Scalable Synthesis of Novel Beta-Elemene Based Histone Deacetylase Inhibitors for Oncology Applications
Scalable Synthesis of Novel Beta-Elemene Based Histone Deacetylase Inhibitors for Oncology Applications
The pharmaceutical industry is constantly seeking novel scaffolds to improve the efficacy and safety profile of epigenetic therapies, particularly histone deacetylase (HDAC) inhibitors. Patent CN112707833A introduces a groundbreaking approach by integrating beta-elemene, a natural sesquiterpene with known antitumor properties, into the structure of HDAC inhibitors. This innovation replaces the conventional aromatic hydrophobic CAP groups with the unique steric and electronic environment of the beta-elemene skeleton. For R&D directors and procurement specialists, this represents a significant opportunity to access a new class of high-purity pharmaceutical intermediates that leverage natural product chemistry for enhanced biological activity. The patent details comprehensive synthetic routes for four distinct series of compounds, utilizing robust chemical transformations that are well-suited for industrial application.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional HDAC inhibitors typically rely on simple aromatic rings, such as benzene or heterocycles, to serve as the surface recognition CAP group. While effective, these structures often suffer from limitations in selectivity, leading to off-target effects and toxicity issues in clinical settings. Furthermore, the synthesis of complex aromatic caps often requires harsh conditions, expensive transition metal catalysts, and multi-step purification processes that drive up the cost of goods. The reliance on purely synthetic aromatic systems also limits the structural diversity available for optimizing the fit within the narrow hydrophobic pocket of the HDAC active site. Consequently, there is a pressing need in cost reduction in API manufacturing for platforms that offer greater structural complexity and inherent bioactivity without compromising synthetic feasibility.
The Novel Approach
The methodology outlined in the patent addresses these challenges by employing beta-elemene as a bioactive CAP group. This sesquiterpene possesses a rigid cyclohexane skeleton with three chiral centers and multiple pi bonds, providing a high electron cloud density and a specific spatial volume that mimics the reported hydrophobic regions of HDAC inhibitors. By coupling this natural scaffold with various linking units and zinc-binding groups (ZBGs), the inventors have created a versatile library of compounds. The synthetic strategy avoids the need for complex aromatic functionalization, instead relying on efficient N-alkylation and amidation reactions. This shift not only simplifies the supply chain by utilizing readily available natural product extracts but also potentially enhances the therapeutic index of the resulting drugs through synergistic anticancer mechanisms.
Mechanistic Insights into Beta-Elemene Functionalization and Linker Assembly
The core of this technology lies in the precise functionalization of the beta-elemene skeleton to attach the pharmacophore. The process begins with the conversion of beta-elemene into a reactive monochloro-intermediate, which then undergoes nucleophilic substitution with piperazine to form the key beta-elemene-piperazine core (Intermediate D-10). This core serves as the foundation for all four series of inhibitors. For the aliphatic series (Series A), the synthesis involves N-alkylation with amino acid derivatives followed by hydrolysis or aminolysis to install the linker and ZBG. This route is particularly advantageous for creating flexible linkers that can adapt to the enzyme's binding channel.
