Advanced Synthesis of Triazine Hydroxyethyl Ethylenediamine Polycarboxylic Acid for Industrial Rust Protection
Advanced Synthesis of Triazine Hydroxyethyl Ethylenediamine Polycarboxylic Acid for Industrial Rust Protection
The development of high-performance rust inhibitors is critical for the longevity and efficiency of mechanical systems, particularly in the realm of metalworking fluids and industrial coatings. Patent CN102250029A introduces a groundbreaking preparation method for triazine hydroxyethyl ethylenediamine polycarboxylic acid, a compound designed to offer superior corrosion resistance through a robust molecular architecture. This innovation leverages the inherent stability of the triazine ring derived from cyanuric chloride, combined with the chelating capabilities of hydroxyethyl ethylenediamine groups. By utilizing a step-wise nucleophilic substitution strategy, the process ensures precise control over the functionalization of the triazine core, resulting in a product with exceptional anti-rust and anti-hard water performance. For R&D directors and procurement specialists seeking reliable industrial rust inhibitor suppliers, this technology represents a significant leap forward in balancing performance with economic feasibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional rust inhibitors often rely on simple organic acids or phosphates which may lack the thermal stability required for modern high-speed machining operations. Many conventional syntheses involve multi-step processes with expensive precursors or require harsh reaction conditions that degrade the final product's purity. Furthermore, older generation additives frequently struggle in hard water environments, leading to precipitation and loss of efficacy in cooling systems. The reliance on unstable intermediates in legacy methods can also introduce batch-to-batch variability, complicating quality control for large-scale manufacturers. These limitations necessitate a shift towards more robust chemical scaffolds that can withstand rigorous industrial demands while maintaining cost-effectiveness.
The Novel Approach
The patented method addresses these challenges by employing cyanuric chloride as a foundational building block, capitalizing on its low cost and high reactivity. The process involves a controlled displacement of chlorine atoms on the triazine ring with hydroxyethyl ethylenediamine, creating a stable intermediate rich in nitrogen and oxygen donor atoms. This intermediate is subsequently carboxymethylated using chloroacetic acid, introducing multiple carboxyl groups that enhance water solubility and metal chelation. The result is a polycarboxylic acid derivative that forms a durable protective film on metal surfaces, effectively blocking corrosive agents. This approach not only simplifies the synthetic route but also utilizes commodity chemicals, drastically reducing the barrier to entry for cost reduction in industrial additive manufacturing.
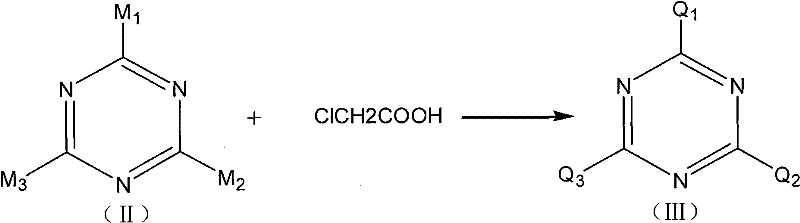
Mechanistic Insights into Nucleophilic Substitution and Carboxymethylation
The core of this synthesis lies in the sequential nucleophilic substitution reactions on the triazine ring, a mechanism that offers distinct advantages in terms of selectivity and yield. Initially, the chlorine atoms of cyanuric chloride are displaced by the amine groups of hydroxyethyl ethylenediamine under alkaline catalysis at temperatures between 60°C and 80°C. This step is critical as it establishes the organic framework of the inhibitor, embedding the hydroxyethyl and ethylene diamine moieties which are essential for surface adsorption. The reaction conditions are carefully optimized to prevent the hydrolysis of the remaining chlorine atoms, ensuring that the subsequent carboxymethylation step proceeds efficiently. The use of alkaline catalysts such as sodium hydroxide or sodium carbonate facilitates the deprotonation of the amine, enhancing its nucleophilicity and driving the reaction to completion within 3 to 5 hours.
Following the formation of the amino-substituted intermediate, the process moves to the carboxymethylation stage, where chloroacetic acid reacts with the secondary amines and hydroxyl groups. As illustrated in the structural analysis below, this step generates a complex mixture of polycarboxylic acid sodium salts, where the degree of substitution can vary. 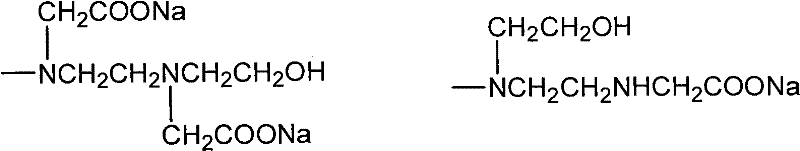 This structural diversity is actually beneficial, as it creates a broad spectrum of molecular weights and polarities that improve the additive's performance across different pH levels and water hardness conditions. The final acid precipitation step at 20°C to 50°C converts these soluble salts into the free acid form, which can be easily isolated by filtration. This mechanistic pathway ensures high-purity triazine derivatives with minimal byproduct formation, a key requirement for sensitive industrial applications.
