Revolutionizing Flame Retardant Manufacturing: Continuous High-Purity BDP Synthesis
The global demand for high-performance, non-halogenated flame retardants has surged, particularly driven by the rapid expansion of the new energy vehicle sector and the stringent safety requirements for engineering plastics like PC/ABS and PPO/HIPS. In response to these market pressures, patent CN112409404B introduces a groundbreaking continuous industrial production method for high-purity bisphenol A-bis(diphenyl phosphate), commonly known as BDP. This technology represents a paradigm shift from traditional intermittent batch synthesis to a sophisticated continuous flow process, addressing critical pain points regarding product consistency, impurity profiles, and operational safety. By implementing a split-feeding strategy for bisphenol A and utilizing a multistage crosslinking reactor system, this method achieves a product yield exceeding 97% while maintaining a color number below 50. For R&D directors and procurement specialists seeking a reliable flame retardant supplier, this patent outlines a pathway to securing a supply chain that is not only more robust but also capable of delivering ultra-low impurity levels, specifically controlling triphenyl phosphate (TPP) to 0.1% and isopropenyl phenyl diphenyl phosphate to less than 50 ppm.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the manufacturing of bisphenol A-bis(diphenyl phosphate) has relied heavily on intermittent batch synthesis, a methodology fraught with inherent inefficiencies and quality control challenges. In traditional batch reactors, the simultaneous addition of all reactants often leads to uneven heat distribution and localized concentration spikes, which trigger undesirable side reactions. Specifically, the presence of solid bisphenol A in large quantities within the phosphorus oxychloride medium slows down the reaction kinetics, necessitating higher temperatures and longer retention times that promote the decomposition of bisphenol A into allyl phenol. This decomposition is the root cause of persistent impurities such as isopropenyl phenyl diphenyl phosphate, which compromises the thermal stability and color of the final polymer application. Furthermore, batch processes are labor-intensive and prone to safety hazards; the frequent opening and closing of reactors increase the risk of leakage and operator exposure to corrosive gases, while the batch-to-batch variability makes it difficult to guarantee the stringent purity specifications required by top-tier automotive and electronics manufacturers.
The Novel Approach
The continuous industrial production method disclosed in the patent fundamentally reengineers the synthesis pathway to eliminate these systemic flaws through precise process control and reactor design. Instead of a single charge, bisphenol A is strategically divided into two portions, with 70-80% reacting initially with phosphorus oxychloride and a Lewis acid catalyst in a continuous multistage crosslinking reactor. This staged approach ensures that the reaction proceeds under optimal kinetic conditions without the mass transfer limitations associated with solid accumulation. Following the removal of excess phosphorus oxychloride via film evaporation, the remaining bisphenol A is introduced along with an organic base, driving the crosslinking reaction to completion while neutralizing generated hydrogen chloride. This is followed by a continuous multistage esterification with phenol, where consumed phenol is dynamically replenished to maintain reaction equilibrium. The result is a seamless, closed-loop production system that drastically reduces energy consumption, minimizes human intervention, and delivers a finished product with exceptional purity and stability, setting a new benchmark for cost reduction in polymer additive manufacturing.
Mechanistic Insights into Lewis Acid-Catalyzed Continuous Phosphorylation
The core chemical innovation lies in the manipulation of reaction kinetics through the split-feeding mechanism and the utilization of Lewis acid catalysis, specifically anhydrous magnesium chloride, to facilitate the phosphorylation of bisphenol A. In the initial crosslinking stage, the Lewis acid activates the phosphorus oxychloride, making it more susceptible to nucleophilic attack by the hydroxyl groups of the first portion of bisphenol A. By maintaining the reaction temperature between 80°C and 120°C across four distinct reactor stages, the process ensures a gradual and controlled conversion, preventing the thermal degradation that plagues batch systems. The subsequent addition of the second portion of bisphenol A in the presence of an organic amine, such as triethylamine, serves a dual purpose: it acts as a reactant to complete the oligomerization and simultaneously functions as an acid scavenger. This immediate neutralization of hydrogen chloride gas shifts the chemical equilibrium towards the product side, effectively suppressing the reverse reaction and preventing the formation of chlorinated byproducts that could act as precursors to color bodies.
Furthermore, the continuous esterification stage employs a dynamic replenishment strategy for phenol, which is critical for minimizing the formation of triphenyl phosphate (TPP), a common and detrimental impurity. In conventional methods, the depletion of phenol towards the end of the reaction allows unreacted phosphorus species to self-condense or react incompletely, leading to TPP accumulation. By continuously supplying fresh phenol into the second, third, and fourth stages of the esterification reactor, the system maintains a high molar excess of the capping agent throughout the entire residence time. This ensures that all terminal acyl chloride groups are efficiently capped with phenoxy groups, resulting in the target bis(diphenyl phosphate) structure with minimal end-group defects. The rigorous control over stoichiometry and residence time in this continuous flow environment is what enables the suppression of isopropenyl phenyl diphenyl phosphate to trace levels below 50 ppm, a specification that is vital for preventing discoloration in high-end engineering plastics.
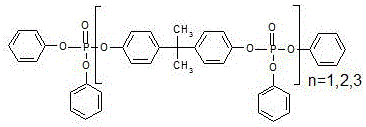
How to Synthesize Bisphenol A-bis(diphenyl phosphate) Efficiently
The synthesis of high-purity BDP via this continuous method requires precise coordination of feed rates, temperature gradients, and pressure controls across multiple reactor units. The process begins with the premixing of phosphorus oxychloride, the Lewis acid catalyst, and the majority of the bisphenol A feed, which is then pumped into a series of heated reactors operating under slight negative pressure to facilitate the removal of gaseous byproducts. Following the initial crosslinking, the stream passes through a thin-film evaporator to strip unreacted phosphorus oxychloride before entering the secondary reaction zone for final oligomerization with the remaining bisphenol and organic base. The resulting intermediate is then merged with phenol for the final esterification sequence, after which the crude product undergoes a continuous purification train involving acid washing to remove catalyst residues, alkali washing to neutralize acidic components, and water washing to eliminate salts.