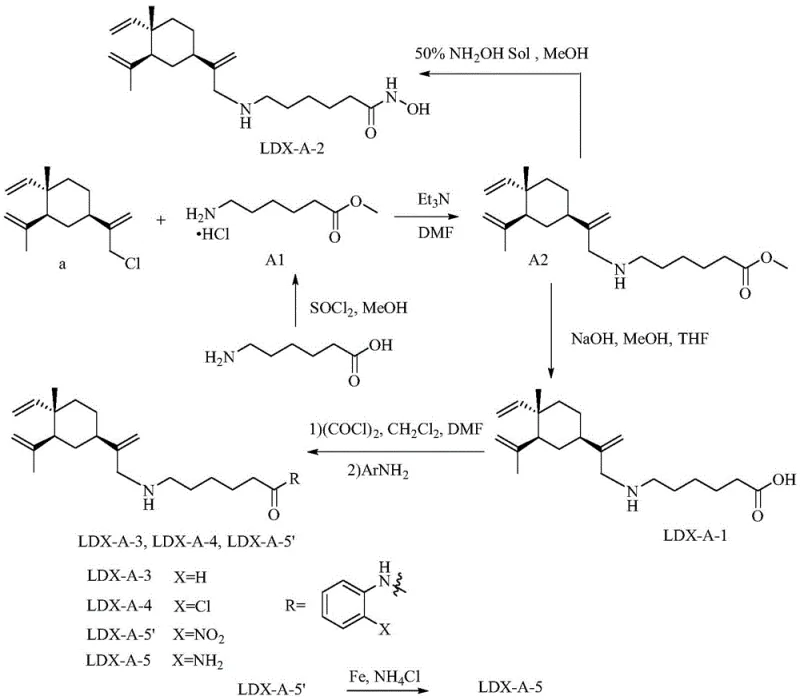
In contrast, the aromatic series (Series B and C) utilize more rigid linkers to probe different regions of the HDAC active site. Series B employs a carbamate linkage formed via CDI activation, connecting the piperazine core to a benzyl alcohol derivative, followed by Knoevenagel condensation to extend the conjugation. Series C utilizes a direct acrylamide linkage formed through Knoevenagel-Doebner condensation of terephthalaldehyde derivatives. These diverse chemistries allow for fine-tuning of the molecule's physicochemical properties, such as solubility and metabolic stability. The ability to switch between aliphatic and aromatic linkers while maintaining the same beta-elemene CAP group demonstrates the modularity of this platform, making it an ideal candidate for a reliable pharmaceutical intermediates supplier looking to offer a broad portfolio of epigenetic modulators.
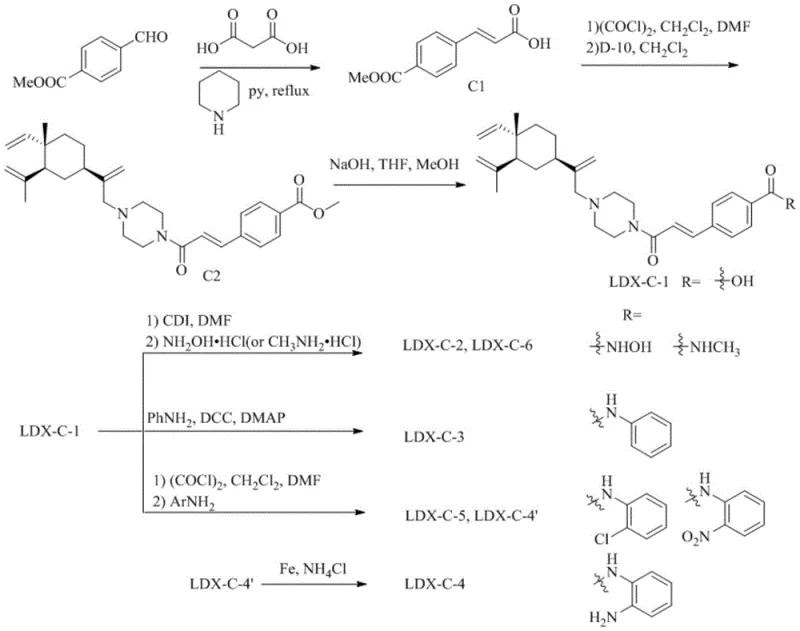
How to Synthesize Beta-Elemene HDAC Inhibitor Intermediates Efficiently
The synthesis of these novel inhibitors follows a logical progression starting from the abundant natural product beta-elemene. The initial chlorination and piperazine substitution steps are critical for establishing the chiral integrity of the CAP group. Subsequent steps involve standard peptide coupling or esterification techniques to attach the linker-ZBG moiety. The patent provides detailed experimental conditions, including solvent systems like DMF and dichloromethane, and reagents such as oxalyl chloride and CDI, which are staples in fine chemical manufacturing. For a detailed breakdown of the specific reaction parameters and workup procedures required to achieve high purity, please refer to the standardized guide below.
