Advanced Manufacturing of Dydrogesterone via Streamlined Deprotection and Isomerization for Global Supply
The pharmaceutical landscape for hormonal therapies continues to demand robust, scalable, and high-purity synthetic routes for critical active pharmaceutical ingredients, specifically Dydrogesterone, a potent oral progestogen widely utilized for treating endogenous progesterone deficiencies. Patent CN114437164A, published in May 2022, introduces a transformative preparation method that addresses the longstanding inefficiencies associated with traditional Dydrogesterone manufacturing. This innovation pivots away from the convoluted, low-yield pathways of the past, such as those disclosed in CN110818760A which relied on cumbersome bromination and photochemical ring-opening sequences, and instead proposes a streamlined approach centered on the acid-catalyzed deprotection of 3,20-bis(ethyldioxy)-9beta-10alpha-pregna-5,7-diene followed by a precise isomerization step. For R&D directors and procurement specialists alike, this patent represents a significant leap forward in process chemistry, offering a pathway that not only simplifies the operational workflow but also drastically enhances the economic viability of producing this high-value hormone. The core breakthrough lies in the meticulous control of reaction conditions, particularly the molar ratios of acid-alcohol solvents and the specific thermal parameters during the transposition phase, which collectively ensure a reproducible yield between 44.5% and 46% while maintaining exceptional chemical purity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Dydrogesterone has been plagued by excessive step counts and reliance on hazardous or difficult-to-control reaction types, creating substantial bottlenecks for supply chain managers and increasing the cost of goods sold for pharmaceutical manufacturers. Prior art methods often necessitated starting from progesterone and proceeding through a gauntlet of carbon-based protection, bromination, elimination, and critically, a light-induced ring-opening step that requires specialized photochemical reactors and poses significant safety and scalability challenges. These conventional routes are inherently inefficient, suffering from low overall yields due to the accumulation of losses at each discrete transformation stage, and they frequently generate complex impurity profiles that are difficult to purge without expensive chromatographic purification. Furthermore, the use of multiple distinct solvent systems and harsh reagents in these older methodologies complicates waste management and environmental compliance, forcing manufacturers to invest heavily in effluent treatment infrastructure. The cumulative effect of these limitations is a fragile supply chain where lead times are extended, production costs are inflated, and the consistency of the final API quality is perpetually at risk due to the sensitivity of photochemical and multi-step sequences.
The Novel Approach
In stark contrast to these legacy processes, the methodology outlined in CN114437164A employs a direct and elegant strategy that leverages the stability of the 3,20-bis(ethyldioxy)-9beta-10alpha-pregna-5,7-diene precursor to bypass the need for complex functional group manipulations. By utilizing a carefully prepared acid-alcohol solution—formed by introducing hydrogen chloride into methanol, ethanol, or isopropanol—the process effects a clean deprotection of the ketal groups under mild conditions, immediately generating the requisite diketone intermediate without the formation of significant byproducts. This intermediate is then subjected to a thermodynamic transposition in a hydrophobic solvent like toluene, catalyzed by acetic acid under reflux, which efficiently rearranges the diene system from the 5,7-position to the biologically active 4,6-position characteristic of Dydrogesterone. This novel approach eliminates the need for photochemical equipment entirely, replacing it with standard heating and stirring operations that are universally available in chemical manufacturing facilities, thereby reducing capital expenditure and operational complexity. The result is a synthesis that is not only shorter and safer but also demonstrably more robust, capable of delivering high-purity product through simple crystallization techniques rather than relying on resource-intensive purification methods.
Mechanistic Insights into Acid-Catalyzed Deprotection and Isomerization
The chemical elegance of this process is rooted in the precise mechanistic control exerted during the deprotection and subsequent isomerization phases, which are critical for ensuring the stereochemical integrity and purity of the final Dydrogesterone molecule. The initial deprotection step involves the protonation of the ethyldioxy groups on the steroid backbone by the hydrogen chloride-alcohol complex, facilitating the hydrolysis of the ketals to reveal the 3,20-dione functionality while preserving the sensitive 9-beta, 10-alpha stereochemistry of the A/B ring junction. Following neutralization with alkaline water, which precipitates the intermediate and removes acidic residues, the material undergoes a thermodynamically driven isomerization where the conjugated diene system migrates from the 5,7-positions to the 4,6-positions. This transposition is catalyzed by acetic acid in a hydrophobic medium, likely proceeding through an enol or enolate intermediate that allows for the equilibration of the double bonds to the more stable conjugated position found in the target API. Understanding this mechanism is vital for R&D teams, as it highlights the importance of maintaining strict anhydrous conditions during the reflux stage to prevent hydration side reactions, and underscores the necessity of precise solvent ratios to drive the equilibrium towards the desired product.
