Revolutionizing Azaphilone Production: Advanced Biocatalysis for Commercial Scale
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to produce bioactive compounds with higher efficiency and purity. A significant breakthrough in this domain is documented in patent CN113308443B, which details a novel Monascus monooxygenase mutant capable of catalyzing the synthesis of azaphilone derivatives with unprecedented efficiency. Azaphilones, characterized by their unique pyranoquinone bicyclic structure, possess valuable medical properties including anti-cancer, anti-bacterial, and anti-inflammatory activities, making them highly sought-after intermediates for drug development. Traditionally, the supply of these compounds has been constrained by the limitations of natural extraction, but this new biocatalytic approach offers a transformative solution for reliable azaphilone supplier networks seeking to stabilize their supply chains. By leveraging specific protein engineering techniques, this technology addresses the critical bottlenecks of low catalytic activity and unstable yields that have historically plagued the production of these complex molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the procurement of azaphilone compounds has relied heavily on extraction from natural plant sources or fermentation using wild-type microorganisms, both of which present substantial logistical and economic challenges for a procurement manager. Plant-based extraction is inherently vulnerable to external environmental factors such as climate change, seasonal variations, and geographic limitations, leading to significant fluctuations in both yield and quality consistency. Furthermore, the cultivation of specific plant species requires extensive land use and long growth cycles, which drastically increases the lead time for high-purity azaphilones and complicates inventory planning for large-scale manufacturers. When relying on wild-type microbial fermentation, the catalytic efficiency of the native enzymes is often suboptimal, resulting in low conversion rates that necessitate complex and costly downstream purification processes to remove impurities. These inefficiencies translate directly into higher manufacturing costs and a fragile supply chain that struggles to meet the rigorous demands of the global pharmaceutical market.
The Novel Approach
In stark contrast to these traditional methods, the technology disclosed in patent CN113308443B introduces a genetically engineered monooxygenase mutant that fundamentally reshapes the economics of azaphilone manufacturing. By specifically mutating the arginine at the 176th site of the wild-type monooxygenase to alanine, the inventors have created a biocatalyst with dramatically improved kinetic properties and substrate specificity. This novel approach allows for the direct conversion of precursor substrates into the target azaphilone product with a reaction efficiency that is more than double that of the wild-type enzyme, effectively bypassing the yield ceilings of conventional fermentation. The result is a streamlined production process that not only enhances cost reduction in pharmaceutical intermediate manufacturing but also ensures a consistent, high-quality output that is independent of agricultural variables. This shift represents a move towards a more resilient and predictable supply model, essential for supply chain heads managing global distribution networks.
Mechanistic Insights into R176A Monooxygenase Catalysis
The core of this technological advancement lies in the precise structural modification of the enzyme's active site, which facilitates a more efficient condensation of carbonyl and aldehyde groups within the substrate molecule. The substitution of the bulky arginine residue with a smaller alanine residue at position 176 likely reduces steric hindrance within the catalytic pocket, allowing for better substrate binding and faster turnover rates during the oxidation process. This mechanistic optimization is critical for R&D directors focused on impurity profiles, as the enhanced specificity of the mutant enzyme minimizes the formation of side products that typically complicate purification. The reaction proceeds under mild conditions, utilizing NADPNa as a cofactor in a PBS buffer system, which preserves the integrity of the sensitive pyranoquinone structure while maximizing the conversion of 2-aldehyde-acetophenone. Understanding this catalytic cycle is essential for scaling the process, as it highlights the enzyme's robustness and its capacity to maintain high activity over extended reaction periods without significant degradation.
Furthermore, the high selectivity of this mutant enzyme directly contributes to the exceptional purity of the final product, which is reported to exceed 99% after separation and purification. This level of purity is paramount for applications in drug synthesis, where trace impurities can have profound effects on the safety and efficacy of the final therapeutic agent. The ability to achieve such high purity standards simplifies the downstream processing workflow, reducing the need for extensive chromatography or recrystallization steps that often drive up production costs and time. By controlling the reaction at the molecular level through protein engineering, manufacturers can ensure a clean impurity spectrum that meets the stringent regulatory requirements of international health authorities. This mechanistic control transforms the production of azaphilones from an art into a precise science, enabling the commercial scale-up of complex biocatalytic intermediates with confidence.
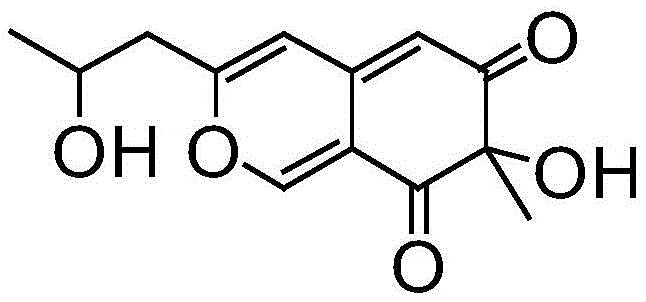
How to Synthesize Azaphilone Efficiently
Implementing this synthesis route requires a systematic approach to genetic engineering and fermentation optimization to fully realize the potential of the mutant monooxygenase. The process begins with the construction of the expression strain, where the mutant gene is cloned into a suitable vector such as pET30a(+) and transformed into an Escherichia coli host for high-level protein expression. Following induction with IPTG, the enzyme is harvested and purified using nickel affinity chromatography, ensuring that only the active biocatalyst is introduced into the reaction system. The detailed standardized synthesis steps see the guide below, which outlines the precise conditions for substrate dissolution, cofactor addition, and reaction monitoring to achieve the reported 92.8% yield. Adhering to these protocols is essential for reproducing the high efficiency and purity demonstrated in the patent data.
