Optimizing Bicalutamide Intermediate Production via Novel Trifluoromethyl Benzonitrile Synthesis
The global demand for high-performance oncology therapeutics continues to drive rigorous innovation in the synthesis of key pharmaceutical intermediates. Specifically, the production of 4-amino-2-trifluoromethyl benzonitrile, a critical building block for the anti-androgen drug Bicalutamide, has historically faced significant bottlenecks regarding yield consistency and environmental compliance. Patent CN1810775A introduces a transformative three-step methodology that fundamentally restructures the synthetic pathway, shifting away from hazardous reduction protocols toward efficient nucleophilic aromatic substitutions. This technical breakthrough not only addresses the escalating regulatory pressures on waste management but also provides a robust framework for achieving commercial-grade purity specifications essential for modern API manufacturing. For R&D directors and procurement strategists, understanding the mechanistic advantages of this route is paramount for securing a competitive edge in the supply of high-value fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for generating 4-amino-2-trifluoromethyl benzonitrile have long been plagued by operational inefficiencies and severe environmental drawbacks that hinder large-scale industrial adoption. As illustrated in the reaction scheme below, the legacy process typically initiates with o-trifluoromethyl aniline, necessitating a cumbersome four-step sequence involving diazotization, nitration, amino displacement, and finally, reduction using iron powder. 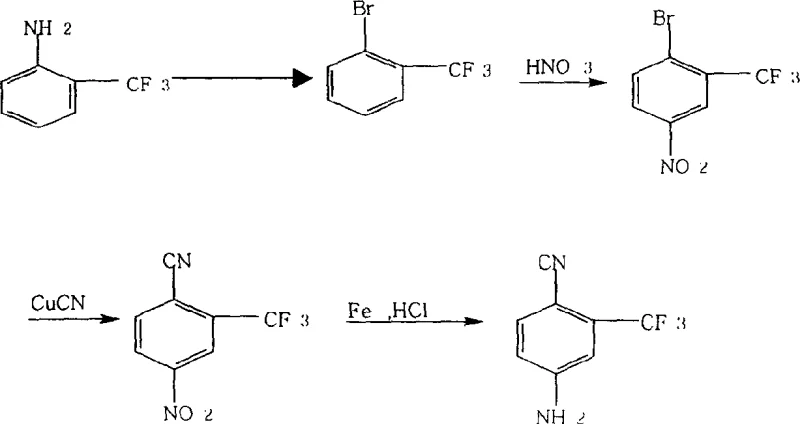 This reliance on stoichiometric iron powder reduction creates substantial quantities of hazardous solid waste, commonly referred to as iron mud, which requires complex and costly disposal procedures. Furthermore, the extraction of the target compound from the reaction mixture is notoriously difficult due to emulsification and product loss, often requiring five to six repetitive extraction cycles that drastically consume organic solvents. The cumulative effect of these inefficient unit operations results in a dismal overall yield of approximately 30%, alongside inconsistent product color and purity profiles that fail to meet the stringent requirements of top-tier pharmaceutical clients.
This reliance on stoichiometric iron powder reduction creates substantial quantities of hazardous solid waste, commonly referred to as iron mud, which requires complex and costly disposal procedures. Furthermore, the extraction of the target compound from the reaction mixture is notoriously difficult due to emulsification and product loss, often requiring five to six repetitive extraction cycles that drastically consume organic solvents. The cumulative effect of these inefficient unit operations results in a dismal overall yield of approximately 30%, alongside inconsistent product color and purity profiles that fail to meet the stringent requirements of top-tier pharmaceutical clients.
The Novel Approach
In stark contrast, the patented methodology delineated in CN1810775A offers a streamlined, three-step alternative that begins with the readily available m-trifluoromethyl fluorobenzene, effectively bypassing the need for hazardous diazotization and metal reduction entirely. This novel approach leverages a strategic sequence of regioselective bromination, copper-catalyzed cyanation, and high-pressure aminolysis to construct the target molecular architecture with exceptional precision. By utilizing dibromohydantoin for bromination and cuprous cyanide in a quinoline medium for cyanation, the process ensures high conversion rates while minimizing the formation of troublesome by-products. The elimination of the iron reduction step not only removes a major source of heavy metal contamination but also simplifies the downstream purification workflow, allowing for the direct isolation of the product through straightforward crystallization or distillation. Consequently, this modernized route delivers a total yield ranging from 73% to 75%, representing a more than twofold improvement in material efficiency compared to conventional techniques.
Mechanistic Insights into Regioselective Bromination and Cyanation
The success of this synthetic strategy hinges on the precise control of regioselectivity during the initial functionalization of the aromatic ring, which dictates the purity of all subsequent intermediates. The first step involves the electrophilic bromination of m-trifluoromethyl fluorobenzene using dibromohydantoin in a glacial acetic acid medium, where the strong electron-withdrawing nature of the trifluoromethyl group directs the incoming bromine atom to the desired position ortho to the fluorine. This specific orientation is critical because it sets the stage for the subsequent nucleophilic aromatic substitution, ensuring that the cyano group is installed exactly where needed for the final biological activity of the Bicalutamide molecule. The use of concentrated sulfuric acid as a catalyst in this step further enhances the electrophilicity of the brominating agent, driving the reaction to completion within a reasonable timeframe of five to seven hours under reflux conditions. Such controlled reactivity minimizes the formation of poly-brominated impurities, thereby reducing the burden on purification systems in later stages of the manufacturing process.
