Scaling Green Fenazaquin Production: Technical Insights for Global Agrochemical Leaders
The global agrochemical industry is constantly seeking sustainable manufacturing pathways that balance efficiency with environmental stewardship, a challenge directly addressed by the innovative technology disclosed in patent CN102558073A. This patent introduces a groundbreaking green synthesis method for fenazaquin, a potent quinazoline-based miticide widely used to protect crops such as almonds, apples, and cotton from spider mites. Unlike conventional processes that rely on hazardous chlorine gas and generate substantial solid waste, this novel approach leverages advanced chlorinating agents like phosphorus oxychloride, thionyl chloride, or bis(trichloromethyl) carbonate in the presence of a DMF catalyst. For R&D directors and procurement managers alike, this represents a significant opportunity to optimize the production of high-purity agrochemical intermediates while mitigating the regulatory and disposal costs associated with traditional phosphine-based chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of fenazaquin has relied on a pathway that utilizes triphenylphosphine and chlorine gas to activate the hydroxyl group of the quinazoline ring. As illustrated in the reaction scheme below, this traditional method involves the formation of a reactive phosphine-chlorine complex which subsequently chlorinates the substrate. However, this stoichiometric reaction inevitably produces one mole of triphenylphosphine oxide solid waste for every mole of product generated. This solid byproduct not only complicates the filtration and purification steps but also creates a significant burden on wastewater treatment facilities due to the difficulty in degrading phosphine oxides. Furthermore, the handling of elemental chlorine gas poses severe safety risks and requires specialized containment infrastructure, increasing both capital expenditure and operational complexity for manufacturers.
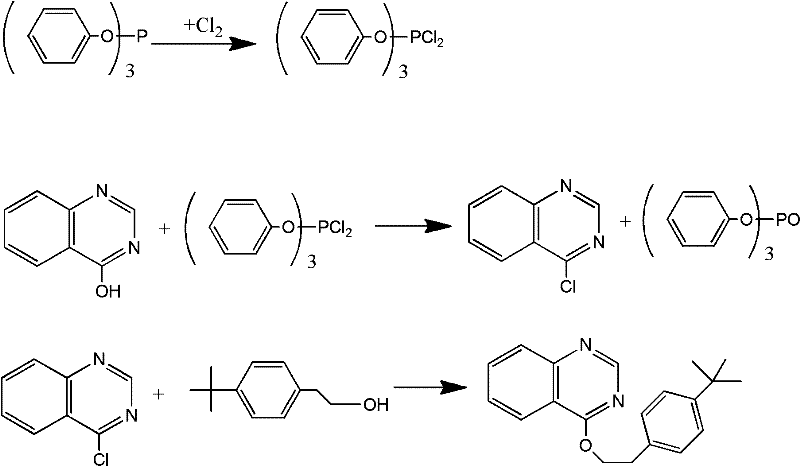
The Novel Approach
In stark contrast, the methodology described in CN102558073A circumvents these issues by employing soluble chlorinating reagents that do not generate intractable solid waste. The new process initiates with the dissolution of 4-hydroxyquinazoline in common organic solvents like dichloromethane or dichloroethane, catalyzed by a small amount of N,N-dimethylformamide (DMF). The subsequent addition of reagents such as bis(trichloromethyl) carbonate or phosphorus oxychloride facilitates a clean conversion to the 4-chloroquinazoline intermediate. Crucially, excess chlorinating agents can be removed simply by heating and distillation, leaving a reaction mixture that is ready for the final etherification step with p-t-butylphenethyl alcohol. This streamlined workflow eliminates the need for filtering tons of phosphine oxide sludge, thereby simplifying the technical process and significantly reducing the generation of hazardous wastewater.
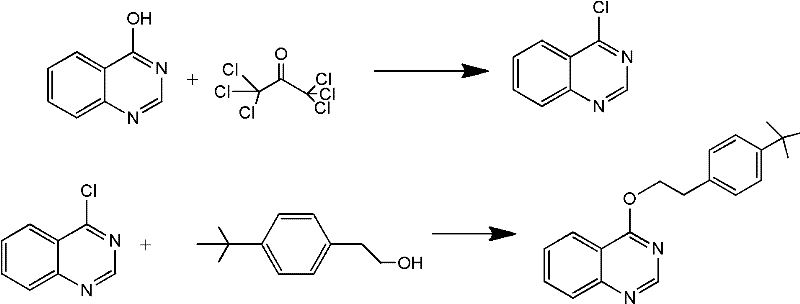
Mechanistic Insights into DMF-Catalyzed Chlorination and Etherification
The core of this technological breakthrough lies in the efficient activation of the hydroxyl group on the quinazoline ring through a Vilsmeier-Haack type mechanism or direct nucleophilic substitution facilitated by the DMF catalyst. When DMF interacts with chlorinating agents like thionyl chloride or bis(trichloromethyl) carbonate, it forms a highly reactive iminium salt intermediate (often referred to as the Vilsmeier reagent). This electrophilic species aggressively attacks the oxygen of the 4-hydroxyquinazoline, converting the poor leaving group (-OH) into an excellent leaving group, which is then displaced by a chloride ion to form the 4-chloroquinazoline. This mechanistic pathway is far more atom-economical than the phosphine-mediated route because the byproducts are typically gaseous (such as CO2 and SO2 from triphosgene or thionyl chloride) or volatile liquids that can be easily separated, ensuring that the reaction mass remains homogeneous and easy to handle throughout the synthesis.