- Preparation of monochloro-beta-elemene intermediate via reaction with sodium hypochlorite in dichloromethane.
- Nucleophilic substitution of the chloro-intermediate with anhydrous piperazine to form the beta-elemene-piperazine core.
- Coupling the core with aliphatic or aromatic linkers followed by functionalization with zinc-binding groups like hydroxamic acids or amides.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of beta-elemene-based intermediates offers several strategic advantages over traditional synthetic scaffolds. The primary raw material, beta-elemene, is derived from natural sources (Curcuma wenyujin), ensuring a sustainable and potentially lower-cost feedstock compared to petrochemical-derived aromatics. The synthetic routes described avoid the use of precious metal catalysts like palladium or platinum, which are subject to volatile pricing and strict residual metal limits in pharmaceutical products. This elimination of expensive catalytic steps translates directly into substantial cost savings and simplified downstream processing.
- Cost Reduction in Manufacturing: The synthetic pathways rely on commodity chemicals such as piperazine, malonic acid, and common amino acids, which are available in bulk quantities at stable prices. By avoiding complex cross-coupling reactions that require specialized ligands and inert atmospheres, the overall process mass intensity (PMI) is reduced. The use of standard solvents and ambient pressure reactions further lowers the capital expenditure required for equipment, facilitating a more economical production model for commercial scale-up of complex oncology intermediates.
- Enhanced Supply Chain Reliability: Diversifying the source of the CAP group from synthetic aromatics to a natural product extract mitigates the risk associated with petrochemical supply fluctuations. The robustness of the N-alkylation and amidation steps ensures high reproducibility across different batches, which is critical for maintaining consistent quality in GMP environments. Furthermore, the modular nature of the synthesis allows for rapid adaptation; if a specific ZBG becomes unavailable, the core intermediate can be easily coupled with alternative linkers, ensuring continuity of supply for your drug development programs.
- Scalability and Environmental Compliance: The reactions described operate under mild conditions, typically ranging from room temperature to moderate reflux, which minimizes energy consumption. The absence of heavy metal catalysts significantly reduces the burden on wastewater treatment facilities, aligning with increasingly stringent environmental regulations. The final purification steps involve standard column chromatography or recrystallization, techniques that are easily transferable from laboratory to pilot plant scales, thereby reducing lead time for high-purity pharmaceutical intermediates entering clinical trials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these beta-elemene derivatives. Understanding these aspects is crucial for integrating this technology into your existing drug discovery pipeline. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for your decision-making process.
Q: What is the structural advantage of using beta-elemene in HDAC inhibitors?
A: Beta-elemene serves as a bulky, hydrophobic CAP group that mimics the surface recognition region of traditional inhibitors but offers unique steric properties and inherent anticancer activity, potentially enhancing selectivity for Class I HDACs.
Q: Are the synthesis routes suitable for large-scale manufacturing?
A: Yes, the described methods utilize common solvents like dichloromethane, DMF, and methanol, and standard reactions such as N-alkylation and amidation, which are highly amenable to commercial scale-up without requiring exotic catalysts.
Q: What types of Zinc Binding Groups (ZBG) are compatible with this scaffold?
A: The platform supports diverse ZBGs including hydroxamic acids, amides, and fatty acid derivatives, allowing for the tuning of potency and selectivity profiles against specific HDAC isoforms.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Elemene Derivatives Supplier
The integration of beta-elemene into HDAC inhibitor scaffolds represents a sophisticated convergence of natural product chemistry and medicinal design. At NINGBO INNO PHARMCHEM, we possess the technical expertise to navigate the complexities of terpenoid functionalization and amide bond formation. Our facility is equipped to handle the specific solvent systems and purification requirements necessary for these sensitive intermediates. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from preclinical research to clinical supply is seamless. Our stringent purity specifications and rigorous QC labs guarantee that every batch meets the exacting standards required for oncology drug development.
We invite you to collaborate with us to optimize these synthetic routes for your specific needs. Whether you require custom analogs with modified linkers or large-scale production of the core intermediates, our team is ready to assist. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your project timelines and reduce overall development costs.
