Advanced Synthesis of 1-Isopropylpyrazole-5-Boronic Acid Pinacol Ester for Commercial Scale-Up
Advanced Synthesis of 1-Isopropylpyrazole-5-Boronic Acid Pinacol Ester for Commercial Scale-Up
The pharmaceutical industry is constantly seeking robust and scalable synthetic routes for critical intermediates, particularly those serving novel therapeutic areas such as Sickle Cell Disease (SCD). Patent CN114181237A introduces a significant advancement in the synthesis of 1-isopropylpyrazole-5-boronic acid pinacol ester, a key building block for the investigational drug Woseltantor. This compound functions as a hemoglobin polymerization inhibitor, offering hope for patients suffering from this debilitating genetic disorder. The disclosed method addresses long-standing challenges in organic synthesis regarding yield optimization and impurity control. By re-engineering the solvent system and reaction conditions, this technology provides a pathway to high-purity materials essential for downstream drug substance manufacturing. For R&D directors and procurement specialists, understanding the nuances of this patented process is vital for securing a reliable supply chain. The innovation lies not just in the chemical transformation but in the strategic selection of reaction media that facilitates easier purification and enhances overall process efficiency. This report delves into the technical specifics and commercial implications of adopting this novel methodology for large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for similar pyrazole derivatives often rely on high-boiling polar aprotic solvents such as dimethylformamide (DMF) or dimethyl sulfoxide (DMSO). While these solvents are effective for dissolving reactants and facilitating nucleophilic substitution, they present significant downstream processing challenges. The primary issue arises during the work-up phase, where the high boiling point of the solvent makes it difficult to separate completely from the reaction product. Residual solvent molecules can persist even after standard extraction procedures. In multi-step syntheses, these residues become critical liabilities. Specifically, when the intermediate is subjected to sensitive organometallic reactions in subsequent steps, trace amounts of amides or sulfoxides can act as quenching agents. This leads to a drastic reduction in yield and the formation of complex impurity profiles that are difficult to remove. Furthermore, the energy consumption required to distill or remove these high-boiling solvents is substantial, impacting both the economic and environmental footprint of the manufacturing process. The accumulation of impurities from solvent decomposition, such as dimethylamine from DMF, further complicates the purification landscape, often necessitating additional chromatographic steps that are not feasible on a commercial scale.
The Novel Approach
The patented methodology offers a transformative solution by utilizing isopropanol as the primary reaction solvent for the initial alkylation step. Isopropanol possesses a boiling point significantly lower than that of the target intermediate, 1-isopropylpyrazole. This physical property difference is exploited to achieve a highly efficient separation strategy. Upon completion of the alkylation reaction, the solvent can be easily removed via distillation under reduced pressure, leaving behind the product with minimal contamination. This straightforward work-up procedure ensures that the intermediate entering the second step is of exceptionally high purity. The elimination of high-boiling solvent residues prevents the quenching of sensitive reagents in subsequent transformations. Consequently, the overall yield of the final boronic ester is markedly improved compared to conventional methods. This approach not only simplifies the operational workflow but also reduces the need for extensive purification protocols. The strategic choice of solvent demonstrates a deep understanding of process chemistry, where physical properties are leveraged to enhance chemical outcomes. This innovation sets a new standard for the synthesis of heterocyclic intermediates used in complex pharmaceutical applications.
Mechanistic Insights into N-Alkylation and Lithiation-Boronation
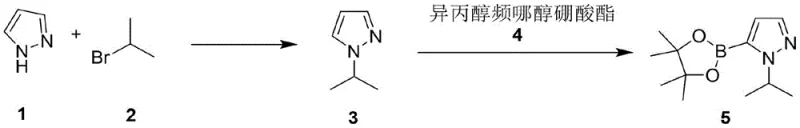
The core of this synthesis involves a two-step sequence that requires precise control over reaction conditions to maximize efficiency. The first step is an N-alkylation reaction where pyrazole reacts with 2-bromopropane in the presence of a mixed base system. The use of potassium hydroxide and potassium carbonate in isopropanol creates an optimal environment for deprotonation and nucleophilic attack. A phase transfer catalyst, such as tetrabutylammonium bromide, is employed to enhance the solubility of the inorganic base in the organic phase, thereby accelerating the reaction rate. The careful selection of the base ratio is critical; an excess of base can lead to side reactions, while insufficient base results in incomplete conversion. The reaction is conducted at elevated temperatures to ensure complete consumption of the starting material. Following the reaction, the mixture is cooled and filtered to remove inorganic salts. The filtrate is then subjected to distillation, where the low-boiling isopropanol is removed, and the product is isolated. This step is crucial as it sets the stage for the subsequent organometallic transformation by providing a clean substrate free from interfering impurities.
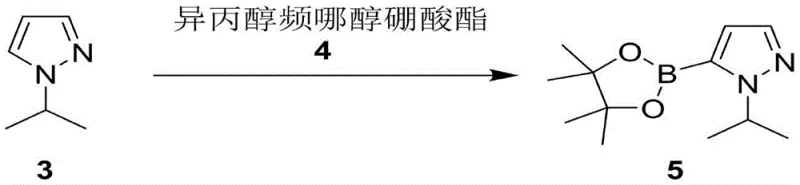
The second step involves a lithiation-boronation sequence, which is highly sensitive to the quality of the starting material. The purified 1-isopropylpyrazole is dissolved in tetrahydrofuran and cooled to cryogenic temperatures. n-Butyllithium is added dropwise to generate the lithiated intermediate at the 5-position of the pyrazole ring. This organolithium species is highly reactive and susceptible to quenching by any protic impurities or electrophilic contaminants carried over from the first step. If high-boiling solvents like DMF were used previously, their decomposition products could react with the n-butyllithium, consuming the reagent and lowering the yield. By ensuring the intermediate is free from such contaminants, the lithiation proceeds efficiently. The resulting anion is then trapped with isopropyl pinacol borate to form the final boronic ester. The reaction is quenched with aqueous ammonium chloride, and the product is isolated through extraction and crystallization. This mechanistic understanding highlights the importance of the solvent switch in the first step for the success of the second step.
