Advancing Chiral Nitrogen Heterocycle Production with Metal-Free One-Pot Cascade Technology
The pharmaceutical industry continuously seeks efficient pathways to construct nitrogen-containing heterocyclic scaffolds, which are foundational to modern drug discovery. According to patent CN110590644B, a novel synthetic methodology has been developed to address the longstanding challenges in producing chiral 1,2-dihydropyridine compounds. These structures are critical precursors for polysubstituted pyridines and chiral piperidines, motifs found in nearly 59% of FDA-approved small molecule drugs. The disclosed technology utilizes a sophisticated "one-pot cascade" strategy that integrates an asymmetric Mannich reaction, a Wittig olefination, and an intramolecular cyclization. This approach represents a significant leap forward in organic synthesis, offering a metal-free alternative that bypasses the limitations of traditional transition metal catalysis. For R&D directors and process chemists, this patent provides a robust framework for accessing high-value chiral building blocks with exceptional stereocontrol, potentially accelerating the development of next-generation therapeutic agents targeting various biological pathways.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral 1,2-dihydropyridines has been fraught with significant technical hurdles that hinder large-scale application. Traditional methods often rely on intramolecular cyclization reactions starting from natural amino acids, as demonstrated by early work from Kawabata et al. While these methods can achieve high enantiomeric excess, they suffer from poor universality due to the limited availability and structural diversity of chiral amino acid starting materials. Furthermore, alternative strategies involving nucleophilic addition to pyridinium salts, such as those reported by Shibasaki and Ma Dawei, typically require the use of expensive transition metal catalysts like copper or aluminum complexes. These metal-dependent processes introduce severe complications, including the necessity for rigorous heavy metal removal to meet pharmaceutical safety standards, poor atom economy due to the use of chiral auxiliaries, and restricted substrate scope that limits the diversity of substituents at the 2, 4, and 5 positions of the pyridine ring. Consequently, these conventional routes often result in higher production costs and complex purification workflows that are ill-suited for the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology outlined in patent CN110590644B introduces a transformative "three-step one-pot" synthesis that effectively circumvents these historical bottlenecks. By employing simple and readily available imine and aldehyde raw materials, the process constructs the chiral 1,2-dihydropyridine skeleton through a seamless sequence of asymmetric Mannich reaction, Wittig reaction, and Lewis acid-catalyzed cyclization. Crucially, this novel approach operates without the involvement of transition metals, utilizing organocatalysts like L-Proline for the initial stereoselective step and silicon tetrachloride for the final ring closure. This metal-free paradigm not only simplifies the reaction setup but also drastically reduces the environmental footprint and downstream purification burden. The reaction conditions are relatively mild, typically ranging from 0°C to 100°C, and the one-pot nature eliminates the need to isolate unstable intermediates, thereby improving overall yield and operational efficiency. This innovation provides a reliable pharmaceutical intermediate supplier with a versatile platform to generate diverse libraries of nitrogen heterocycles that were previously difficult or prohibitively expensive to access.
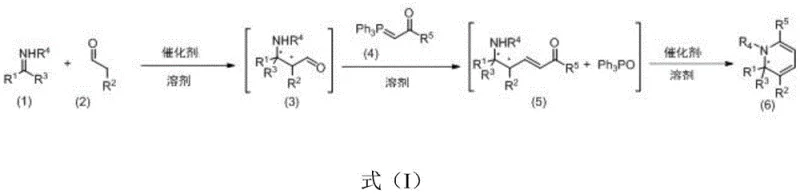
Mechanistic Insights into the Metal-Free Cascade Cyclization
The core of this technological breakthrough lies in the precise orchestration of three distinct chemical transformations within a single reaction vessel. The process initiates with an asymmetric Mannich reaction between an imine compound and an aldehyde, catalyzed by a chiral amine or amino acid derivative. This step is critical for establishing the initial stereocenter with high fidelity, setting the stage for the subsequent chirality transfer. Following the formation of the chiral aldehyde intermediate, the system undergoes a Wittig reaction with a phosphorus ylide. This olefination step extends the carbon chain and introduces the necessary unsaturation required for the final ring closure. Unlike traditional multi-step syntheses where intermediates are isolated and purified, this protocol allows the crude reaction mixture to proceed directly to the final stage. The culmination of the sequence is an intramolecular cyclization catalyzed by a Lewis acid, preferably silicon tetrachloride or trichlorosilane. This catalyst activates the carbonyl group, facilitating a nucleophilic attack by the nitrogen atom to close the six-membered ring. The synergy between these steps ensures that the stereochemical information generated in the first step is preserved throughout the cascade, resulting in final products with enantiomeric excess values consistently exceeding 90%.