- Extract wild-type monooxygenase coding DNA from Monascus and perform site-directed mutagenesis at position 176 (Arginine to Alanine) to create the recombinant plasmid.
- Transform the recombinant plasmid into Escherichia coli host cells and induce expression using IPTG at 18°C to produce the mutant enzyme.
- React the purified mutant enzyme with 2-aldehyde-acetophenone substrate in PBS buffer with NADPNa cofactor at 35°C to achieve 92.8% conversion yield.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this enzymatic technology offers a compelling value proposition centered around stability, efficiency, and long-term cost optimization. By shifting from variable plant extraction to controlled microbial fermentation, companies can secure a reliable azaphilone supplier relationship that is immune to the vagaries of weather and harvest cycles. The dramatic increase in reaction yield from 44.2% to 92.8% implies a substantial reduction in raw material consumption per unit of product, which directly correlates to significant cost savings in manufacturing operations without the need for expensive capital investment in new infrastructure. Additionally, the simplified purification process resulting from high product purity reduces the consumption of solvents and energy, further enhancing the environmental profile and economic viability of the production line. These factors combine to create a supply chain that is not only more cost-effective but also more agile and responsive to market demands.
- Cost Reduction in Manufacturing: The elimination of inefficient wild-type strains and the adoption of the high-yield mutant enzyme drastically reduces the cost per kilogram of the final product by maximizing substrate conversion. Since the process avoids the use of expensive transition metal catalysts and relies on renewable biological systems, the operational expenditure is significantly lowered while maintaining high throughput. This efficiency gain allows for competitive pricing strategies in the global market, making high-purity azaphilone more accessible for broader pharmaceutical applications. The qualitative improvement in yield means that less waste is generated, reducing disposal costs and enhancing the overall sustainability of the manufacturing process.
- Enhanced Supply Chain Reliability: Unlike plant-based sourcing which is subject to seasonal disruptions and geographic constraints, this microbial fermentation method can be performed year-round in controlled industrial facilities. This consistency ensures that lead times for high-purity azaphilones are predictable and can be optimized to meet just-in-time delivery schedules required by large pharmaceutical clients. The use of standard E. coli expression systems also means that the technology can be easily transferred between manufacturing sites, providing redundancy and security against localized production failures. This reliability is a critical asset for supply chain heads tasked with maintaining continuous operations in a volatile global market.
- Scalability and Environmental Compliance: The biocatalytic nature of this process aligns perfectly with green chemistry principles, as it operates under mild conditions and generates less hazardous waste compared to traditional chemical synthesis. Scaling this process from laboratory to industrial levels is facilitated by the well-understood fermentation technologies associated with E. coli, allowing for rapid capacity expansion from 100 kgs to 100 MT scales. The reduced need for harsh solvents and the high selectivity of the enzyme minimize the environmental footprint, ensuring compliance with increasingly strict international environmental regulations. This scalability ensures that the supply can grow in tandem with market demand without compromising on quality or sustainability standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this mutant monooxygenase technology in industrial settings. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation, providing clarity on the feasibility and advantages of the new method. Understanding these details is crucial for stakeholders evaluating the integration of this biocatalytic route into their existing production portfolios. The data supports a strong case for the superiority of this method over conventional extraction techniques in terms of both performance and economic viability.
Q: How does the mutant monooxygenase improve azaphilone yield compared to wild-type?
A: The mutant enzyme, featuring an R176A amino acid substitution, demonstrates significantly enhanced catalytic activity, increasing reaction yield from 44.2% in the wild-type strain to 92.8%, while maintaining product purity above 99%.
Q: What are the advantages of enzymatic synthesis over plant extraction for azaphilones?
A: Enzymatic synthesis eliminates dependency on climate, plant species, and planting areas, ensuring consistent supply continuity and stable quality, whereas plant extraction is subject to agricultural variability and lower yields.
Q: Is this biocatalytic process suitable for large-scale industrial manufacturing?
A: Yes, the process utilizes Escherichia coli expression systems which are well-established for industrial fermentation, allowing for scalable production with simplified downstream purification due to high selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azaphilone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this enzymatic synthesis route and are fully equipped to bring this technology from patent to commercial reality. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are designed to handle complex biocatalytic processes with stringent purity specifications, supported by rigorous QC labs that verify every batch against the highest international standards. We understand that the transition to a new manufacturing technology requires a partner who can navigate the complexities of process optimization and regulatory compliance with expertise and dedication.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this advanced enzymatic technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to contact us to request specific COA data and route feasibility assessments that will demonstrate the tangible benefits of partnering with us. By leveraging our expertise in fine chemical intermediates, we can help you secure a competitive edge in the market through superior product quality and supply chain resilience.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
