Scalable Production of High-Purity Acridine Inner Salts for Advanced Chemiluminescence Diagnostics
Scalable Production of High-Purity Acridine Inner Salts for Advanced Chemiluminescence Diagnostics
The landscape of in vitro diagnostics (IVD) is rapidly shifting towards chemiluminescence immunoassays (CLIA) due to their superior sensitivity and stability compared to traditional enzyme-linked methods. At the heart of this technology lies the acridine ester family of chemiluminescent reagents, specifically acridine inner salts, which serve as critical labeling markers for antigens and antibodies. However, the widespread adoption of these reagents has historically been bottlenecked by complex synthesis routes, low yields, and the reliance on prohibitively expensive coupling agents. Patent CN111170994B introduces a transformative preparation method that addresses these industrial pain points by substituting high-cost activators with economical alternatives and optimizing solvent systems for facile purification. This technical breakthrough not only enhances the atom economy of the synthesis but also enables a robust crystallization process, marking a significant leap forward for manufacturers seeking reliable chemiluminescence reagent suppliers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for preparing acridine inner salts, such as those disclosed in earlier patents like CN201310250582.4, suffer from severe inefficiencies that hinder commercial viability. The most glaring issue is the reliance on O-(N-Succinimidyl)-N,N,N',N'-tetramethyluronium tetrafluoroborate (TSTU) as the activating agent. TSTU is an extremely costly reagent that significantly inflates the bill of materials, making the final product economically unfeasible for high-volume diagnostic applications. Furthermore, these legacy processes typically utilize dimethylformamide (DMF) as the reaction solvent. While DMF is a powerful polar aprotic solvent, its high boiling point and strong solvating power create a nightmare for downstream processing. In the conventional method, the target acridine inner salt remains dissolved in the DMF matrix as a viscous oil even after reaction completion. This physical state prevents standard separation techniques like filtration or simple crystallization, forcing manufacturers to resort to preparative chromatography. Chromatography is inherently batch-limited, solvent-intensive, and difficult to scale, resulting in low throughput and inconsistent purity profiles that are unacceptable for GMP-grade pharmaceutical intermediates.
The Novel Approach
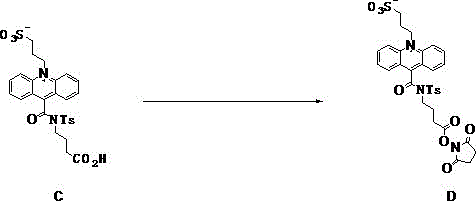
The methodology outlined in patent CN111170994B fundamentally re-engineers the synthesis workflow to prioritize scalability and cost-efficiency. The core innovation lies in the strategic replacement of TSTU with N-hydroxysuccinimide (NHS), activated in situ by N,N'-carbonyldiimidazole (CDI). NHS is a commodity chemical with a fraction of the cost of TSTU, offering immediate and substantial raw material savings without compromising reactivity. More critically, the process switches the reaction medium to acetonitrile. Unlike DMF, acetonitrile exhibits a differential solubility profile: it dissolves the activated intermediate sufficiently to allow the reaction to proceed but has poor solubility for the final zwitterionic product. This property is exploited to drive the product out of solution as a solid precipitate directly upon reaction completion or upon the addition of an anti-solvent like tert-butyl methyl ether. This shift from an "oily residue requiring chromatography" to a "crystalline solid amenable to filtration" is a game-changer for process chemistry, enabling simple isolation, washing, and drying steps that are easily transferable to multi-kilogram reactors.
Mechanistic Insights into CDI-Mediated Esterification
To fully appreciate the robustness of this synthesis, one must examine the mechanistic underpinnings of the CDI-mediated activation. The reaction begins with the carboxylic acid moiety on the propyl side chain of the acridine precursor (Compound C). When CDI is introduced into the acetonitrile solution, it reacts with the carboxylic acid to form an unstable acyl imidazole intermediate, releasing imidazole as a byproduct. This acyl imidazole is a highly reactive electrophile, far more susceptible to nucleophilic attack than the parent acid. Upon the subsequent addition of N-hydroxysuccinimide, the hydroxyl group of the NHS acts as a nucleophile, attacking the carbonyl carbon of the acyl imidazole. This displaces the imidazole leaving group and forms the stable N-hydroxysuccinimide ester (Compound D). The choice of acetonitrile is mechanistically sound because it stabilizes the polar transition states while not interfering with the electrophilic centers. Furthermore, the zwitterionic nature of the acridine inner salt, possessing both a sulfonate anion and an acridinium cation, creates a highly polar structure that is naturally insoluble in moderately polar solvents like acetonitrile once the lipophilic protecting groups or precursors are transformed, driving the precipitation thermodynamics.
Impurity control is another critical aspect where this mechanism excels. In the old DMF-based routes, side reactions such as hydrolysis of the active ester or decomposition of the acridine ring could occur due to prolonged heating or difficulty in removing moisture from the hygroscopic DMF. The new method operates effectively at room temperature (20-40°C), minimizing thermal degradation risks. Additionally, the crystallization step serves as a powerful purification mechanism. As the product crystallizes from the acetonitrile matrix, impurities such as unreacted starting material, urea byproducts from CDI, or excess NHS remain in the mother liquor due to their higher solubility. This "self-purifying" aspect of the crystallization ensures that the isolated solid achieves high purity levels (>98% by HPLC) without the need for iterative chromatographic passes, which often lead to product loss and increased waste generation.

