Scalable Synthesis of Posaconazole Intermediate via Efficient Acylation and Methylenation Strategies
The global demand for broad-spectrum antifungal agents continues to surge, driven by the increasing prevalence of drug-resistant fungal strains and the critical need for effective treatments in immunocompromised patients. Posaconazole, a second-generation triazole antifungal, stands out as a cornerstone therapy for invasive fungal infections, necessitating a robust and efficient supply chain for its key precursors. Patent CN114409505A introduces a groundbreaking preparation method for the vital posaconazole intermediate, 2-[2-(2,4-difluorophenyl)-2-propen-1-yl]-1,3-propanediol. This technical breakthrough addresses long-standing inefficiencies in synthetic routes by leveraging a direct acylation strategy that significantly enhances yield and operational safety. As a reliable posaconazole intermediate supplier, understanding the nuances of this patented technology is essential for securing a competitive edge in the pharmaceutical market. The structural integrity and purity of this intermediate are paramount, as they directly influence the efficacy and safety profile of the final API.
![Chemical structure of 2-[2-(2,4-difluorophenyl)-2-propen-1-yl]-1,3-propanediol (Compound I)](/insights/img/posaconazole-intermediate-acylation-pharma-supplier-20260314012616-01.png)
The target molecule, depicted above, serves as a critical building block in the assembly of the posaconazole backbone. Its synthesis has historically been plagued by complex multi-step sequences involving hazardous reagents and difficult purification protocols. The innovation presented in CN114409505A simplifies this landscape by utilizing m-difluorobenzene as a readily available starting material, reacting it with 3-hydroxymethyl-4-butyrolactone. This approach not only streamlines the synthetic pathway but also aligns with modern green chemistry principles by reducing the generation of toxic byproducts. For R&D directors and procurement specialists, this represents a tangible opportunity to optimize cost structures while ensuring the consistent availability of high-purity pharmaceutical intermediates required for GMP manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this specific diol intermediate has relied on methodologies that are inherently fraught with economic and safety challenges. One prominent prior art route, disclosed in patents such as EP2789610, employs a Grignard reaction involving expensive trimethyl chloromethyl silane derivatives. 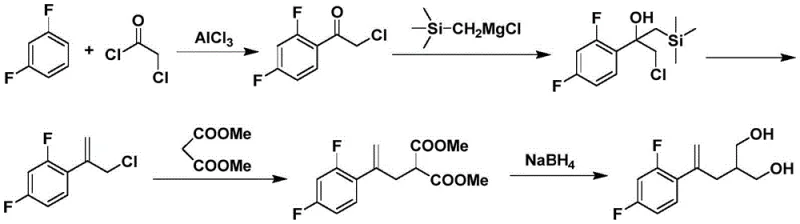
This conventional pathway, illustrated above, requires stringent anhydrous conditions and low temperatures to maintain the stability of the organometallic species, leading to high energy consumption and complex reactor requirements. Furthermore, the use of chloroacetyl chloride in related alkylation strategies introduces severe handling risks due to its strong irritation and corrosive nature, complicating waste treatment and increasing environmental compliance costs. Another existing method utilizes 1,2,3-trichloropropane for alkylation, which suffers from poor regioselectivity and necessitates harsh elimination conditions using sodium tert-butoxide. These factors collectively result in lower overall yields, often hovering around 55%, and create significant bottlenecks in the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the novel methodology outlined in CN114409505A offers a streamlined, two-step sequence that bypasses these historical hurdles. The core innovation lies in the direct Friedel-Crafts acylation of m-difluorobenzene with 3-hydroxymethyl-4-butyrolactone, followed by a carbonyl methylenation step. 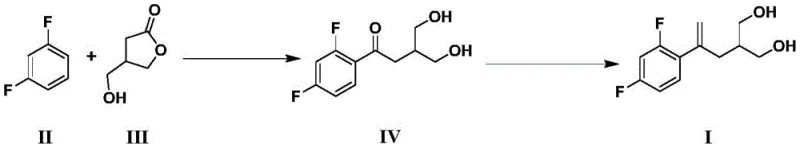
As shown in the reaction scheme above, this route eliminates the need for hazardous silane reagents and difficult Grignard preparations. Instead, it leverages the nucleophilicity of the aromatic ring and the electrophilicity of the lactone carbonyl under catalytic conditions. The subsequent methylenation converts the ketone functionality into the requisite exocyclic double bond with high fidelity. This strategic shift results in a total yield exceeding 74%, a substantial improvement over prior art. For a procurement manager, this translates to cost reduction in pharmaceutical intermediates manufacturing by minimizing raw material waste and maximizing throughput per batch. The simplicity of the operation also reduces the dependency on highly specialized labor, further driving down production costs.
