Advanced One-Step Synthesis of 1-Aryl-2-Tetralones for High-Value Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries constantly seek more efficient pathways to access complex scaffolds like tetralones, which serve as critical precursors for bioactive agents and photochromic dyes. Patent CN102548651A introduces a transformative methodology for the selective synthesis of 1-aryl-2-tetralones, addressing long-standing challenges in regioselectivity and step economy. Unlike conventional multi-step sequences that suffer from poor atom economy and difficult purification, this innovation utilizes a sophisticated palladium-catalyzed cross-coupling strategy. By employing a specialized bis-phosphino-ferrocene palladium(II) catalyst system, the process enables the direct coupling of aryl halides to the 1-position of 2-tetralone derivatives. This breakthrough not only simplifies the synthetic route but also offers exceptional control over the substitution pattern, effectively eliminating the formation of unwanted 3-aryl isomers that typically plague traditional syntheses.
For R&D directors and process chemists, the implications of this technology are profound, offering a robust platform for generating diverse libraries of substituted tetralones with high purity. The ability to bypass intermediate isolation steps significantly reduces solvent consumption and waste generation, aligning with modern green chemistry principles. Furthermore, the mild reaction conditions and tolerance for various functional groups on both the aryl halide and the tetralone ring suggest broad applicability across different therapeutic areas. As we delve deeper into the technical specifics, it becomes clear that this method represents a significant leap forward in the manufacturing of high-value pharmaceutical intermediates, providing a reliable foundation for scaling up complex molecular architectures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-aryl-2-tetralones has been a cumbersome endeavor, typically relying on a circuitous route that begins with the reaction of 1-tetralone with aryl magnesium bromides, commonly known as the Grignard reaction. This traditional pathway, often referred to as the Green reaction, necessitates a subsequent dehydration step to form an alkene, followed by epoxidation and a final rearrangement to yield the desired ketone. Each of these sequential transformations introduces opportunities for yield loss, impurity formation, and significant operational complexity. The requirement for stoichiometric amounts of organometallic reagents and the need for rigorous anhydrous conditions throughout multiple stages drive up both material costs and safety risks. Moreover, the separation of the final product from byproducts and residual metals often requires extensive chromatographic purification, which is notoriously difficult to scale and economically inefficient for large-volume production.
The Novel Approach
In stark contrast, the methodology disclosed in the patent data streamlines this entire sequence into a single, elegant coupling event. By directly reacting an aryl halide with 2-tetralone in the presence of a palladium catalyst and a base, the process achieves the installation of the aryl group at the alpha-position of the ketone with remarkable efficiency. This one-pot transformation eliminates the need for protecting groups, intermediate isolations, and the harsh reagents associated with epoxidation and rearrangement chemistry. The result is a dramatic reduction in the overall process mass intensity (PMI) and a significant shortening of the production timeline. For procurement and supply chain teams, this translates to a more resilient manufacturing process that is less susceptible to bottlenecks caused by multi-step processing delays.
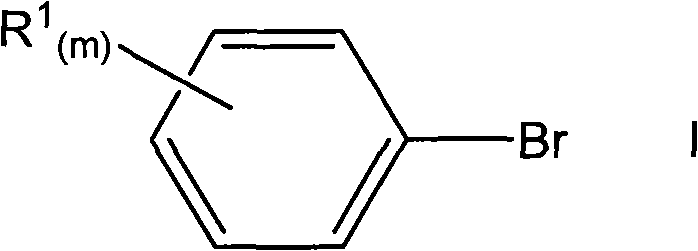
The visual representation of this reaction highlights the simplicity of the inputs versus the complexity of the output. Where previous methods required navigating through unstable intermediates like epoxides, this direct coupling leverages the inherent reactivity of the 2-tetralone enolate equivalent under palladium catalysis. This shift from a linear, multi-step dependency to a convergent, single-step operation is the hallmark of modern process intensification, offering immediate advantages in terms of throughput and operational expenditure.