How to Synthesize Acridine Inner Salt Efficiently
The synthesis of the target acridine inner salt (Compound D) is the final and most critical step in the overall manufacturing chain, transforming the purified carboxylic acid precursor (Compound C) into the bioconjugatable active ester. This procedure leverages the unique solubility characteristics of acetonitrile to bypass complex purification bottlenecks. The process begins by dissolving the zwitterionic acid precursor in dry acetonitrile, followed by activation with CDI to generate the reactive acyl species. Once activation is confirmed, N-hydroxysuccinimide is introduced to capture the activated intermediate. The reaction progress is monitored to ensure complete conversion before inducing precipitation. For a detailed breakdown of the operational parameters, stoichiometry, and workup procedures required to replicate this high-yield transformation, please refer to the standardized synthesis guide below.
- Dissolve the acridine carboxylic acid precursor (Compound C) in acetonitrile and activate with CDI at room temperature.
- Add N-hydroxysuccinimide (NHS) to the reaction mixture and stir until the active ester formation is complete.
- Induce crystallization by adding a non-polar anti-solvent such as tert-butyl methyl ether, followed by filtration and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from the legacy TSTU/DMF process to the NHS/Acetonitrile protocol represents a significant opportunity for cost optimization and risk mitigation. The primary driver of value is the drastic reduction in raw material expenditure. By eliminating TSTU, a specialized and expensive coupling reagent, and replacing it with bulk-available NHS, the direct material cost per kilogram of product is significantly reduced. This cost structure improvement is compounded by the elimination of chromatographic purification. Column chromatography requires vast quantities of high-purity solvents and silica gel, along with significant labor hours for operation and solvent recovery. Replacing this with a simple filtration and wash step drastically cuts utility consumption, waste disposal costs, and processing time, leading to a leaner and more cost-effective manufacturing operation.
- Cost Reduction in Manufacturing: The economic benefits of this new route extend beyond simple reagent substitution. The ability to isolate the product via crystallization rather than chromatography means that the process intensity is lowered. There is no longer a need for expensive preparative HPLC columns or the associated downtime for column packing and equilibration. The solvents used, primarily acetonitrile and tert-butyl methyl ether, are common industrial solvents with well-established recovery and recycling streams, further enhancing the economic efficiency. Moreover, the higher atom economy of using NHS compared to the bulky TSTU reagent means less chemical waste is generated per unit of product, reducing the environmental compliance burden and associated disposal fees.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commoditized reagents. TSTU is a niche chemical that may face supply volatility or long lead times from specialized vendors. In contrast, N-hydroxysuccinimide and CDI are produced by multiple global suppliers, ensuring a stable and competitive supply base. The robustness of the crystallization process also improves batch-to-batch consistency. In the old process, the isolation of an oil often led to variable purities and difficulties in defining specifications. The new process yields a defined crystalline solid with a sharp melting point and consistent X-ray diffraction pattern, simplifying quality control and ensuring that every shipment meets the stringent requirements of diagnostic kit manufacturers.
- Scalability and Environmental Compliance: From a scale-up perspective, this process is inherently safer and more manageable. The exotherm of the CDI activation is manageable in acetonitrile, and the room temperature conditions reduce the energy load on the facility. The elimination of DMF is particularly noteworthy from an environmental, health, and safety (EHS) standpoint. DMF is a reproductive toxin subject to increasingly strict exposure limits and discharge regulations globally. By switching to acetonitrile, the facility reduces its regulatory risk profile and simplifies wastewater treatment protocols. The high yields reported in the patent examples, maintaining efficiency even when scaling from milligrams to 100 grams, suggest that the kinetics and mass transfer characteristics of the reaction are favorable for large-scale tank reactors, facilitating a smooth transition from pilot plant to commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of acridine inner salts using this novel methodology. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering clarity on process capabilities and product quality. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their diagnostic supply chains.
Q: Why is acetonitrile preferred over DMF for this synthesis?
A: Acetonitrile allows for direct crystallization of the product due to lower solubility, whereas DMF retains the product in solution as an oil, necessitating difficult chromatographic purification.
Q: What is the cost advantage of using NHS over TSTU?
A: N-hydroxysuccinimide is significantly less expensive than TSTU and offers better atom economy, drastically reducing raw material costs for large-scale manufacturing.
Q: Can this process be scaled to industrial quantities?
A: Yes, the patent demonstrates successful scaling from milligram to 100g batches with consistent high purity (>98%) and yield, indicating strong potential for metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acridine Inner Salt Supplier
As the demand for high-sensitivity chemiluminescence diagnostics continues to surge, securing a stable supply of high-quality acridine inner salts is paramount for diagnostic manufacturers. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced synthetic methodologies like the one described in CN111170994B to deliver superior intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We operate under stringent purity specifications and utilize rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and potency of every batch, guaranteeing that our acridine derivatives perform consistently in your final assay kits.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes, demonstrating how our efficient crystallization-based process can improve your margins. Please contact our technical procurement team today to request specific COA data for our acridine inner salt inventory and to discuss route feasibility assessments for your next-generation diagnostic projects.