From an impurity control perspective, the one-pot design offers substantial advantages over stepwise approaches. Unstable intermediates, particularly the chiral aldehydes and dihydropyridine precursors, are prone to oxidation and isomerization if exposed to air or prolonged storage. By keeping these species in solution and immediately converting them to the more stable cyclic product, the process minimizes the formation of degradation byproducts. Furthermore, the byproduct of the Wittig reaction, triphenylphosphine oxide, is noted in the patent to have a promoting effect on the subsequent cyclization, turning a potential waste product into a beneficial component of the reaction matrix. This intricate balance of reactivity allows for the synthesis of high-purity chiral 1,2-dihydropyridines without the need for chromatographic purification between steps, significantly streamlining the manufacturing workflow and reducing solvent waste generation.
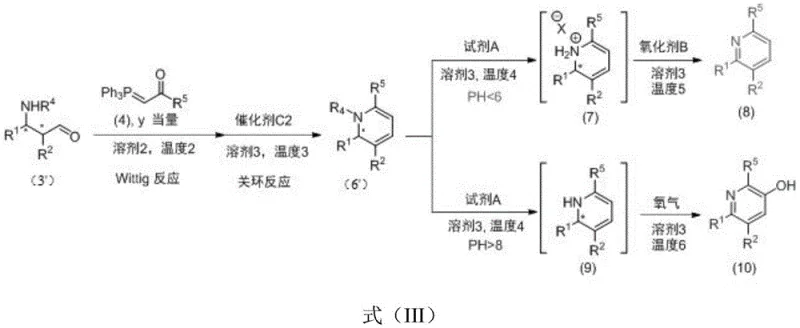
How to Synthesize Chiral 1,2-Dihydropyridine Efficiently
The practical implementation of this synthesis route is designed for operational simplicity and robustness, making it highly attractive for process development teams aiming to establish scalable manufacturing protocols. The procedure begins by combining the imine substrate and aldehyde in a suitable solvent such as acetonitrile or dichloromethane, followed by the addition of the chiral catalyst at controlled temperatures typically around 0°C. Once the Mannich adduct is formed, the ylide reagent is introduced directly to the same vessel, and the temperature is raised to facilitate the Wittig olefination. Finally, the Lewis acid catalyst is added to trigger the cyclization, completing the construction of the heterocyclic core. This streamlined workflow eliminates multiple workup and isolation stages, reducing the total processing time and labor costs associated with traditional multi-step syntheses. For detailed operational parameters and specific stoichiometric ratios tailored to your specific substrate requirements, please refer to the standardized synthesis guide below.
- Perform an asymmetric Mannich reaction between an imine compound and an aldehyde compound using a chiral catalyst to generate a chiral aldehyde intermediate.
- Conduct a Wittig reaction on the intermediate using a ylide compound to extend the carbon chain and form an unsaturated ketone precursor.
- Execute an intramolecular ring-closing reaction catalyzed by a Lewis acid such as silicon tetrachloride to finalize the chiral 1,2-dihydropyridine structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this metal-free cascade technology translates into tangible strategic benefits that extend beyond mere chemical efficiency. The elimination of transition metal catalysts removes a major cost driver associated with both the purchase of expensive noble metals and the specialized equipment required for their removal, such as scavenger resins or extensive filtration systems. This simplification of the downstream processing directly contributes to substantial cost savings in API manufacturing, allowing for more competitive pricing of the final active pharmaceutical ingredients. Additionally, the reliance on simple, commercially available starting materials like imines and aldehydes ensures a stable and resilient supply chain, mitigating the risks associated with sourcing complex or proprietary chiral pools that are often subject to market volatility and long lead times.
