Optimizing Clopidogrel Intermediate Production: A Deep Dive into High-Efficiency Esterification
The pharmaceutical industry continuously demands more efficient pathways for synthesizing critical active pharmaceutical ingredient (API) intermediates, particularly for high-volume cardiovascular medications like Clopidogrel. Patent CN101560202B introduces a transformative preparation method for 2-(2-thienyl) ethanol p-toluenesulfonate, a pivotal precursor in the Clopidogrel synthesis value chain. This technology leverages a specialized pyridine-catalyzed esterification strategy that fundamentally alters the kinetic profile of the reaction compared to traditional methodologies. By integrating specific nucleophilic catalysts such as N,N-dimethylaminopyridine (DMAP) into a toluene-based system, the process achieves a dramatic reduction in reaction duration, compressing what was previously a multi-day operation into a swift 1.5 to 3-hour window. This acceleration is not merely a matter of speed but represents a significant leap in process chemistry optimization, directly addressing the bottlenecks of throughput and thermal stability that have long plagued the manufacturing of sulfonate esters. For R&D directors and process chemists, this patent offers a validated blueprint for enhancing purity profiles while mitigating the formation of hydrolytic impurities that typically degrade yield in conventional acid chloride reactions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art techniques, such as those disclosed in U.S. Pat 2007/0225320, relied heavily on the use of triethylamine solely as an acid binding agent without the benefit of nucleophilic catalysis. In these legacy processes, the esterification between Thiophene-2-Ethanol and p-toluenesulfonyl chloride was notoriously sluggish, often requiring extended reaction times of up to 30 hours even at elevated temperatures around 30°C. Such prolonged exposure to reactive conditions inevitably led to significant side reactions, most notably the facile hydrolysis of the sensitive sulfonate ester product. This hydrolytic degradation not only suppressed the overall yield but also complicated the downstream purification processes, necessitating rigorous and costly separation steps to remove acidic byproducts and unreacted starting materials. Furthermore, the thermal instability of the product under long-duration heating posed a safety risk and limited the scalability of the process, making it economically unviable for high-tonnage commercial production where reactor occupancy time is a critical cost driver.
The Novel Approach
The innovative methodology described in CN101560202B overcomes these historical deficiencies by introducing a pyridine-based catalytic system that activates the sulfonyl chloride species more effectively. By conducting the esterification in the presence of catalysts like DMAP, pyridine, or hexahydropyridine, the reaction kinetics are vastly improved, allowing the transformation to reach completion within a mere 1.5 to 3 hours. This rapid conversion minimizes the residence time of the product in the reaction mixture, thereby drastically reducing the opportunity for hydrolytic decomposition. The process operates under mild thermal conditions, typically maintaining temperatures between -5°C and 25°C, which further preserves the structural integrity of the thermally sensitive thiophene moiety. As demonstrated in the patent embodiments, this approach consistently delivers yields exceeding 85%, with optimized conditions reaching an impressive 98% purity, establishing a new benchmark for efficiency in the synthesis of this critical pharmaceutical intermediate.
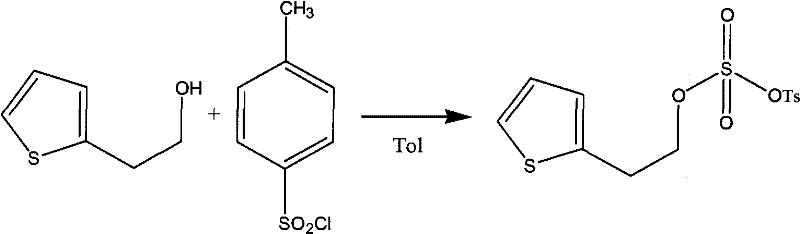
Mechanistic Insights into Pyridine-Catalyzed Esterification
The core of this technological advancement lies in the nucleophilic catalysis mechanism facilitated by the pyridine derivatives. In the absence of a catalyst, the direct attack of the alcohol oxygen on the sulfur atom of the sulfonyl chloride is kinetically hindered by the steric bulk and the electronic nature of the leaving group. However, when a nucleophilic catalyst like DMAP is introduced, it initially attacks the sulfur center of the p-toluenesulfonyl chloride to form a highly reactive N-sulfonylpyridinium intermediate. This cationic species is significantly more electrophilic than the original sulfonyl chloride, making it far more susceptible to nucleophilic attack by the Thiophene-2-Ethanol. This two-step pathway lowers the activation energy of the rate-determining step, explaining the observed acceleration in reaction rate. The subsequent displacement of the pyridine catalyst by the alcohol regenerates the catalyst for another cycle, ensuring that only catalytic amounts (1% to 10% by weight) are required to drive the reaction to completion efficiently.
From an impurity control perspective, this mechanism offers distinct advantages regarding the suppression of side products. The rapid formation of the target sulfonate ester means that the concentration of free p-toluenesulfonyl chloride in the reaction medium drops quickly, reducing the likelihood of competing hydrolysis reactions with trace moisture. Additionally, the use of triethylamine as a stoichiometric acid scavenger ensures that the hydrogen chloride generated during the reaction is immediately neutralized, preventing acid-catalyzed degradation of the thiophene ring or the ester linkage. The combination of low-temperature operation and rapid kinetics creates a 'kinetic trap' where the desired product is formed faster than it can decompose. This precise control over the reaction environment results in a cleaner crude product profile, simplifying the workup procedure and reducing the burden on final purification steps, which is crucial for meeting the stringent quality standards required for API intermediates.