This structural diversity is actually beneficial, as it creates a broad spectrum of molecular weights and polarities that improve the additive's performance across different pH levels and water hardness conditions. The final acid precipitation step at 20°C to 50°C converts these soluble salts into the free acid form, which can be easily isolated by filtration. This mechanistic pathway ensures high-purity triazine derivatives with minimal byproduct formation, a key requirement for sensitive industrial applications.
How to Synthesize Triazine Hydroxyethyl Ethylenediamine Polycarboxylic Acid Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and minimize impurities. The process begins with the preparation of the reaction vessel equipped with reflux and temperature control systems to handle the exothermic nature of the substitution reactions. Operators must maintain strict alkaline conditions throughout the initial stages to ensure complete conversion of the cyanuric chloride. Detailed standard operating procedures regarding molar ratios, specifically maintaining a cyanuric chloride to amine ratio of 1:3 to 1:3.5, are essential for reproducibility. For a comprehensive breakdown of the specific operational parameters and safety protocols, please refer to the standardized synthesis guide below.
- React cyanuric chloride with hydroxyethyl ethylenediamine under alkaline catalysis at 60-80°C for 3-5 hours to form the amino-substituted intermediate.
- Perform carboxymethylation by reacting the intermediate with chloroacetic acid under alkaline conditions at 50-70°C for 4-6 hours.
- Conduct acid precipitation at 20-50°C for 1-3 hours, followed by filtration and drying to isolate the final polycarboxylic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patent offers compelling advantages that directly address the pain points of supply chain volatility and rising raw material costs. By anchoring the synthesis on cyanuric chloride, a widely available and inexpensive commodity chemical, the process decouples production costs from the fluctuations associated with specialty fine chemicals. The elimination of transition metal catalysts, which are often required in other coupling reactions, removes the need for expensive downstream purification steps to remove heavy metal residues. This simplification of the workflow translates into shorter production cycles and reduced energy consumption, providing a clear pathway for commercial scale-up of complex polycarboxylic acids. Furthermore, the aqueous nature of the reaction medium aligns with modern environmental regulations, minimizing the generation of hazardous organic waste.
- Cost Reduction in Manufacturing: The utilization of cyanuric chloride and chloroacetic acid as primary feedstocks significantly lowers the bill of materials compared to traditional inhibitor syntheses. Since these reagents are produced on a massive global scale, their pricing is stable and predictable, allowing for accurate long-term budgeting. Additionally, the high atom economy of the substitution reactions means that a larger proportion of the input mass is converted into the final product, reducing waste disposal costs. The absence of exotic catalysts further reduces the operational expenditure, making this a highly attractive option for reducing lead time for high-purity rust inhibitors.
- Enhanced Supply Chain Reliability: The raw materials required for this process, including sodium hydroxide and sodium carbonate, are staple chemicals with robust global supply networks. This diversity in sourcing options mitigates the risk of supply disruptions that often plague manufacturers relying on single-source specialty intermediates. The synthesis itself is robust and tolerant to minor variations in feedstock quality, ensuring consistent output even when supply chains are stressed. For procurement managers, this reliability translates into secure inventory levels and the ability to meet sudden spikes in demand from the automotive or machinery sectors without delay.
- Scalability and Environmental Compliance: The reaction conditions, ranging from 50°C to 80°C, are mild and easily achievable in standard stainless steel reactors, facilitating seamless scale-up from pilot plants to multi-ton production facilities. The workup procedure, which involves simple filtration and acid precipitation, avoids the need for complex distillation or chromatography, thereby lowering capital expenditure on equipment. Moreover, the process generates primarily saline wastewater which is easier to treat than solvent-heavy waste streams, ensuring compliance with increasingly stringent environmental discharge standards. This combination of scalability and eco-friendliness positions the product as a sustainable choice for forward-thinking industrial partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this triazine-based rust inhibitor. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits for potential partners. Understanding these details is crucial for evaluating the fit of this technology within your existing manufacturing portfolio.
Q: What are the key advantages of using cyanuric chloride in this synthesis?
A: Cyanuric chloride offers a stable triazine backbone with highly reactive chlorine atoms, allowing for controlled step-wise substitution. This results in a robust molecular structure with excellent rust-inhibiting properties and hard water resistance.
Q: How does this method improve upon traditional rust inhibitor production?
A: This method utilizes inexpensive and readily available raw materials like cyanuric chloride and hydroxyethyl ethylenediamine. The process avoids complex purification steps often required in traditional methods, leading to significant cost reductions and simplified supply chains.
Q: Is the final product suitable for high-temperature industrial applications?
A: Yes, the triazine ring provides exceptional thermal stability. The resulting polycarboxylic acid maintains its structural integrity and corrosion protection capabilities even under the demanding conditions found in metalworking fluids and industrial cooling systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazine Hydroxyethyl Ethylenediamine Polycarboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this patented synthesis route in delivering next-generation corrosion protection solutions. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of triazine derivative performs consistently in your formulations. We are committed to leveraging our technical expertise to optimize this process further, driving down costs while maintaining the highest standards of quality and safety.
We invite you to collaborate with us to unlock the full commercial value of this innovative rust inhibitor. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and logistical requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how we can become your strategic partner in advancing industrial chemical solutions.