- Cost Reduction in Manufacturing: The absence of transition metals fundamentally alters the cost structure of the synthesis. By avoiding copper, aluminum, or palladium catalysts, the process eliminates the need for expensive ligand systems and the rigorous analytical testing required to certify low residual metal levels in the final product. This reduction in material and testing costs, combined with the high atom economy of the one-pot design, results in a significantly lower cost of goods sold. Furthermore, the ability to perform the reaction in common solvents like ethyl acetate or dichloromethane without requiring cryogenic conditions for all steps reduces energy consumption and utility costs, enhancing the overall economic viability of the production process.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route is bolstered by its use of commodity chemicals that are widely available from multiple global suppliers. Unlike methods dependent on scarce natural amino acids or specialized chiral auxiliaries, the imine and aldehyde precursors can be sourced reliably, ensuring continuity of supply even during market disruptions. The high yields and excellent stereoselectivity reported in the patent examples mean that less raw material is wasted to side reactions, maximizing the output from each batch. This efficiency allows manufacturers to maintain leaner inventory levels while still meeting production targets, thereby improving cash flow and reducing the capital tied up in working stock.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, the metal-free nature of this process aligns perfectly with the industry's shift towards greener chemistry. The avoidance of heavy metals simplifies waste treatment protocols, as the effluent does not require specialized handling for toxic metal disposal. This ease of waste management facilitates smoother regulatory approvals and reduces the environmental compliance burden on manufacturing sites. Moreover, the one-pot cascade design inherently reduces solvent usage and energy demand by consolidating three reaction steps into a single vessel operation. This intensification of the process makes it highly amenable to commercial scale-up, enabling the production of complex nitrogen heterocycles from 100 kgs to 100 MT annual volumes with consistent quality and minimal environmental impact.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method compares to existing industry standards. Understanding these nuances is essential for evaluating the feasibility of integrating this route into your current manufacturing portfolio.
Q: What are the primary advantages of this metal-free synthesis method compared to traditional transition metal catalysis?
A: The primary advantage is the elimination of expensive and toxic transition metal catalysts, which removes the need for costly heavy metal removal steps during purification. This significantly reduces production costs and simplifies regulatory compliance for pharmaceutical applications, while maintaining high enantioselectivity above 90% ee.
Q: Can this synthetic route be adapted to produce polysubstituted pyridines and piperidines?
A: Yes, the synthesized chiral 1,2-dihydropyridine intermediates serve as versatile precursors. They can be directly oxidized and aromatized in a one-pot sequence to form polysubstituted pyridines, or subjected to catalytic hydrogenation to yield chiral polysubstituted piperidines, providing a unified platform for diverse nitrogen heterocycle libraries.
Q: How does the one-pot cascade design impact process scalability and impurity control?
A: The one-pot design minimizes unit operations by avoiding the isolation of unstable intermediates, which reduces material loss and solvent consumption. This streamlined approach enhances process robustness and scalability, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates while ensuring consistent impurity profiles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 1,2-Dihydropyridine Supplier
The technological potential of the metal-free one-pot cascade synthesis described in patent CN110590644B offers a compelling opportunity for pharmaceutical companies seeking to optimize their supply chains for nitrogen heterocycle intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative academic and patent literature into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high stereoselectivity and purity specifications achieved in the lab are maintained at an industrial scale. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of chiral 1,2-dihydropyridine or derived pyridine meets the exacting standards required for global drug development programs.
We invite you to collaborate with us to leverage this advanced synthetic methodology for your specific project needs. Whether you require custom synthesis of novel analogs or large-scale production of established intermediates, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this metal-free technology can enhance the efficiency and profitability of your pharmaceutical manufacturing operations.
