hydrogen. This precise control over the reaction medium prevents the scrambling of isotopes that plagues other methods. The resulting intermediate, BnSCF2D, retains the deuterium atom on the difluoromethyl group with high fidelity, serving as a perfect precursor for the final transformation. This mechanistic precision is vital for R&D directors who require consistent isotopic labeling for accurate ADME (Absorption, Distribution, Metabolism, and Excretion) profiling.
Following the formation of the deuterated sulfide, the process involves an oxidative chlorination step using chlorine gas in chloroform at controlled low temperatures (0-5°C). This generates a reactive sulfur-chlorine species which is immediately trapped by sodium benzenesulfinate. This second step is a classic nucleophilic substitution at the sulfur center, displacing the chloride with the benzenesulfonyl group to yield the final PhSO2SCF2D product. The choice of sodium benzenesulfinate is strategic, as it provides a stable leaving group capability in subsequent applications while rendering the reagent itself stable for storage. The entire sequence is designed to minimize side reactions and maximize the recovery of the valuable deuterated material, ensuring that the impurity profile remains clean and manageable for downstream pharmaceutical applications.
How to Synthesize PhSO2SCF2D Efficiently
The synthesis of this high-value deuterated reagent requires strict adherence to moisture-free conditions and precise temperature control to maintain isotopic purity. The process begins with the activation of the thiol followed by the introduction of the difluorocarbene equivalent in the presence of a deuterium source. Detailed operational parameters, including specific molar ratios of sodium hydride to benzyl mercaptan (1:1 to 1:1.2) and the concentration of the alkaline heavy water, are critical for success. The subsequent chlorination and sulfonylation steps must be monitored closely to prevent over-oxidation or decomposition of the sensitive fluorinated intermediates. For a comprehensive, step-by-step guide including exact stoichiometry, solvent volumes, and purification techniques such as column chromatography eluent systems, please refer to the standardized protocol below.
- React benzyl mercaptan with sodium hydride in anhydrous ether, followed by treatment with alkaline heavy water and a difluorocarbene reagent to form the deuterated intermediate BnSCF2D.
- Chlorinate the intermediate BnSCF2D using chlorine gas in chloroform at low temperature to generate the reactive sulfur-chlorine species.
- React the chlorinated species with sodium benzenesulfinate at room temperature to yield the final stable reagent PhSO2SCF2D via column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this patented synthesis route offers substantial strategic benefits beyond mere technical performance. The ability to produce a shelf-stable deuterated reagent eliminates the need for pharmaceutical companies to invest in complex, hazardous in-situ generation setups, thereby reducing capital expenditure on specialized equipment and safety infrastructure. Furthermore, the high yield and purity achieved in this process translate directly into cost efficiency, as less raw material is wasted on failed batches or extensive purification efforts. For supply chain heads, the robustness of this method ensures a reliable continuity of supply, mitigating the risks associated with sourcing scarce deuterated building blocks from multiple vendors. This stability is crucial for maintaining uninterrupted production schedules for clinical trial materials and commercial API manufacturing.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of readily available starting materials like benzyl mercaptan and sodium benzenesulfinate significantly lower the raw material costs. Additionally, the high selectivity of the reaction reduces the burden on downstream purification processes, such as chromatography or crystallization, which are often the most cost-intensive stages in fine chemical production. By streamlining the workflow and minimizing waste generation, this process delivers a leaner, more economical manufacturing model that aligns with the industry's drive for sustainable cost reduction in pharmaceutical manufacturing.
- Enhanced Supply Chain Reliability: The reagent PhSO2SCF2D is characterized by its stability at normal temperature and pressure, which simplifies logistics and storage requirements. Unlike volatile or moisture-sensitive alternatives that require cold chain shipping or inert atmosphere handling, this product can be transported and stored using standard protocols. This durability reduces the risk of spoilage during transit and allows for larger batch sizes to be produced and stocked, ensuring that procurement managers can secure long-term inventory buffers against market fluctuations or raw material shortages.
- Scalability and Environmental Compliance: The reaction conditions, primarily operating between 0°C and room temperature, are highly amenable to scale-up in standard stainless steel reactors without the need for cryogenic cooling or high-pressure vessels. This thermal accessibility facilitates the commercial scale-up of complex deuterated intermediates from gram to multi-kilogram scales with minimal engineering modifications. Moreover, the process avoids the use of highly toxic heavy metals, simplifying waste treatment and ensuring compliance with increasingly stringent environmental regulations regarding heavy metal residues in active pharmaceutical ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this deuterated synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the operational realities and potential applications. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this reagent into their existing drug discovery workflows.
Q: Why is the PhSO2SCF2D reagent superior to non-deuterated analogues for drug metabolism?
A: The carbon-deuterium (C-D) bond is significantly stronger than the carbon-hydrogen (C-H) bond, often by a factor of 6 to 9. Incorporating the SCF2D group into drug candidates can drastically slow down metabolic degradation at that specific site, thereby improving the pharmacokinetic profile and half-life of the therapeutic agent without altering its biological target affinity.
Q: How does this patent address the issue of isotopic dilution in previous methods?
A: Conventional methods using reagents like TMSCF2Br often generate water as a byproduct, which leads to H/D exchange and lowers the deuterium content. This patented process utilizes a specific sequence involving sodium hydride and alkaline heavy water (D2O) in anhydrous ether, effectively suppressing proton sources and ensuring the final product maintains extremely high deuterium incorporation purity.
Q: Is the PhSO2SCF2D reagent stable for long-term storage and shipping?
A: Yes, unlike many reactive difluoromethylthiolating agents that require in-situ generation, the PhSO2SCF2D reagent described in this patent is a shelf-stable liquid. It can be stored at normal temperature and pressure without significant deterioration, making it highly suitable for global supply chains and reducing the logistical complexity for pharmaceutical manufacturers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable PhSO2SCF2D Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that high-quality deuterated intermediates play in accelerating the development of innovative medicines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation to verify isotopic enrichment and chemical purity. As a trusted partner, we understand the unique challenges of handling fluorinated and deuterated compounds and have optimized our facilities to handle these specialized chemistries safely and effectively.
We invite you to collaborate with us to optimize your supply chain for deuterated drug candidates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating how our manufacturing capabilities can reduce your overall cost of goods. We encourage you to contact us to request specific COA data for our deuterated reagents and to discuss route feasibility assessments for your target molecules. Let us help you overcome synthetic bottlenecks and bring your next-generation therapeutics to market faster.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