Mechanistic Insights into Friedel-Crafts Acylation and Methylenation
The success of this synthetic route hinges on the precise control of the initial acylation step. In this mechanism, a Lewis acid catalyst, such as aluminum trichloride or zinc chloride, activates the carbonyl group of the 3-hydroxymethyl-4-butyrolactone. This activation generates a highly electrophilic acylium ion equivalent, which is then attacked by the electron-rich m-difluorobenzene ring. The presence of fluorine atoms on the benzene ring modulates its reactivity, requiring careful selection of catalyst strength and reaction temperature, typically between 40°C and 120°C. The choice of solvent A, ranging from dichloromethane to nitrobenzene, plays a critical role in stabilizing the transition state and solubilizing the catalyst complex. This step forms the keto-alcohol intermediate (Compound IV), which retains the hydroxymethyl group essential for the final diol structure. The high selectivity observed in this step minimizes the formation of poly-acylated byproducts, ensuring a clean reaction profile that simplifies downstream processing.
Following the isolation of Compound IV, the second critical transformation involves carbonyl methylenation. This step effectively replaces the carbonyl oxygen with a methylene group to establish the allylic system. The patent discloses the use of reagents such as methyl triphenyl phosphine salts (Wittig reagents) or Nysted reagents. When using phosphonium salts, a strong base like potassium tert-butoxide is employed to generate the reactive ylide species in situ. This ylide attacks the ketone of Compound IV, forming an oxaphosphetane intermediate that collapses to release triphenylphosphine oxide and the desired alkene. Alternatively, the use of titanium-based reagents like the Nysted reagent offers a different mechanistic pathway that can be advantageous for specific substrate scopes. The control of impurities during this elimination/addition sequence is vital, as residual phosphine oxides or titanium salts can be difficult to remove. The patented process optimizes stoichiometry and temperature (-10°C to 40°C) to drive the reaction to completion while maintaining the integrity of the sensitive diol functionality.
How to Synthesize 2-[2-(2,4-difluorophenyl)-2-propen-1-yl]-1,3-propanediol Efficiently
Implementing this synthesis requires adherence to specific operational parameters to ensure reproducibility and safety on a manufacturing scale. The process begins with the rigorous drying of solvents and reagents to prevent catalyst deactivation during the acylation phase. Following the reaction, the workup involves careful quenching into ice water to decompose the catalyst complex, followed by extraction with organic solvents like ethyl acetate or dichloromethane. The resulting organic phase must be concentrated under reduced pressure to remove residual water, a critical step before proceeding to the methylenation reaction, as moisture can quench the methylenation reagents. The detailed standardized synthesis steps, including specific molar ratios, addition rates, and purification crystallization protocols using toluene and n-heptane, are provided in the guide below.
- Perform acylation reaction between m-difluorobenzene and 3-hydroxymethyl-4-butyrolactone using a Lewis acid catalyst in solvent A at 40-120°C.
- Isolate the intermediate compound IV through extraction and concentration, ensuring water content is below 0.1%.
