Optimizing Ceritinib Intermediate Production via Efficient One-Pot Catalytic Oxidation
Optimizing Ceritinib Intermediate Production via Efficient One-Pot Catalytic Oxidation
The pharmaceutical industry's relentless pursuit of more efficient synthetic routes for oncology treatments has brought significant attention to the preparation of key intermediates for Anaplastic Lymphoma Kinase (ALK) inhibitors. Specifically, the patent CN106854182B, published in early 2020, outlines a groundbreaking preparation method for 2,5-dichloro-N-(2-(isopropylsulfonyl)phenyl)pyrimidin-4-amine, a critical building block in the synthesis of Ceritinib. This advanced protocol addresses longstanding inefficiencies in the manufacturing of this complex heterocyclic compound by integrating a streamlined one-pot strategy that combines nucleophilic substitution and catalytic oxidation. By leveraging specific rare metal catalysts such as sodium tungstate, the process achieves exceptional purity levels exceeding 99% while drastically reducing energy consumption and operational complexity. For R&D directors and process chemists, this represents a pivotal shift towards greener, more economically viable manufacturing paradigms that do not compromise on the stringent quality standards required for active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations detailed in CN106854182B, the synthesis of this pyrimidine derivative was plagued by significant operational hurdles that impacted both cost and throughput. Existing literature, such as the methodology described in WO2016138648, relied on multi-step procedures that were not only labor-intensive but also suffered from poor reaction safety profiles and extended processing times. A critical bottleneck in these conventional routes was the excessive reaction duration, often requiring up to 38 hours to reach completion, which severely limited reactor turnover rates and increased utility costs. Furthermore, the traditional approaches often necessitated harsh conditions or complex work-up procedures to isolate intermediates, leading to cumulative yield losses and the generation of substantial chemical waste. These factors collectively created a supply chain vulnerability, making it difficult for manufacturers to guarantee consistent availability of high-purity intermediates for downstream API production without incurring prohibitive expenses.
The Novel Approach
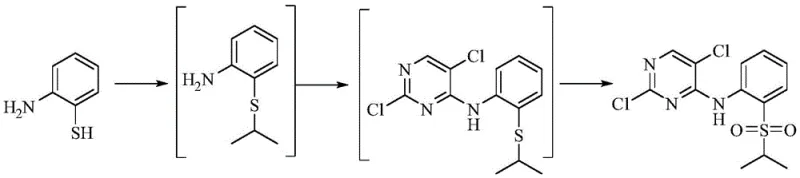
In stark contrast to these legacy methods, the novel approach introduced in the patent utilizes a sophisticated one-pot synthesis strategy that elegantly consolidates multiple transformation steps into a single reaction vessel. This methodology initiates with the nucleophilic substitution of 2-aminothiophenol with bromoisopropane under controlled alkaline conditions, followed immediately by coupling with 2,4,5-trichloropyrimidine without the need for intermediate isolation. The process culminates in a highly selective oxidation step mediated by a rare metal catalyst, converting the sulfide moiety directly to the requisite sulfone group. By eliminating intermediate purification stages, this route not only accelerates the overall timeline but also minimizes material loss and solvent usage. The result is a robust, scalable process capable of delivering yields as high as 87.2% with purity specifications meeting the rigorous demands of global regulatory bodies, thereby establishing a new benchmark for efficiency in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Tungstate-Catalyzed Oxidative Sulfonylation
The chemical elegance of this synthesis lies in its precise control over reactivity, particularly during the final oxidative transformation which is critical for defining the biological activity of the final drug substance. The mechanism begins with the deprotonation of the thiol group in 2-aminothiophenol by a base such as sodium carbonate or triethylamine, generating a highly nucleophilic thiolate anion that attacks bromoisopropane to form the thioether linkage. Subsequently, the remaining amino group acts as a nucleophile attacking the electron-deficient C4 position of the 2,4,5-trichloropyrimidine ring, displacing a chloride ion to establish the core pyrimidine-amine scaffold. The final and most crucial stage involves the oxidation of the sulfur atom from the sulfide state to the sulfone state using hydrogen peroxide activated by a tungsten-based catalyst. This catalytic cycle ensures that the oxidation proceeds selectively to the sulfone without over-oxidizing sensitive functional groups or degrading the pyrimidine ring, a common failure mode in non-catalytic oxidation attempts.
Furthermore, the choice of solvent plays a pivotal role in stabilizing the transition states and ensuring homogeneous reaction conditions throughout the prolonged heating periods. The patent highlights the efficacy of polar aprotic solvents like acetonitrile or moderately polar esters like ethyl acetate, which facilitate the dissolution of both organic substrates and inorganic bases. The use of sodium tungstate or sodium phosphotungstate as the catalyst creates a peroxo-tungsten species in situ upon addition of 30% hydrogen peroxide, which acts as the active oxygen transfer agent. This mechanistic pathway allows the reaction to proceed at moderate temperatures, typically around 60°C, preventing thermal degradation of the product. Understanding these mechanistic nuances is essential for process chemists aiming to replicate this success on a commercial scale, as slight deviations in catalyst loading or peroxide addition rates could impact the impurity profile and overall yield of the final crystalline product.