Mechanistic Insights into Bis-Phosphino-Ferrocene Palladium Catalysis
The success of this selective synthesis hinges on the unique properties of the catalyst system, specifically the use of bis-phosphino-ferrocene ligands coordinated to palladium(II). The ferrocene backbone provides a rigid, sterically demanding environment that dictates the orientation of the substrate during the oxidative addition and reductive elimination steps of the catalytic cycle. Unlike simpler phosphine ligands, the bulky di-tert-butylphosphino groups often employed in this system create a specific pocket that favors interaction at the less hindered 1-position of the tetralone ring. This steric guidance is crucial for suppressing the thermodynamic tendency for substitution at the 3-position, which is a common side reaction in non-catalyzed or poorly catalyzed alkylation attempts.
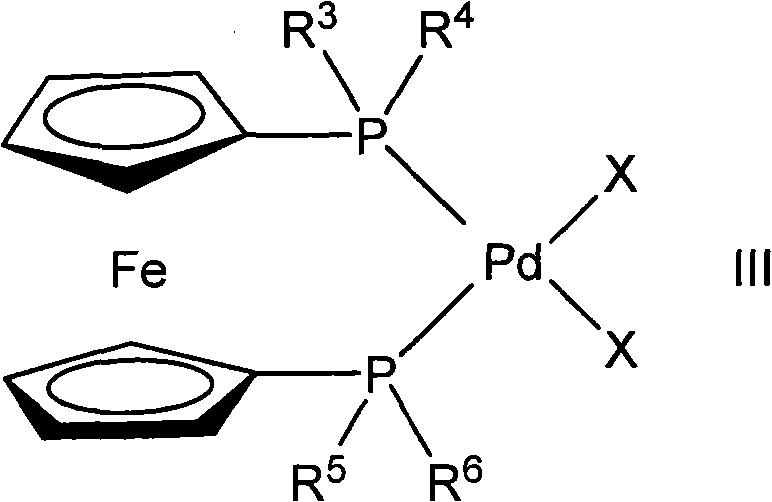
Furthermore, the electronic properties of the ferrocene moiety modulate the electron density at the palladium center, facilitating the activation of the aryl halide bond while maintaining stability under the basic reaction conditions required for enolization. The presence of an alkali metal alkoxide, such as potassium methoxide, serves a dual purpose: it generates the reactive enolate species from the 2-tetralone and acts as a base to neutralize the acid byproduct formed during the coupling. This synergistic relationship between the transition metal catalyst and the base ensures that the reaction proceeds smoothly to completion, often within a few hours, without the need for excessive heating or pressure.
Regioselectivity is perhaps the most critical parameter for R&D teams concerned with impurity profiles. The patent data indicates that this catalytic system can achieve selectivity ratios as high as 99:1 in favor of the 1-aryl isomer over the 3-aryl isomer. This level of control is unprecedented for this class of compounds and drastically simplifies downstream purification. By minimizing the formation of structural isomers, the process reduces the burden on analytical quality control and ensures that the final API intermediate meets stringent purity specifications without the need for recrystallization loops that sacrifice yield.
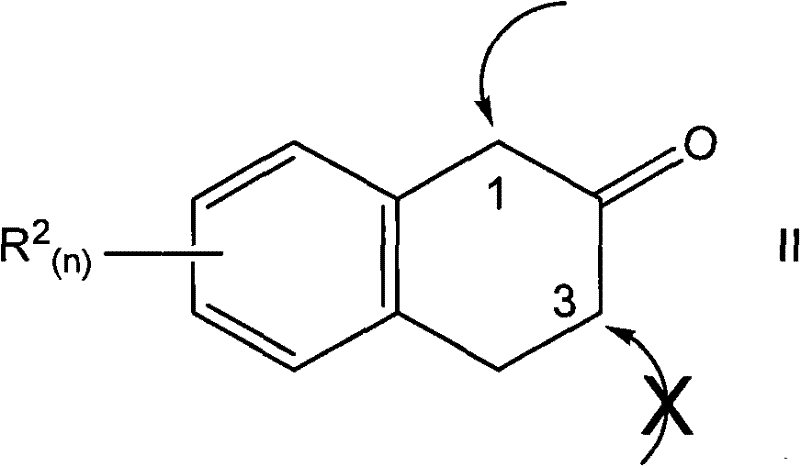
How to Synthesize 1-Aryl-2-Tetralone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the preparation of the catalytic mixture and the maintenance of an inert atmosphere to prevent catalyst deactivation. The general procedure involves dissolving the palladium catalyst and the alkoxide base in an anhydrous solvent such as THF or dioxane, followed by the sequential addition of the tetralone and aryl halide substrates. The reaction is typically heated to reflux or maintained at a controlled elevated temperature to drive the kinetics to completion. While the specific stoichiometry can be optimized based on the electronic nature of the substrates, the robustness of the catalyst allows for low loading levels, often around 2 mol%, which is economically favorable for large-scale operations.