Following bromination, the installation of the nitrile functionality is achieved through a modified Rosenmund-von Braun reaction using cuprous cyanide in quinoline, a solvent that facilitates the solubility of the copper salt and stabilizes the transition state. This cyanation step proceeds via a nucleophilic aromatic substitution mechanism where the carbon-nitrogen triple bond replaces the carbon-bromine bond, a transformation that is highly sensitive to temperature and stoichiometry. The patent specifies a molar ratio of cuprous cyanide to the bromo-intermediate of approximately 1:1 to 1.1:1, which is optimized to prevent the formation of homocoupling by-products while ensuring complete consumption of the valuable halide precursor. The resulting 4-fluoro-2-trifluoromethyl benzonitrile is then subjected to aminolysis with liquid ammonia in ethanol at elevated temperatures, where the fluorine atom acts as an excellent leaving group due to the activation by the adjacent electron-withdrawing groups. This final substitution yields the target amine with a purity exceeding 99%, demonstrating the robustness of the mechanistic design in controlling the impurity profile throughout the entire synthetic sequence.
How to Synthesize 4-Amino-2-Trifluoromethyl Benzonitrile Efficiently
Implementing this patented synthesis requires strict adherence to the specified reaction conditions and reagent ratios to maximize yield and safety, particularly given the use of pressurized liquid ammonia and cyanide salts. The process is designed to be scalable, moving seamlessly from laboratory verification to multi-ton production without the need for specialized high-pressure equipment beyond standard industrial autoclaves. Operators must ensure precise temperature control during the exothermic bromination phase and maintain an inert atmosphere during the cyanation step to prevent oxidation of the copper catalyst.
- Perform regioselective bromination of m-trifluoromethyl fluorobenzene using dibromohydantoin in glacial acetic acid.
- Execute copper-catalyzed cyanation using cuprous cyanide in quinoline to replace the bromine atom.
- Conclude with high-pressure aminolysis using liquid ammonia in ethanol to yield the final amino-nitrile product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers compelling economic and logistical benefits that directly impact the bottom line and operational resilience. By eliminating the need for iron powder reduction and the associated solvent-intensive extraction processes, manufacturers can achieve significant cost reduction in API manufacturing through decreased raw material consumption and lower waste disposal fees. The simplified workflow reduces the number of unit operations, which in turn shortens the overall production cycle time and allows for faster turnover of batch reactors, enhancing the overall capacity utilization of the facility. Furthermore, the reliance on commodity chemicals such as glacial acetic acid, ethanol, and liquid ammonia ensures that the supply chain remains robust and less susceptible to the volatility often seen with specialized reagents. This stability is crucial for maintaining continuous supply to downstream pharmaceutical partners who require just-in-time delivery of critical intermediates for their own production schedules.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents, combined with the drastic reduction in solvent usage for extraction, leads to substantial cost savings per kilogram of finished product. The higher overall yield means that less starting material is required to produce the same amount of final API intermediate, effectively lowering the cost of goods sold. Additionally, the removal of the iron sludge waste stream significantly reduces environmental compliance costs and the logistical burden of hazardous waste transport and treatment. These factors collectively contribute to a more economically viable production model that can withstand market fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: Sourcing reliability is markedly improved as the process utilizes widely available industrial chemicals rather than niche reagents that may face supply constraints. The shortened synthetic route reduces the number of potential failure points in the manufacturing process, thereby increasing the predictability of delivery timelines. With fewer steps and simpler workup procedures, the risk of batch failures due to operational errors is minimized, ensuring a consistent flow of material to customers. This reliability is essential for long-term contracts with multinational pharmaceutical companies that prioritize supply security above all else.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with reaction conditions that are easily manageable in large-scale stainless steel reactors without requiring exotic materials of construction. The reduction in hazardous waste generation aligns with increasingly strict global environmental regulations, future-proofing the manufacturing site against tighter emission standards. The ability to treat emissions easily and the consistent product quality reduce the need for re-processing or scrapping batches, further enhancing the sustainability profile of the operation. This alignment with green chemistry principles makes the supplier a more attractive partner for environmentally conscious corporate buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis route for 4-amino-2-trifluoromethyl benzonitrile. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational feasibility. Understanding these details is vital for technical teams evaluating the integration of this intermediate into their broader supply chain strategies.
Q: What is the primary advantage of the new synthesis route over traditional methods?
A: The new route eliminates the use of hazardous iron powder reduction and complex diazotization steps, significantly reducing heavy metal waste and simplifying purification while increasing total yield from approximately 30% to over 73%.
Q: How does this process impact the purity profile of the final intermediate?
A: By avoiding iron sludge contamination and utilizing precise nucleophilic substitutions, the process consistently achieves HPLC purity levels exceeding 99%, which is critical for downstream API synthesis.
Q: Are the raw materials for this patented method readily available for scale-up?
A: Yes, the process relies on commercially accessible reagents such as glacial acetic acid, quinoline, and liquid ammonia, ensuring robust supply chain continuity without reliance on specialized or restricted precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Amino-2-Trifluoromethyl Benzonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of oncology drugs like Bicalutamide depends heavily on the consistent availability of high-quality intermediates produced via robust and scalable pathways. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the fluctuating demands of the global pharmaceutical market with agility and precision. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 4-amino-2-trifluoromethyl benzonitrile meets the exacting standards required for API synthesis. Our commitment to process excellence means that we can deliver the benefits of this patented route to our partners, providing a secure and efficient source of this critical building block.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with our technical procurement team, you can access specific COA data and route feasibility assessments that demonstrate the tangible value of switching to our optimized supply chain. Let us help you streamline your sourcing strategy and secure a competitive advantage in the fast-paced pharmaceutical industry through our dedicated service and technical expertise.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
