Advanced Copper-Catalyzed Synthesis of N-Boc-trans-cyclohexanediamine for Commercial Scale-up
The pharmaceutical and fine chemical industries continuously seek robust synthetic routes for protected diamines, which serve as critical building blocks for peptide mimetics and polymer initiators. Patent CN113493398B introduces a significant advancement in the preparation of N-Boc-trans-cyclohexanediamine, addressing long-standing challenges in selectivity and raw material utilization. This innovative methodology utilizes a copper-catalyzed oxidation system with tert-butyloxycarbonyl hydrazine and hydrogen peroxide, offering a distinct alternative to traditional Boc-protection strategies. By shifting the paradigm from direct Boc-anhydride protection to an oxidative coupling mechanism, the process achieves superior control over mono-protection, minimizing the formation of di-protected byproducts that complicate downstream purification. For R&D directors and procurement specialists, this represents a tangible opportunity to enhance the efficiency of pharmaceutical intermediate supply chains while maintaining rigorous purity standards required for GMP manufacturing environments.
The strategic value of this patent lies in its ability to maximize the conversion of the valuable diamine starting material, which is often the cost-driving component in the synthesis. Traditional methods frequently rely on a large excess of the diamine to suppress di-protection, leading to significant economic loss and complex recovery processes. In contrast, the disclosed invention employs a stoichiometric approach where the oxidant and catalyst drive the reaction towards the desired mono-Boc product with high fidelity. This technical breakthrough not only improves the overall atom economy but also simplifies the work-up procedure, making it an attractive candidate for commercial scale-up of complex pharmaceutical intermediates. As we delve deeper into the mechanistic and operational details, the potential for cost reduction in pharmaceutical intermediate manufacturing becomes increasingly evident through the elimination of wasteful reagent usage and streamlined processing steps.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of mono-Boc protected cyclohexanediamines has relied heavily on the reaction between the diamine and di-tert-butyl dicarbonate (Boc2O) under alkaline conditions. While conceptually straightforward, this conventional approach suffers from inherent selectivity issues that plague large-scale production. To achieve acceptable yields of the mono-protected species, manufacturers are often forced to use a substantial excess of the diamine starting material, sometimes in a molar ratio that renders the process economically inefficient. This excess material does not simply disappear; it remains in the reaction mixture, requiring energy-intensive distillation or crystallization steps to recover and recycle, which adds to the operational expenditure. Furthermore, the formation of the di-Boc impurity is difficult to suppress completely, often resulting in product streams that require extensive chromatographic purification, a step that is notoriously difficult to scale and expensive to operate in a commercial setting. The reliance on Boc2O also introduces safety and handling concerns, as the reagent can be unstable under certain conditions and generates tert-butanol and CO2 as byproducts, complicating the reaction profile.
The Novel Approach
The methodology disclosed in CN113493398B circumvents these traditional bottlenecks by employing a catalytic oxidative coupling strategy. Instead of using Boc2O, the process utilizes tert-butyloxycarbonyl hydrazine as the source of the Boc group, activated by a copper catalyst and hydrogen peroxide. This shift in chemistry allows for a much more precise control over the reaction trajectory, favoring the formation of the mono-protected product while inherently suppressing the di-protected species. The reaction operates under mild conditions, typically between 10-15°C, which reduces the thermal load on the reactor and minimizes the risk of thermal runaway, a critical safety consideration for reliable pharmaceutical intermediate supplier operations. By optimizing the equivalent weight of hydrogen peroxide and the concentration of the solvent, the process ensures that the raw material cyclohexanediamine is fully consumed, eliminating the need for costly excesses. This novel approach not only enhances the chemical yield but also significantly simplifies the downstream processing, as the impurity profile is cleaner and more manageable, directly translating to improved operational efficiency and reduced production costs.
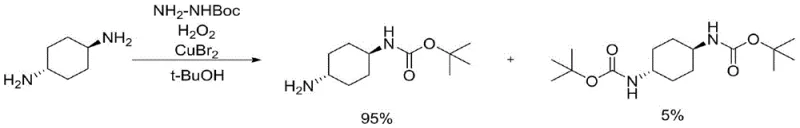
Mechanistic Insights into Copper-Catalyzed Oxidative Coupling
The core of this technological advancement lies in the unique catalytic cycle driven by the copper species. The proposed mechanism involves the oxidation of the copper catalyst, such as copper bromide or copper chloride, by hydrogen peroxide to generate a higher oxidation state species capable of activating the hydrazine derivative. This activated complex captures a hydrogen molecule from the tert-butylhydrazine, generating a diazo precursor that subsequently loses nitrogen to form a carbonyl radical. This radical species then undergoes a single-electron oxidation addition with the substrate, facilitated by the copper catalyst, to install the mono-tert-butoxycarbonyl protecting group. The cycle is completed by the reduction of the copper species back to its active state, allowing the catalytic turnover to continue efficiently. This mechanistic pathway is distinct from nucleophilic substitution mechanisms seen with Boc2O, offering a different selectivity profile that is less prone to over-protection. Understanding this cycle is crucial for R&D teams looking to replicate or optimize the process, as it highlights the importance of maintaining the correct oxidation potential through controlled addition of the oxidant.
Impurity control is a paramount concern in the synthesis of high-purity pharmaceutical intermediates, and this patent offers a sophisticated solution through kinetic control. The formation of the di-tert-butoxycarbonyl protecting group is effectively reduced to a range of 2-5% of the reaction system by strictly managing the reaction parameters. The electron-donating nature of the amino group in the mono-protected intermediate makes it less reactive towards the second protection step under these specific oxidative conditions, providing a natural selectivity filter. By maintaining the reaction temperature at 10-15°C and controlling the dropping speed of hydrogen peroxide, the concentration of the active radical species is kept at a level that favors the initial protection without driving the second substitution. This precise control minimizes the burden on the purification team, as the di-Boc impurity can be easily separated during the acidic extraction phase, where it remains in the organic layer while the desired mono-Boc product is retained in the aqueous phase. This level of impurity management is essential for meeting the stringent quality specifications required by downstream drug substance manufacturers.