How to Synthesize 1-Isopropylpyrazole-5-Boronic Acid Pinacol Ester Efficiently
Implementing this synthesis route requires adherence to specific operational parameters to ensure reproducibility and safety. The process begins with the preparation of the alkylation mixture, where the order of addition and temperature control are paramount. The use of a mixed base system requires careful monitoring to prevent exotherms during the addition of the alkyl halide. Following the reaction, the distillation process must be controlled to avoid thermal degradation of the product. The second step demands strict inert atmosphere conditions and low-temperature management to handle the pyrophoric n-butyllithium safely. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating this high-yielding process.
- Perform N-alkylation of pyrazole with 2-bromopropane using potassium hydroxide and carbonate in isopropanol with a phase transfer catalyst.
- Distill the reaction mixture to remove the low-boiling solvent and isolate 1-isopropylpyrazole with high purity.
- Conduct lithiation with n-butyllithium at low temperature followed by reaction with pinacol borate to yield the final ester.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthesis route offers substantial benefits for procurement managers and supply chain leaders looking to optimize their sourcing strategies for pharmaceutical intermediates. The shift from high-boiling polar solvents to isopropanol represents a significant reduction in processing complexity and cost. Isopropanol is a commodity chemical that is readily available globally, ensuring supply chain stability and reducing the risk of raw material shortages. Furthermore, the ease of solvent recovery through distillation allows for potential recycling, which aligns with sustainability goals and reduces waste disposal costs. The improved yield of the final product means that less starting material is required to produce the same amount of active intermediate, directly impacting the cost of goods sold. This efficiency gain is crucial for maintaining competitive pricing in the pharmaceutical market. Additionally, the robustness of the process reduces the likelihood of batch failures, ensuring consistent supply continuity for downstream drug manufacturers. The simplified purification process also shortens the production cycle time, allowing for faster turnaround on orders.
- Cost Reduction in Manufacturing: The elimination of expensive and difficult-to-remove high-boiling solvents significantly lowers the operational costs associated with solvent purchase and waste management. The ability to recover and reuse isopropanol further enhances the economic viability of the process. By avoiding the need for complex chromatographic purification to remove solvent residues, the manufacturing process becomes more streamlined and cost-effective. The higher overall yield reduces the consumption of raw materials per unit of product, contributing to substantial cost savings. These factors combined create a more efficient production model that can withstand market fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: The reliance on common, commercially available reagents such as isopropanol, potassium hydroxide, and tetrahydrofuran minimizes supply chain risks. Unlike specialized solvents that may have limited suppliers, these commodities are produced by multiple manufacturers worldwide, ensuring a stable supply. The robustness of the synthesis route reduces the dependency on highly specialized equipment or conditions, making it easier to transfer technology between manufacturing sites. This flexibility is essential for building a resilient supply chain that can adapt to changing demand or logistical challenges. Consistent product quality and reliable delivery schedules are critical for maintaining trust with pharmaceutical partners.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations such as distillation and crystallization that are easily adapted from laboratory to plant scale. The reduced use of hazardous high-boiling solvents lowers the environmental impact of the manufacturing process, facilitating compliance with increasingly stringent environmental regulations. The simplified work-up procedure generates less waste, contributing to a greener manufacturing footprint. This alignment with environmental, social, and governance (ESG) criteria is becoming a key differentiator for suppliers in the global market. The ability to scale up without compromising yield or purity ensures that commercial production targets can be met efficiently.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of this critical pharmaceutical intermediate. These answers are derived from the detailed technical data provided in the patent documentation and reflect the practical implications of the disclosed technology. Understanding these aspects is crucial for making informed decisions about process adoption and supplier selection. The information provided here aims to clarify the advantages of this novel route over traditional methods.
Q: Why is isopropanol preferred over DMF for the alkylation step?
A: Isopropanol has a lower boiling point than the product, allowing for easy removal via distillation. High-boiling solvents like DMF leave residues that interfere with subsequent lithiation steps.
Q: What is the impact of solvent purity on the lithiation yield?
A: Impurities from high-boiling solvents can quench n-butyllithium, drastically reducing the yield of the boronic ester. High purity intermediates are critical for success.
Q: Is this synthesis route scalable for commercial production?
A: Yes, the use of common solvents like isopropanol and THF, along with standard distillation and crystallization techniques, makes this route highly suitable for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Isopropylpyrazole-5-Boronic Acid Pinacol Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our technical team has extensively analyzed the synthesis route described in patent CN114181237A and is fully equipped to implement this advanced methodology for commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch meets the highest industry standards. We understand the complexities involved in organometallic chemistry and have the expertise to manage the sensitive lithiation steps safely and efficiently. Partnering with us means gaining access to a supply chain that is both robust and responsive to your specific requirements.
We invite you to collaborate with us to optimize your supply chain for this essential intermediate. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality expectations. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your projects. By leveraging our manufacturing capabilities and technical expertise, we can help you reduce lead times and secure a stable supply of high-purity materials. Let us support your drug development goals with our commitment to quality and innovation.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