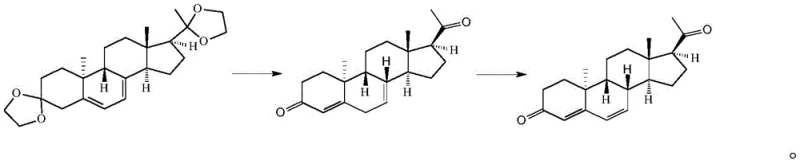
Furthermore, the control of impurities in this synthesis is achieved through a combination of kinetic control during the reaction and thermodynamic control during the crystallization workup. The patent data indicates that deviations in solvent molar ratios, particularly using less than the specified 30-40 equivalents of hydrophilic solvent during crystallization, can lead to incomplete precipitation or the co-crystallization of impurities, whereas excess solvent can paradoxically increase impurity levels by solubilizing unwanted byproducts. The chromatographic data provided in the patent examples demonstrates the efficacy of this control, showing a dominant product peak with an area percentage exceeding 99.9%, with trace impurities such as Impurity A and Impurity B remaining well below detection limits or at negligible levels of 0.01%. This high level of purity is a direct consequence of the selective nature of the acid-catalyzed transposition, which avoids the radical mechanisms associated with bromination and photolysis that typically generate a wide array of structural analogs and degradation products. For quality assurance professionals, this mechanistic clarity offers confidence that the process is robust against minor variations in raw material quality, provided the critical process parameters regarding temperature and stoichiometry are strictly adhered to.
How to Synthesize Dydrogesterone Efficiently
To implement this advanced synthesis route effectively, manufacturing teams must adhere to a standardized protocol that prioritizes the precise preparation of the acid-alcohol catalyst and the controlled addition of reagents to manage exotherms and crystallization kinetics. The process begins with the generation of the acidic medium, followed by the introduction of the steroid precursor, and concludes with a rigorous workup involving neutralization, extraction, and sequential crystallizations to ensure maximum recovery and purity. While the general outline is straightforward, the devil is in the details, specifically regarding the timing of ammonia water addition and the duration of the reflux period, which are critical for optimizing the yield within the reported 44.5% to 46% range. Detailed standard operating procedures for this specific transformation are essential for technology transfer, ensuring that the nuances of the transposition step are captured accurately at scale.
- Prepare an acid-alcohol solution by introducing hydrogen chloride into a solvent such as methanol, ethanol, or isopropanol, then react with 3,20-bis(ethyldioxy)-9beta-10alpha-pregna-5,7-diene to effect deprotection.
- Neutralize the reaction mixture with alkaline water to crystallize and isolate the deprotected intermediate, ensuring removal of acidic residues before the next stage.
- Reflux the intermediate in a hydrophobic solvent like toluene with acetic acid to catalyze the transposition of the diene system, followed by extraction and crystallization to obtain pure Dydrogesterone.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers profound advantages for procurement managers and supply chain heads who are tasked with reducing the total cost of ownership for hormonal APIs while ensuring uninterrupted supply. The elimination of photochemical steps and bromination reagents removes significant cost drivers associated with specialized equipment maintenance, hazardous waste disposal, and the procurement of expensive, light-sensitive reagents. By shifting to a thermal process driven by commodity chemicals like acetic acid, toluene, and ethanol, the manufacturing footprint becomes significantly leaner, allowing for faster batch turnover and reduced cycle times which directly translates to improved cash flow and inventory management. Moreover, the simplicity of the workup, which relies on standard liquid-liquid extraction and crystallization rather than complex chromatography, drastically reduces solvent consumption and processing time, leading to substantial operational cost savings that can be passed down the supply chain. This efficiency makes the process highly attractive for large-scale production, where even marginal improvements in throughput and yield can result in significant financial benefits over the lifecycle of the product.