Following the chlorination, the process seamlessly transitions to the etherification stage without the need for isolating the sensitive chloro-intermediate, which is prone to hydrolysis. The reaction mixture, still containing the activated species, is treated with p-t-butylphenethyl alcohol at controlled temperatures ranging from 0°C to 100°C. The nucleophilic oxygen of the alcohol attacks the electron-deficient carbon at the 4-position of the quinazoline ring, displacing the chloride and forming the final fenazaquin ether linkage. The patent data indicates that maintaining strict temperature control during this exothermic step is critical for minimizing side reactions and ensuring high purity. By avoiding the isolation of the intermediate, the process reduces exposure to moisture and air, thereby suppressing the formation of hydrolysis impurities and enhancing the overall yield and quality of the final agrochemical active ingredient.
How to Synthesize Fenazaquin Efficiently
To implement this green synthesis route effectively, manufacturers must adhere to precise operational parameters regarding reagent addition and temperature management. The process begins by suspending 4-hydroxyquinazoline in a dry organic solvent and adding the DMF catalyst, followed by the controlled dropwise addition of the chosen chlorinating agent. Detailed standardized synthetic steps, including specific molar ratios and reflux times for different chlorinating reagents, are outlined in the technical guide below to ensure reproducibility and safety during scale-up.
- Dissolve 4-hydroxyquinazoline in an organic solvent such as dichloromethane and add a catalytic amount of N,N-dimethylformamide (DMF).
- Dropwise add a chlorinating reagent (e.g., POCl3, SOCl2, or bis(trichloromethyl) carbonate) at controlled temperatures between -5°C and 120°C to form the chloro-intermediate.
- Remove excess chlorinating agent by heating, then react the mixture with p-t-butylphenethyl alcohol at 0-100°C, followed by washing and recrystallization to obtain pure fenazaquin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this green synthesis method offers profound strategic benefits that extend beyond mere regulatory compliance. By shifting away from the legacy triphenylphosphine-chlorine route, companies can achieve substantial cost savings through the elimination of expensive phosphine reagents and the associated costs of solid waste disposal. The simplified workup procedure, which relies on distillation and washing rather than complex filtration of solids, drastically reduces the cycle time per batch, allowing for higher throughput and better utilization of existing reactor capacity. This efficiency gain translates directly into improved supply chain reliability, ensuring that critical agrochemical intermediates can be delivered to customers with reduced lead times and greater consistency.
- Cost Reduction in Manufacturing: The economic impact of removing triphenylphosphine oxide generation cannot be overstated, as it eliminates the need for purchasing costly phosphine reagents and paying for the hazardous waste treatment of the resulting solid byproduct. Additionally, the ability to recover and recycle excess chlorinating agents like phosphorus oxychloride through distillation further lowers the raw material consumption per kilogram of product. This leaner material profile results in a significantly reduced cost of goods sold (COGS), providing a competitive pricing advantage in the global agrochemical market without compromising on quality or purity standards.
- Enhanced Supply Chain Reliability: The reagents utilized in this novel process, such as thionyl chloride, phosphorus oxychloride, and DMF, are commodity chemicals with robust and stable global supply chains, unlike specialized phosphine derivatives which may face availability fluctuations. The simplified process flow also reduces the risk of production delays caused by equipment clogging from solid waste or extended filtration times. Consequently, manufacturers can maintain a more consistent production schedule, ensuring uninterrupted supply of fenazaquin to downstream formulators and protecting against market volatility caused by production bottlenecks.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns perfectly with modern green chemistry principles by minimizing waste at the source rather than treating it post-generation. The absence of toxic chlorine gas handling and the reduction in wastewater load make the process easier to permit and scale in regions with stringent environmental regulations. This future-proofs the manufacturing asset, allowing for seamless commercial scale-up of complex quinazoline derivatives from pilot plant quantities to multi-ton annual production without the need for major infrastructure upgrades for waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of how this method compares to incumbent technologies in terms of yield, safety, and operational feasibility.
Q: How does this green synthesis method reduce environmental impact compared to traditional routes?
A: Traditional methods utilize triphenylphosphine and chlorine gas, generating stoichiometric amounts of solid triphenylphosphine oxide waste and requiring hazardous gas handling. The patented green method replaces these with liquid or soluble chlorinating agents (like POCl3 or triphosgene) that can be distilled off or washed away, drastically reducing solid waste generation and wastewater treatment loads.
Q: What are the key advantages for supply chain stability when adopting this route?
A: This route utilizes widely available industrial reagents such as phosphorus oxychloride and thionyl chloride, avoiding the supply bottlenecks associated with specialized phosphine reagents. Furthermore, the simplified workup procedure, which eliminates the filtration of solid phosphine oxide byproducts, shortens the production cycle time and enhances overall throughput reliability.
Q: Can this process be scaled for commercial production of high-purity fenazaquin?
A: Yes, the process is designed for scalability. The reaction conditions allow for temperature control between -5°C and 120°C, and the removal of excess reagents via distillation is a standard unit operation in chemical plants. The resulting product achieves high purity (up to 99% in optimized examples) through simple recrystallization, making it suitable for large-scale agrochemical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fenazaquin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting sustainable and efficient manufacturing technologies to meet the evolving demands of the global agrochemical sector. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and robust. We are committed to delivering high-purity fenazaquin and related quinazoline intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities.
We invite you to collaborate with us to leverage this advanced green synthesis route for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can drive value and sustainability for your organization.
