Advanced Silver-Catalyzed Synthesis of p-Hydroxyphenylhydrazine Compounds for Commercial Scale-Up
Advanced Silver-Catalyzed Synthesis of p-Hydroxyphenylhydrazine Compounds for Commercial Scale-Up
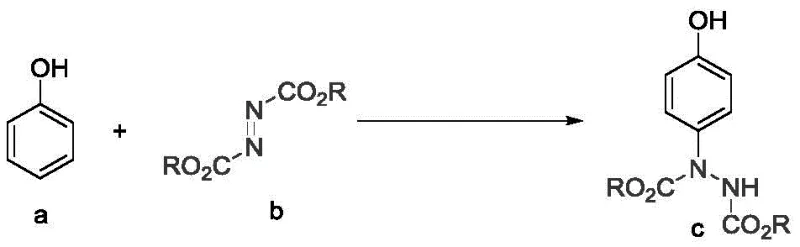
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to functionalize phenolic scaffolds with high precision, particularly for the synthesis of critical intermediates like p-hydroxyphenylhydrazine derivatives. A significant technological breakthrough in this domain is documented in Chinese Patent CN111362842B, published in April 2022, which introduces a highly efficient preparation method for p-hydroxyphenylhydrazine compounds. This patent outlines a novel transition metal-catalyzed protocol that utilizes silver oxide (Ag2O) to achieve direct para-carbon-hydrogen bond amination of phenolic compounds. For R&D directors and process chemists, this represents a paradigm shift from traditional electrophilic substitutions, offering a pathway to access valuable p-aminophenol precursors with unprecedented regioselectivity. The methodology is not merely a laboratory curiosity but a scalable process designed for industrial viability, utilizing inexpensive raw materials and operating under remarkably mild conditions.
The strategic importance of this technology lies in its ability to bypass the inherent difficulties associated with phenol functionalization. Phenols are ubiquitous building blocks in drug discovery, yet controlling the site of substitution is notoriously challenging due to the similar electron densities at the ortho and para positions. The invention detailed in CN111362842B solves this by leveraging the unique coordination properties of silver catalysts in an aqueous environment. This approach ensures that the nitrogen source, typically an azodicarboxylate, attacks exclusively at the para-position, thereby streamlining the purification workflow and maximizing atom economy. As a reliable p-hydroxyphenylhydrazine supplier, understanding these mechanistic nuances is crucial for delivering high-purity materials that meet the stringent specifications of global pharmaceutical clients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of phenolic compounds has been plagued by issues of regiocontrol and environmental impact. Traditional electrophilic aromatic substitution reactions often result in complex mixtures of ortho and para isomers, necessitating energy-intensive separation techniques such as repeated crystallization or preparative chromatography to isolate the desired para-product. Furthermore, many existing protocols rely on harsh Lewis acid catalysts or require the installation and subsequent removal of bulky directing groups to force ortho-selectivity, which adds unnecessary synthetic steps and reduces overall yield. When attempting para-functionalization without protection, the reactivity of the free hydroxyl group often leads to side reactions or polymerization, especially under acidic or oxidative conditions. These inefficiencies translate directly into higher manufacturing costs and longer lead times, creating significant bottlenecks for procurement managers tasked with securing cost reduction in pharmaceutical intermediates manufacturing. Additionally, the reliance on volatile organic solvents in conventional methods poses safety hazards and complicates waste management, conflicting with modern sustainability goals.
The Novel Approach
In stark contrast, the method disclosed in CN111362842B offers a streamlined, green alternative that addresses these痛点 directly. By employing a silver oxide catalyst in a water-based system, the process achieves excellent para-selectivity without the need for protecting groups or exotic reagents. The reaction proceeds smoothly at low temperatures, typically in an ice bath, which preserves the integrity of sensitive functional groups and minimizes thermal runaway risks. As illustrated in the specific example of diethyl 1-(4-hydroxyphenyl)hydrazine-1,2-dicarboxylate synthesis, the process delivers impressive yields, such as the reported 91% isolated yield in Example 1, demonstrating its practical efficiency.
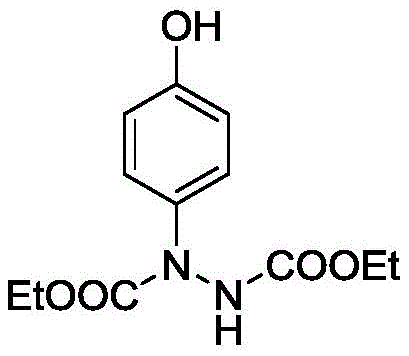
This high level of performance is achieved using readily available starting materials like phenol and diethyl azodicarboxylate, ensuring a stable supply chain. The use of water as the solvent not only reduces costs but also simplifies the workup procedure, as the product can be easily extracted into an organic phase while inorganic salts remain in the aqueous layer. This novel approach effectively transforms a complex multi-step synthesis into a single-pot operation, significantly enhancing the commercial scale-up of complex hydrazine derivatives.
Mechanistic Insights into Ag2O-Catalyzed Para-Selective Amination
The core innovation of this technology resides in the unique interaction between the silver catalyst and the phenolic substrate. Unlike traditional electrophilic substitutions that rely solely on electronic activation, the silver oxide likely acts as a soft Lewis acid that coordinates with the phenolic oxygen or potentially activates the azodicarboxylate species. This coordination creates a transient supramolecular assembly that directs the incoming nitrogen nucleophile specifically to the para-position of the aromatic ring. The steric bulk of the catalyst-substrate complex may further disfavor attack at the ortho-position, thereby enforcing high regioselectivity. Understanding this mechanism is vital for process optimization, as it explains why the reaction tolerates various substituents on the azodicarboxylate, ranging from ethyl and isopropyl to tert-butyl and benzyl esters, as evidenced by the diverse scope presented in the patent examples.