How to Synthesize 2,5-dichloro-N-(2-(isopropylsulfonyl)phenyl)pyrimidin-4-amine Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters regarding temperature gradients and reagent addition sequences to maximize the formation of the desired sulfone while minimizing side reactions. The process is designed to be operator-friendly, utilizing standard reflux equipment and common laboratory reagents that are readily available in most chemical manufacturing facilities. Detailed below is the standardized operational framework derived from the patent examples, which serves as a guideline for achieving the reported high purity and yield metrics. Operators should note that the exothermic nature of the oxidation step requires careful monitoring, and the final recrystallization from ethanol is critical for removing trace metal residues and ensuring the product meets pharmaceutical grade specifications.
- Perform nucleophilic substitution of 2-aminothiophenol with bromoisopropane under alkaline conditions at reflux.
- Without isolation, react the intermediate with 2,4,5-trichloropyrimidine to form the pyrimidine core structure.
- Execute catalytic oxidation using sodium tungstate and hydrogen peroxide to convert the sulfide group to the final sulfone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this patented methodology offers profound implications for cost structures and inventory reliability within the oncology drug supply chain. The consolidation of multiple reaction steps into a single pot dramatically reduces the number of unit operations required, which directly translates to lower labor costs, reduced equipment occupancy time, and decreased consumption of auxiliary materials like filtration media and drying agents. Moreover, the significant reduction in total reaction time—from nearly two days in older methods to roughly one day—enhances asset utilization rates, allowing manufacturers to respond more agilely to fluctuating market demands for ALK inhibitors. This efficiency gain is not merely theoretical; it is grounded in the tangible simplification of the workflow, which reduces the probability of human error and batch failures that often plague complex multi-step syntheses.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation and purification steps removes the need for expensive chromatography or extensive washing procedures, leading to substantial savings in solvent procurement and waste disposal fees. By utilizing cost-effective catalysts like sodium tungstate instead of precious metals, the process avoids the volatility associated with noble metal pricing and eliminates the need for complex metal scavenging technologies. Additionally, the high yield reported in the patent examples means that less raw material is required to produce the same amount of finished goods, effectively lowering the cost of goods sold (COGS) per kilogram. These cumulative savings create a competitive pricing advantage that can be passed down the supply chain or retained as improved margin.
- Enhanced Supply Chain Reliability: The robustness of this one-pot method ensures consistent batch-to-batch quality, which is paramount for maintaining uninterrupted API production schedules. Because the reaction conditions are mild and the reagents are commodity chemicals, the risk of supply disruption due to specialized raw material shortages is significantly mitigated. The ability to scale this process from gram-scale R&D to hundred-kilogram production without fundamental changes to the chemistry provides supply chain heads with the confidence to commit to long-term contracts. This stability is crucial for pharmaceutical companies managing the complex logistics of bringing life-saving cancer therapies to market, where any delay in intermediate supply can have cascading effects on clinical trial timelines or commercial launch dates.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, the shorter reaction times and lower operating temperatures reduce the facility's overall energy footprint and carbon emissions. The use of hydrogen peroxide as the terminal oxidant generates water as the primary byproduct, aligning with green chemistry principles and simplifying wastewater treatment protocols compared to methods using heavy metal oxidants. The process is explicitly designed for industrialization, meaning that engineering controls for heat dissipation and gas evolution are well-understood and easily implemented at scale. This compliance readiness accelerates the regulatory approval process for the manufacturing site, ensuring that the supply of this critical intermediate remains uninterrupted by environmental audits or safety incidents.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the experimental data and claims within the patent documentation. These insights are intended to clarify the operational feasibility and strategic value of adopting this technology for large-scale production. By understanding the specific advantages related to catalyst selection and reaction kinetics, stakeholders can make informed decisions about integrating this process into their existing manufacturing portfolios.
Q: What are the primary advantages of the method described in CN106854182B compared to prior art?
A: The method significantly reduces reaction time from 38 hours to approximately 20-26 hours total, utilizes a safer one-pot procedure, and achieves higher yields (up to 87.2%) with purity exceeding 98.9%.
Q: Which catalysts are preferred for the oxidation step in this synthesis?
A: The patent specifies rare metal catalysts such as sodium tungstate, tungstic acid, or sodium phosphotungstate, with sodium tungstate being particularly effective for selective oxidation.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the invention explicitly claims suitability for industrialization and hundred-kilogram scale production due to its mild conditions, simple operation, and use of common solvents like ethyl acetate or acetonitrile.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,5-dichloro-N-(2-(isopropylsulfonyl)phenyl)pyrimidin-4-amine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and a commitment to quality excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity profiles demonstrated in CN106854182B are faithfully reproduced in our facilities. We operate with stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest international standards. Our capability to handle complex catalytic oxidations and sensitive heterocyclic chemistry makes us the ideal strategic partner for securing your supply of this vital oncology intermediate.
We invite you to engage with our technical team to discuss how this optimized synthesis can drive value for your specific project needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this more efficient route. We encourage you to contact our technical procurement team today to obtain specific COA data and comprehensive route feasibility assessments tailored to your volume requirements, ensuring a seamless and cost-effective supply chain for your next-generation therapeutics.