- Cost Reduction in Manufacturing: The primary driver for cost reduction in this process is the complete removal of the photochemical ring-opening step, which traditionally requires expensive UV reactors and consumes significant energy, alongside the elimination of bromination reagents that generate costly halogenated waste streams. By replacing these with a simple acid-catalyzed reflux in toluene, the process utilizes low-cost, high-volume solvents and reagents that are readily available on the global market, insulating the supply chain from price volatility associated with specialty chemicals. Additionally, the high purity achieved directly from crystallization minimizes the need for downstream purification steps such as preparative HPLC or repeated recrystallizations, further lowering the variable costs per kilogram of produced API. This streamlined approach ensures that the cost of goods sold is optimized, providing a competitive edge in the pricing of the final pharmaceutical formulation.
- Enhanced Supply Chain Reliability: Supply chain reliability is significantly bolstered by the use of robust, non-hazardous reagents and the avoidance of light-sensitive intermediates that require cold chain logistics or dark storage conditions. The starting material, 3,20-bis(ethyldioxy)-9beta-10alpha-pregna-5,7-diene, is stable and can be sourced or synthesized in bulk without the degradation risks associated with the unstable intermediates found in older pathways. Furthermore, the process tolerance to slight variations in reaction time and temperature, as evidenced by the consistent yields across different examples in the patent, reduces the risk of batch failures and production delays. This robustness ensures a steady flow of material to downstream formulation units, mitigating the risk of stockouts that can occur with more finicky synthetic routes.
- Scalability and Environmental Compliance: Scalability is inherent in this design, as the unit operations—neutralization, reflux, extraction, and crystallization—are standard in almost every pharmaceutical manufacturing facility, requiring no bespoke engineering or retrofits. From an environmental standpoint, the process generates significantly less hazardous waste compared to bromination-based routes, as it avoids the production of heavy metal salts or halogenated organic byproducts that are difficult to treat. The use of recyclable solvents like toluene and ethanol further aligns with green chemistry principles, facilitating easier regulatory approval and reducing the environmental compliance burden on the manufacturing site. This ease of scale-up from pilot plant to commercial tonnage ensures that supply can be rapidly ramped up to meet market demand without the long lead times associated with installing specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Dydrogesterone synthesis route, drawing directly from the experimental data and claims presented in the patent documentation. These insights are designed to clarify the operational benefits and technical feasibility for stakeholders evaluating this technology for potential licensing or contract manufacturing agreements. Understanding these specifics is crucial for making informed decisions about integrating this process into existing production portfolios.
Q: What are the critical yield parameters for this Dydrogesterone synthesis?
A: The patented process achieves a consistent yield range of 44.5% to 46% by strictly controlling solvent molar ratios and crystallization times, significantly outperforming traditional multi-step routes that suffer from cumulative yield losses.
Q: How does this method improve impurity profiles compared to prior art?
A: By utilizing a direct deprotection and isomerization strategy rather than complex bromination and photochemical ring-opening steps, the process minimizes side reactions, resulting in a final product with purity exceeding 99.9% as verified by HPLC analysis.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the method relies on standard unit operations such as reflux, neutralization, and crystallization using common solvents like toluene and ethanol, making it highly amenable to scale-up from kilogram to multi-ton commercial manufacturing without specialized photochemical equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dydrogesterone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting advanced synthetic routes like the one described in CN114437164A to secure a competitive advantage in the global hormonal therapy market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific solvent systems and thermal requirements of this deprotection-isomerization sequence, guaranteeing stringent purity specifications and rigorous QC labs that validate every batch against the highest international standards. We are committed to delivering high-purity Dydrogesterone that meets the exacting needs of pharmaceutical formulators, leveraging our technical expertise to optimize yield and minimize impurities.
We invite procurement leaders and R&D directors to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic benefits and operational efficiencies this method offers for your specific production volume. We encourage you to reach out for specific COA data and route feasibility assessments to verify the compatibility of this process with your current quality systems and to explore opportunities for long-term collaboration in the supply of high-quality pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