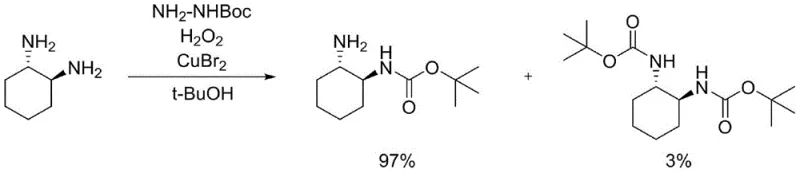
How to Synthesize N-Boc-trans-cyclohexanediamine Efficiently
Implementing this synthesis route requires careful attention to the addition rates and temperature profiles to ensure safety and reproducibility. The process begins with the dissolution of the diamine and hydrazine reagent in a solvent like tert-butanol, followed by the slow introduction of the oxidant. The detailed standardized synthesis steps involve specific quenching and pH adjustment protocols that are critical for isolating the product in high purity. For a comprehensive guide on the exact operational parameters, including stirring speeds and specific work-up sequences, please refer to the technical documentation provided below.
- Dissolve trans-cyclohexanediamine, tert-butyloxycarbonyl hydrazine, and a copper catalyst in an organic solvent such as tert-butanol.
- Add hydrogen peroxide dropwise at 10-15°C while maintaining strict temperature control to manage reaction exotherm.
- Quench with sodium sulfite, adjust pH for extraction, and purify via slurry with N-heptane to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this copper-catalyzed route offers substantial benefits for procurement and supply chain management teams looking to optimize their sourcing strategies. The primary advantage stems from the significant reduction in raw material waste, as the process eliminates the need for excessive diamine usage that characterizes conventional methods. This efficiency gain directly translates to cost reduction in pharmaceutical intermediate manufacturing, allowing for more competitive pricing structures without compromising on quality. Furthermore, the use of common and readily available reagents such as hydrogen peroxide and copper salts ensures a stable supply chain, reducing the risk of disruptions associated with specialized or hazardous reagents. The mild reaction conditions also contribute to lower energy consumption and reduced wear on manufacturing equipment, enhancing the overall sustainability and longevity of the production assets.
- Cost Reduction in Manufacturing: The elimination of excess diamine usage represents a major cost saving opportunity, as the diamine is typically the most expensive component in the reaction mixture. By achieving full conversion of the starting material, the process maximizes the value derived from each kilogram of raw material purchased. Additionally, the simplified work-up procedure reduces the consumption of solvents and auxiliary chemicals required for purification, further lowering the variable costs associated with production. The reduction in di-Boc impurity also minimizes the yield loss typically associated with aggressive purification steps, ensuring that more of the theoretical yield is captured as saleable product. These factors combine to create a leaner, more cost-effective manufacturing process that can withstand market fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: The reliance on stable and commoditized chemicals like hydrogen peroxide and copper bromide enhances the resilience of the supply chain against geopolitical or logistical disruptions. Unlike specialized protecting group reagents that may have limited suppliers, the key inputs for this process are widely available from multiple global sources, ensuring continuity of supply. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, reducing the need for stringent incoming quality control that can delay production starts. This reliability is crucial for maintaining consistent delivery schedules to downstream customers, fostering trust and long-term partnerships in the competitive pharmaceutical intermediate market.
- Scalability and Environmental Compliance: The mild temperature profile and the use of less hazardous oxidants make this process highly scalable from pilot plant to commercial production volumes. The reduced generation of hazardous waste, particularly the avoidance of large quantities of unreacted amines and Boc-anhydride byproducts, simplifies environmental compliance and waste disposal procedures. This alignment with green chemistry principles not only reduces regulatory burdens but also enhances the corporate sustainability profile of the manufacturer. The ability to scale this process efficiently ensures that reducing lead time for high-purity pharmaceutical intermediates is achievable without compromising on safety or environmental standards, meeting the growing demand for sustainable manufacturing practices in the industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the patent specifications and are intended to provide clarity on the process capabilities and limitations. Understanding these details is essential for making informed decisions about technology transfer and procurement strategies.
Q: How does this method control di-Boc impurity formation?
A: By precisely controlling the equivalent weight of hydrogen peroxide and reaction temperature, the formation of the di-tert-butoxycarbonyl protecting group is limited to 2-5% of the reaction system.
Q: What catalysts are suitable for this oxidation process?
A: The patent specifies copper bromide or copper chloride as effective catalysts for facilitating the single-electron oxidation addition required for mono-protection.
Q: Is this process scalable for industrial production?
A: Yes, the mild reaction conditions (10-15°C) and use of common solvents like tert-butanol or acetone support safe commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Boc-trans-cyclohexanediamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory to plant is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which are equipped with state-of-the-art analytical instrumentation to verify every batch. Our dedication to technical excellence means that we can adapt the patented copper-catalyzed route to meet your specific volume requirements while maintaining the highest standards of quality and consistency.
We invite you to collaborate with us to optimize your supply chain and achieve your cost targets without sacrificing quality. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating how this advanced synthesis route can benefit your bottom line. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to evaluate the potential of this technology for your applications with confidence. By partnering with us, you gain access to a reliable source of high-purity pharmaceutical intermediates backed by decades of industry expertise and a commitment to innovation.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
