Advanced Oxidation Technology for High-Purity p-Methoxybenzaldehyde and Commercial Scalability
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes that balance high purity with environmental sustainability. Patent CN103145527A introduces a significant advancement in the production of key aromatic aldehydes, specifically p-methoxybenzaldehyde and p-tert-butylbenzaldehyde. This technology leverages a catalytic oxidation system utilizing cobalt acetate and sodium bromide in a glacial acetic acid medium, followed by a sophisticated separation protocol. Unlike traditional methods that rely heavily on solvent extraction and generate substantial inorganic waste, this novel approach employs high-boiling point solvents and stepwise vacuum distillation to achieve superior product isolation. For R&D Directors and Procurement Managers, this represents a critical opportunity to optimize the supply chain for these essential pharma intermediates. The process not only enhances the purity profile of the final product but also drastically simplifies the downstream processing requirements, offering a compelling value proposition for large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted benzaldehydes from their corresponding toluene derivatives has been plagued by complex separation challenges and significant environmental burdens. Conventional processes typically involve an oxidation step followed by a series of aqueous workups, including alkaline washing to separate acidic byproducts. A major drawback of these legacy methods is the reliance on organic solvents like toluene for extraction purposes to recover the desired benzoic acid byproducts. This extraction step not only increases the operational complexity but also introduces substantial solvent loss and volatility issues. Furthermore, the neutralization of alkaline washes with sulfuric acid generates large volumes of wastewater containing high concentrations of sodium sulfate. This saline wastewater is difficult and costly to treat, posing a significant compliance risk for manufacturers operating under strict environmental regulations. The cumulative effect of these inefficiencies results in higher production costs and a larger carbon footprint, making conventional routes less attractive for modern, sustainability-focused supply chains.
The Novel Approach
The technology disclosed in patent CN103145527A offers a transformative solution by re-engineering the separation phase of the synthesis. Instead of relying on liquid-liquid extraction with volatile solvents, this method introduces a high-boiling point organic solvent, such as methyl biphenyl or a biphenyl-biphenyl ether mixture, into the reaction mixture post-oxidation. This strategic addition allows for the implementation of a stepwise vacuum distillation protocol. By carefully controlling vacuum levels and temperature gradients, the process sequentially separates the unreacted starting material, the target aldehyde, and the carboxylic acid byproduct. This physical separation method eliminates the need for toluene extraction entirely, thereby removing the associated solvent consumption and emissions. Moreover, because the acid byproduct is isolated via distillation and subsequent crystallization from water rather than acidification of a salt solution, the generation of sodium sulfate wastewater is completely avoided. This streamlined approach not only reduces waste disposal costs but also enhances the overall mass balance of the process, delivering a cleaner and more economically viable manufacturing pathway.
Mechanistic Insights into Co/Br-Catalyzed Oxidation
The core of this synthesis lies in the efficient oxidation of the methyl group on the aromatic ring to an aldehyde functionality. The reaction utilizes a cobalt-bromide catalytic system, where cobalt acetate acts as the primary metal catalyst and sodium bromide serves as a promoter. In the presence of glacial acetic acid and molecular oxygen, this system generates radical species that abstract hydrogen atoms from the benzylic position of the p-methoxytoluene or p-tert-butyltoluene substrate. The reaction conditions are carefully controlled, typically maintaining temperatures around 45°C during the oxygen sparging phase to ensure selective oxidation while minimizing over-oxidation to the carboxylic acid, although some acid formation is inevitable and managed via the separation process. The use of oxygen as the terminal oxidant is particularly advantageous from a green chemistry perspective, as the only byproduct of the reduction of oxygen is water. This contrasts sharply with stoichiometric oxidants like chromates or permanganates, which generate heavy metal waste. The catalytic cycle ensures that the metal species are regenerated, allowing for high turnover numbers and efficient use of the catalyst loading.
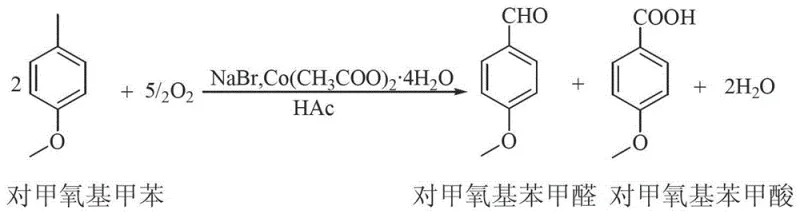
Impurity control is a critical aspect of this mechanism, particularly given the propensity for aldehydes to over-oxidize to carboxylic acids. The process manages this by leveraging the differences in boiling points and solubility properties during the workup. The initial water wash effectively removes the bulk of the cobalt and bromide catalysts along with the acetic acid solvent, preventing metal contamination in the final organic product. The subsequent addition of the high-boiling solvent is crucial for stabilizing the distillation process. It ensures that the target aldehyde, which has a moderate boiling point, can be distilled off under vacuum without thermal degradation, while the higher boiling acid byproduct remains in the pot until higher temperatures are applied. This physical separation mechanism complements the chemical selectivity of the oxidation, ensuring that the final p-methoxybenzaldehyde or p-tert-butylbenzaldehyde meets stringent purity specifications, often exceeding 99% content as demonstrated in the patent examples. The ability to co-produce the benzoic acid in high purity further adds value, turning a potential waste stream into a saleable co-product.