- React compound IV with a carbonyl methylenation reagent (e.g., methyl triphenyl phosphine salt) and an auxiliary agent in solvent B to yield the final diol product.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders, the adoption of this patented technology offers profound strategic benefits beyond mere chemical efficiency. The shift away from exotic reagents like trimethyl chloromethyl silane to commodity chemicals like m-difluorobenzene fundamentally de-risks the supply chain. m-Difluorobenzene is produced globally in massive quantities for various agrochemical and pharmaceutical applications, ensuring a stable and competitive pricing structure. This abundance mitigates the risk of supply disruptions that often plague niche fine chemical intermediates. Furthermore, the simplified reaction conditions—operating at moderate temperatures and atmospheric pressure—reduce the capital expenditure required for specialized reactor infrastructure. Facilities equipped with standard glass-lined or stainless steel reactors can easily accommodate this process, facilitating rapid technology transfer and capacity expansion without significant retrofitting costs.
- Cost Reduction in Manufacturing: The economic impact of this route is driven by the elimination of expensive stoichiometric reagents and the improvement in overall yield. By achieving yields over 74% compared to the 55% of older methods, the effective cost per kilogram of the active intermediate is drastically reduced. Additionally, the avoidance of hazardous chloroacetyl chloride removes the need for specialized scrubbing systems and expensive personal protective equipment, lowering overhead operational expenses. The use of recoverable solvents like toluene and dichloromethane further enhances the economic viability through solvent recycling loops.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures consistent batch-to-batch quality, which is critical for maintaining regulatory compliance in API production. The high purity of the crude product (often exceeding 99% after crystallization) reduces the burden on purification teams and shortens the manufacturing cycle time. This reliability allows for more accurate forecasting and inventory management, reducing the need for excessive safety stock. The simplicity of the process also means that multiple contract manufacturing organizations (CMOs) can potentially qualify the route, creating a diversified supply base that protects against single-source failures.
- Scalability and Environmental Compliance: From an environmental perspective, this process aligns with increasingly stringent global regulations regarding waste disposal and emissions. The atom economy is superior to the alkylation-elimination routes, generating less saline waste and organic sludge. The absence of heavy metal catalysts or persistent organotin/silicon byproducts simplifies wastewater treatment protocols. This 'greener' profile not only reduces disposal costs but also enhances the sustainability credentials of the final pharmaceutical product, a factor of growing importance to end-users and regulatory bodies alike.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims within patent CN114409505A, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their existing supply chains.
Q: What are the primary advantages of the new acylation route over traditional Grignard methods?
A: The new route eliminates the need for expensive and hazardous trimethyl chloromethyl silane and strict Grignard conditions. It utilizes cheap m-difluorobenzene and achieves a total yield over 74%, significantly higher than the 55% yield of conventional alkylation methods.
Q: Which catalysts are suitable for the initial acylation step?
A: The patent specifies a range of effective catalysts including aluminum trichloride, ferric trichloride, zinc chloride, titanium tetrachloride, and even protic acids like sulfuric acid, offering flexibility for process optimization.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrialization with simple operations, safe reaction conditions, and the use of common solvents like dichloromethane and toluene, facilitating easy scale-up and waste management.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Posaconazole Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires more than just chemical knowledge; it demands engineering excellence and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields reported in CN114409505A are realized in our manufacturing facilities. We have invested heavily in rigorous QC labs and analytical capabilities to meet stringent purity specifications, guaranteeing that every batch of posaconazole intermediate we deliver meets the exacting standards required for antifungal API synthesis. Our infrastructure is designed to handle the specific solvent systems and thermal profiles required by this acylation-methylenation sequence safely and efficiently.
We invite pharmaceutical partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized process parameters and bulk purchasing power for raw materials, we can offer a compelling value proposition that enhances your bottom line. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Whether you are looking to secure a secondary source for risk mitigation or seeking a primary partner for long-term supply, our dedicated account managers are ready to discuss how we can support your posaconazole production goals with reliability and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