How to Synthesize 2-(2-thienyl) ethanol p-toluenesulfonate Efficiently
The practical implementation of this synthesis route involves a straightforward sequence of mixing, cooling, and reaction steps that are easily adaptable to standard glass-lined or stainless steel reactors found in modern chemical facilities. The process begins with the dissolution of the sulfonyl chloride in toluene, followed by a controlled cooling phase to ensure the exothermic nature of the subsequent addition does not spike the temperature beyond the optimal range. The sequential addition of the alcohol, catalyst, and base allows for precise management of the reaction exotherm and gas evolution. Detailed standardized operating procedures regarding specific molar ratios, agitation speeds, and quenching protocols are essential for reproducing the high yields reported in the patent literature. Operators must adhere strictly to the temperature constraints to maximize the benefit of the catalytic system.
- Dissolve p-toluenesulfonyl chloride in toluene solvent within a reaction vessel under stirring.
- Cool the solution to below 5°C, then add Thiophene-2-Ethanol, a pyridine catalyst (preferably DMAP), and triethylamine.
- Maintain the reaction temperature between -5°C and 25°C for 1.5 to 3 hours, followed by filtration, washing, and distillation to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this catalytic esterification technology translates into tangible operational efficiencies and cost optimizations throughout the manufacturing value chain. The drastic reduction in reaction time from 30 hours to under 3 hours implies a massive increase in asset utilization, allowing manufacturers to produce significantly more batches per month using the same reactor infrastructure. This increase in throughput capacity directly addresses supply continuity concerns, ensuring that downstream API producers have a reliable flow of intermediates without the risk of production bottlenecks. Furthermore, the high yield profile means that less raw material is wasted per kilogram of finished product, leading to a more sustainable and cost-effective consumption of starting materials like Thiophene-2-Ethanol and Tosyl chloride. The elimination of prolonged heating cycles also results in substantial energy savings, contributing to a lower carbon footprint and reduced utility costs for the manufacturing site.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the intensification of the reaction timeline and the minimization of waste. By eliminating the need for extended heating and reducing the formation of hydrolytic byproducts, the process lowers the overall cost of goods sold (COGS). The high selectivity reduces the load on purification systems, meaning less solvent and energy are required for recrystallization or distillation steps. Additionally, the use of catalytic amounts of relatively inexpensive pyridine derivatives instead of stoichiometric quantities of more costly reagents further optimizes the raw material bill. These factors combine to create a leaner manufacturing process that is highly competitive in the global market for generic pharmaceutical ingredients.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the robustness and speed of this synthetic route. The short reaction window reduces the risk of equipment failure or process deviation affecting a batch, as the exposure time to potential variables is minimized. The use of common, commercially available solvents like toluene and standard reagents ensures that the supply chain is not dependent on exotic or single-source materials that could face availability issues. This reliability allows for more accurate demand forecasting and inventory management, enabling suppliers to respond more agilely to fluctuations in the market demand for Clopidogrel. The consistency of the yield also ensures predictable output volumes, which is critical for maintaining long-term contracts with major pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by its simple operational parameters and lack of extreme conditions. The reaction does not require high pressure or cryogenic temperatures, making it compatible with standard industrial reactor setups. From an environmental standpoint, the higher atom economy and reduced solvent usage for purification align with green chemistry principles. The minimization of waste streams, particularly acidic aqueous layers containing hydrolyzed byproducts, simplifies wastewater treatment requirements. This ease of compliance with environmental regulations reduces the administrative and operational burden on the manufacturing facility, ensuring uninterrupted production schedules and safeguarding the company's social license to operate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2-(2-thienyl) ethanol p-toluenesulfonate, derived directly from the specific embodiments and comparative data found in the patent documentation. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer or process validation. The answers provided reflect the specific advantages of the pyridine-catalyzed route over legacy methods, focusing on yield, purity, and operational safety.
Q: What is the primary advantage of using DMAP in this esterification process?
A: The use of N,N-dimethylaminopyridine (DMAP) as a catalyst significantly accelerates the reaction rate, reducing the completion time from 30 hours in prior art methods to just 1.5 to 3 hours, while simultaneously boosting yields to over 98%.
Q: How does this method address the issue of product hydrolysis?
A: By optimizing the reaction temperature to a controlled range of -5°C to 25°C and utilizing a highly efficient pyridine catalyst, the process minimizes the exposure time and conditions that lead to the facile hydrolysis of the sulfonate ester, thereby preserving yield.
Q: Is this synthesis route suitable for large-scale industrial manufacturing?
A: Yes, the method is explicitly designed for industrial suitability, utilizing common solvents like toluene and standard acid scavengers like triethylamine, resulting in a low-cost, scalable process ideal for commercial API intermediate production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2-thienyl) ethanol p-toluenesulfonate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the efficiency of your final API synthesis depends heavily on the quality and consistency of your starting intermediates. Our technical team has extensively analyzed advanced preparation methods like CN101560202B to ensure our manufacturing processes embody the highest standards of chemical engineering. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you require pilot-scale quantities or full commercial tonnage, our supply remains uninterrupted. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2-(2-thienyl) ethanol p-toluenesulfonate meets the exacting requirements necessary for the synthesis of life-saving cardiovascular medications.
We invite potential partners to engage with our technical procurement team to discuss how our optimized manufacturing capabilities can support your supply chain goals. By leveraging our expertise in process intensification and quality control, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us directly to request specific COA data and route feasibility assessments, allowing you to validate our technical claims and establish a partnership built on transparency, quality, and mutual growth in the competitive pharmaceutical marketplace.