How to Synthesize p-Methoxybenzaldehyde Efficiently
Implementing this synthesis route requires precise control over reaction parameters and distillation conditions to maximize yield and purity. The process begins with the charging of the substituted toluene substrate into a reactor along with glacial acetic acid and the cobalt-bromide catalyst system. Oxygen is then introduced at a controlled rate while maintaining the reaction temperature to drive the oxidation to the desired conversion. Following the reaction, the workup involves a straightforward aqueous wash to remove inorganic species, followed by the addition of the high-boiling solvent. The mixture is then subjected to a multi-stage vacuum distillation sequence.
- Oxidize p-methoxytoluene with oxygen using cobalt acetate and sodium bromide catalysts in glacial acetic acid.
- Wash the reaction mixture with water to remove catalysts and acid, then add a high-boiling solvent.
- Perform stepwise vacuum distillation to separate unreacted toluene, aldehyde product, and benzoic acid byproduct.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process offers tangible benefits that extend beyond mere technical feasibility. The primary advantage lies in the significant reduction of operational costs associated with waste management and solvent consumption. By eliminating the toluene extraction step, the process removes a major source of solvent loss and the associated costs of solvent recovery or disposal. Furthermore, the avoidance of sodium sulfate wastewater generation simplifies the effluent treatment process, reducing the burden on wastewater treatment facilities and lowering compliance costs. These efficiencies translate into a more stable and predictable cost structure for the production of these key intermediates. The simplified process flow also enhances supply chain reliability, as there are fewer unit operations that can become bottlenecks or sources of variability. This robustness is essential for maintaining consistent supply to downstream pharmaceutical customers who require high-quality raw materials for their own synthesis campaigns.
- Cost Reduction in Manufacturing: The elimination of toluene extraction and the associated solvent recovery systems leads to substantial cost savings in utility and material consumption. Without the need to purchase, recover, or dispose of large volumes of toluene, the overall variable cost of production is significantly lowered. Additionally, the removal of the sulfuric acid neutralization step reduces the consumption of bulk chemicals and the generation of solid waste salts. These cumulative savings allow for a more competitive pricing structure for the final aldehyde product, providing a distinct advantage in cost reduction in fine chemical manufacturing. The ability to recover and recycle the high-boiling solvent further contributes to long-term economic efficiency, ensuring that the process remains viable even at fluctuating raw material prices.
- Enhanced Supply Chain Reliability: The streamlined nature of this process, with fewer separation steps and reduced dependency on complex extraction equipment, inherently improves operational reliability. Fewer process steps mean fewer potential points of failure, leading to higher uptime and more consistent production schedules. This reliability is crucial for reducing lead time for high-purity pharma intermediates, as it minimizes the risk of batch failures or delays caused by equipment maintenance or waste treatment issues. The use of standard industrial equipment such as oxidation reactors and distillation columns ensures that the process can be easily replicated across different manufacturing sites, providing flexibility in sourcing and reducing the risk of supply disruption. This robustness makes the supplier a more dependable partner for long-term contracts.
- Scalability and Environmental Compliance: The commercial scale-up of complex aromatic aldehydes is often hindered by environmental constraints, but this technology addresses those challenges directly. By avoiding the generation of saline wastewater and volatile organic compound emissions from toluene, the process aligns well with increasingly strict global environmental regulations. This compliance reduces the risk of regulatory shutdowns or fines, ensuring continuous operation. The distillation-based separation is inherently scalable, allowing for seamless transition from pilot plant to multi-ton production without significant re-engineering. This scalability ensures that the supply can grow in tandem with market demand, supporting the commercial scale-up of complex polymer additives or pharmaceutical intermediates without compromising on environmental standards or product quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology. The answers are derived directly from the patent data and practical manufacturing considerations. Understanding these details helps stakeholders evaluate the feasibility of integrating this material into their supply chains.
Q: How does this process reduce environmental impact compared to traditional methods?
A: This process eliminates the need for toluene extraction and avoids the generation of sodium sulfate wastewater, significantly reducing hazardous waste discharge.
Q: What is the purity level achievable with this distillation method?
A: Through precise vacuum distillation and recrystallization, the process achieves purity levels exceeding 99% for both the aldehyde and acid products.
Q: Can this synthesis route be scaled for industrial production?
A: Yes, the use of standard unit operations like oxidation reactors and vacuum distillation columns makes this route highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Methoxybenzaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes for high-value intermediates like p-methoxybenzaldehyde. Our technical team has thoroughly analyzed the capabilities of the oxidation and distillation technology described in CN103145527A and is well-positioned to implement this process at an industrial scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is seamless and efficient. Our facilities are equipped with state-of-the-art oxidation reactors and high-vacuum distillation columns, allowing us to meet stringent purity specifications consistently. With our rigorous QC labs, we guarantee that every batch of p-methoxybenzaldehyde or p-tert-butylbenzaldehyde meets the exacting standards required by the global pharmaceutical industry, providing you with a reliable pharma intermediate supplier you can trust.
We invite you to collaborate with us to optimize your supply chain for these essential building blocks. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. By leveraging our expertise in catalytic oxidation and separation technologies, we can help you secure a stable, cost-effective, and environmentally responsible supply of these critical chemicals, ensuring your production schedules remain uninterrupted and competitive in the global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