- Prepare the reaction mixture by combining the bis-phosphino-ferrocene palladium(II) catalyst and alkali metal alkoxide (e.g., KOMe) in an anhydrous organic solvent under inert atmosphere.
- Add the 2-tetralone substrate and the aryl halide (e.g., aryl bromide) to the catalytic mixture, ensuring a molar ratio of approximately 1: 1.
- Heat the reaction mixture to reflux or maintain at elevated temperature (e.g., 100°C) until completion, followed by standard aqueous workup and chromatographic purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this direct coupling technology offers substantial benefits that extend beyond mere chemical elegance. For procurement managers, the simplification of the supply chain is a key value driver. By reducing the number of synthetic steps from four or five down to just one, the demand for intermediate raw materials, solvents, and reagents is drastically curtailed. This consolidation means fewer purchase orders, reduced inventory holding costs, and a lower risk of supply disruption for critical reagents like organomagnesium compounds or epoxidizing agents. The reliance on readily available aryl bromides and commodity 2-tetralone further stabilizes the supply base, ensuring consistent availability of starting materials even in volatile market conditions.
- Cost Reduction in Manufacturing: The elimination of multiple reaction vessels, workup procedures, and purification stages leads to a significant decrease in operational expenditures. Without the need for cryogenic conditions often required for Grignard reactions or the specialized equipment for epoxidation, the capital investment and utility costs per kilogram of product are substantially lowered. Additionally, the high selectivity of the reaction minimizes the loss of valuable starting materials to isomeric byproducts, thereby improving the overall effective yield and reducing the cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: The robustness of the palladium-catalyzed protocol allows for greater flexibility in manufacturing scheduling. Since the reaction tolerates a wide range of functional groups and proceeds under relatively mild conditions, it is less prone to batch failures caused by minor deviations in temperature or moisture content. This reliability translates to more predictable lead times for customers, enabling just-in-time delivery models that are essential for agile pharmaceutical development pipelines. The ability to source generic aryl halides from multiple global suppliers further mitigates the risk of single-source dependency.
- Scalability and Environmental Compliance: The patent explicitly mentions the compatibility of this chemistry with continuous flow reactors, such as microreactors, which represents a frontier in scalable manufacturing. Continuous processing allows for precise control over reaction parameters, enhancing safety by minimizing the hold-up volume of reactive intermediates. From an environmental standpoint, the reduction in solvent usage and waste generation aligns with increasingly strict regulatory frameworks regarding industrial emissions. The simplified waste stream, devoid of heavy metal sludge from stoichiometric reagents, facilitates easier disposal and treatment, contributing to a more sustainable corporate footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding the nuances of catalyst selection, substrate scope, and purification requirements is essential for successfully transferring this technology from the bench to commercial production.
Q: What is the primary advantage of this Pd-catalyzed method over traditional Grignard routes?
A: The primary advantage is the drastic reduction in synthetic steps. Traditional methods require a multi-step sequence involving Grignard addition, dehydration, epoxidation, and rearrangement. This novel method achieves the transformation in a single direct coupling step with high regioselectivity.
Q: How does the process control regioselectivity between the 1-position and 3-position?
A: The use of specific bis-phosphino-ferrocene ligands on the palladium center creates a steric and electronic environment that strongly favors coupling at the 1-position of the 2-tetralone ring, often achieving ratios of 99:1 over the 3-substituted isomer.
Q: Is this process suitable for large-scale continuous manufacturing?
A: Yes, the patent explicitly describes the compatibility of this chemistry with continuous flow technology, such as microreactors, allowing for precise temperature control and scalable production from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Aryl-2-Tetralone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the broader context of drug development and commercialization. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 1-aryl-2-tetralone delivered meets the exacting standards required for pharmaceutical applications. Our commitment to quality is matched only by our dedication to process optimization, leveraging advanced catalytic technologies to deliver superior products.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this streamlined method. We encourage you to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a trusted partner in your supply chain. Together, we can accelerate the development of next-generation therapeutics through superior chemical manufacturing.