From an impurity control perspective, the aqueous nature of the reaction medium plays a pivotal role. Water acts not just as a solvent but potentially as a proton shuttle that facilitates the rearomatization step of the C-H activation cycle. This rapid turnover prevents the accumulation of reactive intermediates that could otherwise lead to oligomerization or over-oxidation byproducts. Consequently, the crude reaction mixture is remarkably clean, allowing for straightforward purification via standard column chromatography or even crystallization in optimized large-scale batches. For quality assurance teams, this means that achieving high-purity p-hydroxyphenylhydrazine is more predictable and reproducible, reducing the risk of batch failures and ensuring consistent supply continuity for downstream API synthesis.
How to Synthesize p-Hydroxyphenylhydrazine Efficiently
The operational simplicity of this silver-catalyzed protocol makes it highly attractive for technology transfer from the laboratory to pilot plant scales. The procedure involves a sequential addition of reagents into a reactor containing the catalyst and solvent, followed by a controlled reaction period under cooling. The robustness of the method is highlighted by the patent's claim that scaling the dosage of reactants by 100 times does not adversely affect the yield, a critical indicator of industrial feasibility. Detailed standardized synthesis steps are provided below to guide process engineers in replicating these results.
- Charge the reactor with Ag2O catalyst (1-5 mol%), phenol substrate, azodicarboxylate reactant (1: 2 ratio), and water solvent.
- Maintain the reaction mixture in an ice bath with stirring for 0.5 to 10 hours to ensure high regioselectivity.
- Extract the aqueous reaction mixture with ethyl acetate, concentrate under reduced pressure, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic benefits beyond mere technical elegance. The shift towards a water-based, silver-catalyzed process fundamentally alters the cost structure of producing p-hydroxyphenylhydrazine intermediates. By eliminating the need for expensive organic solvents and reducing the complexity of purification, the overall cost of goods sold (COGS) is significantly optimized. Furthermore, the mild reaction conditions reduce energy consumption associated with heating and cooling, contributing to a lower carbon footprint and aligning with corporate sustainability mandates. These factors combined make the technology a compelling choice for long-term sourcing strategies focused on resilience and efficiency.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the use of commodity chemicals and the elimination of costly purification steps. Since the reaction exhibits high selectivity, the formation of ortho-isomers is negligible, meaning that valuable raw materials are not wasted on unwanted byproducts that must be separated and discarded. Additionally, the catalyst loading is low, typically between 1 to 5 mol%, and silver oxide is a relatively inexpensive transition metal source compared to noble metals like palladium or rhodium. This combination of high atom economy and low catalyst cost results in substantial cost savings in pharmaceutical intermediates manufacturing, allowing buyers to negotiate more favorable pricing tiers without compromising on quality.
- Enhanced Supply Chain Reliability: Supply chain stability is often threatened by the reliance on specialized reagents with long lead times. This method mitigates such risks by utilizing phenol and azodicarboxylates, which are produced globally in massive quantities for various industrial applications. The availability of these starting materials ensures that production schedules are not disrupted by raw material shortages. Moreover, the simplicity of the reaction setup means that manufacturing can be distributed across multiple facilities with standard glass-lined or stainless-steel reactors, reducing the dependency on single-source suppliers and enhancing the overall resilience of the supply network for high-purity phenolic amines.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges, but the patent data indicates that this reaction maintains its efficiency even when scaled up by two orders of magnitude. The use of water as a solvent simplifies waste treatment, as aqueous effluents are easier to treat than halogenated organic waste streams. This ease of handling hazardous materials reduces the regulatory burden and insurance costs associated with chemical manufacturing. Consequently, the process is well-suited for commercial scale-up of complex hydrazine derivatives, enabling manufacturers to rapidly increase capacity to meet surging market demand while maintaining strict adherence to environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this silver-catalyzed synthesis route. These insights are derived directly from the experimental data and claims within patent CN111362842B, providing a transparent view of the technology's capabilities and limitations for potential partners.
Q: What is the primary advantage of the Ag2O catalytic system over traditional Lewis acids?
A: Unlike traditional Lewis acids which often yield mixtures of ortho and para isomers, the Ag2O catalytic system described in CN111362842B achieves exceptional para-selectivity, eliminating the need for difficult separation processes and improving overall material efficiency.
Q: Is this synthesis method considered environmentally friendly?
A: Yes, the process utilizes water as the primary reaction solvent instead of volatile organic compounds (VOCs), aligning with green chemistry principles and significantly reducing the environmental footprint associated with solvent disposal and recovery.
Q: What are the typical reaction conditions required for this transformation?
A: The reaction operates under mild conditions, specifically requiring an ice bath temperature range and short reaction times of 0.5 to 10 hours, which minimizes energy consumption and thermal degradation of sensitive functional groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Hydroxyphenylhydrazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the silver-catalyzed amination technology described in CN111362842B for the production of high-value pharmaceutical intermediates. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch. By integrating this green and efficient synthesis route into our portfolio, we can offer our clients a competitive edge through superior product quality and consistent availability.
We invite global pharmaceutical and agrochemical companies to collaborate with us to leverage this advanced manufacturing capability. Whether you require custom synthesis of specific hydrazine derivatives or large-scale supply of standard intermediates, our technical procurement team is ready to assist. Please contact us today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to reach out for specific COA data and route feasibility assessments to see how our expertise can accelerate your drug development timeline and optimize your supply chain costs.