- Premix 70-80% of bisphenol A with phosphorus oxychloride and a Lewis acid catalyst, then feed into a continuous multistage crosslinking reactor at 80-120°C.
- Remove excess phosphorus oxychloride via film evaporation, then react the residue with the remaining 20-30% bisphenol A and an organic base at 115-120°C.
- Perform continuous multi-stage esterification with phenol replenishment, followed by sequential acid, alkali, and water washing to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from batch to continuous processing described in this patent offers profound strategic advantages beyond mere technical specifications. The most significant impact is seen in the realm of operational efficiency and cost structure; by eliminating the stop-start nature of batch production, the facility can operate at a steady state, maximizing asset utilization and significantly reducing the labor intensity associated with charging, discharging, and cleaning individual reactors. This continuous operation inherently stabilizes the quality of the output, meaning that downstream customers receive material with consistent viscosity and impurity profiles, thereby reducing the need for extensive incoming quality control testing and minimizing the risk of production line stoppages due to off-spec raw materials. Moreover, the closed-system design drastically mitigates the environmental and safety risks associated with handling volatile and corrosive reagents, leading to lower insurance premiums and reduced regulatory compliance burdens.
- Cost Reduction in Manufacturing: The implementation of this continuous process drives down manufacturing costs through several qualitative mechanisms that do not rely on arbitrary percentage claims. By integrating the reaction and purification steps into a unified flow, the need for intermediate storage tanks and the associated pumping operations is eliminated, which directly lowers energy consumption and equipment maintenance costs. Additionally, the high selectivity of the reaction minimizes the formation of heavy ends and tar-like byproducts, which increases the overall mass balance efficiency and reduces the volume of waste solvent that requires treatment or disposal. The ability to recover and recycle solvents continuously within the line further enhances the economic viability, ensuring that the cost reduction in polymer additive manufacturing is sustainable and driven by fundamental process intensification rather than temporary market fluctuations.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the continuous nature of this production method provides a level of predictability that batch processing simply cannot match. Because the system is designed to run for extended periods without interruption, it creates a steady, uninterrupted stream of finished goods, allowing for more accurate inventory planning and shorter lead times for order fulfillment. The robustness of the process against minor fluctuations in raw material quality—due to the buffering capacity of the multistage reactors—ensures that supply continuity is maintained even when facing variations in feedstock. This reliability is crucial for just-in-time manufacturing environments in the automotive and electronics sectors, where any disruption in the supply of critical flame retardants can halt entire assembly lines, making this method a cornerstone for building a resilient supply network.
- Scalability and Environmental Compliance: The modular design of the multistage reactor system allows for straightforward scalability, enabling the commercial scale-up of complex oligomeric flame retardants from pilot volumes to full industrial capacity with minimal re-engineering. Unlike batch reactors where heat transfer becomes a limiting factor as vessel size increases, continuous flow reactors maintain a high surface-area-to-volume ratio, ensuring that thermal control remains precise regardless of production volume. This scalability is paired with superior environmental performance; the closed-loop system prevents the fugitive emissions of hydrogen chloride and phosphorus oxychloride vapors that are common in open batch operations, thereby simplifying the requirements for scrubber systems and ensuring compliance with increasingly stringent global environmental regulations regarding volatile organic compounds and hazardous air pollutants.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the continuous production of bisphenol A-bis(diphenyl phosphate), derived directly from the comparative data and process descriptions within the patent literature. These insights are intended to clarify how the novel continuous methodology overcomes the specific impurity challenges and safety limitations inherent in legacy batch technologies. Understanding these distinctions is essential for technical teams evaluating the feasibility of integrating this high-purity grade into their existing polymer formulations.
Q: How does the continuous process reduce triphenyl phosphate (TPP) impurities compared to batch methods?
A: The continuous process utilizes a split-feeding strategy for bisphenol A and continuous phenol replenishment during esterification. This precise control prevents the excessive local concentration of reactants that typically leads to side reactions forming TPP, reducing content from >1% in batch processes to <0.1%.
Q: What represents the primary safety advantage of this continuous industrial method?
A: Unlike intermittent batch synthesis which poses risks of leakage and requires significant manual intervention, this continuous method operates in closed multistage reactors with negative pressure. This drastically reduces operator exposure to hazardous chemicals like phosphorus oxychloride and eliminates the 'bubble leakage' phenomena common in traditional plants.
Q: Why is controlling isopropenyl phenyl diphenyl phosphate critical for product quality?
A: Isopropenyl phenyl diphenyl phosphate is a harmful impurity that causes product discoloration upon illumination and instability in the final polymer matrix. The patented method controls this impurity to below 50 ppm by optimizing the reaction temperature and using an organic base to neutralize hydrogen chloride, preventing the decomposition of bisphenol A into allyl phenol precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bisphenol A-bis(diphenyl phosphate) Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced continuous manufacturing processes is key to meeting the evolving demands of the global polymer industry. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN112409404B are fully realized in practical, large-scale operations. Our facilities are equipped with state-of-the-art continuous flow reactors and rigorous QC labs capable of verifying stringent purity specifications, including the sub-50 ppm impurity thresholds required for high-end automotive applications. We are committed to delivering high-purity bisphenol A-bis(diphenyl phosphate) that not only meets but exceeds international standards for flame retardancy and thermal stability.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains and reduce total cost of ownership. By leveraging our technical expertise, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and formulation needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of superior material quality and supply chain security.
