Advanced Photochemical Synthesis of 9-Benzylated Fluorenol Derivatives for Commercial Scale-Up
The recent disclosure of patent CN115611713A introduces a groundbreaking synthesis method for 9-benzylated fluorenol derivatives, addressing critical needs in the optoelectronic materials sector. This technology leverages visible light photocatalysis to facilitate the coupling of toluene derivatives with fluorenone under exceptionally mild conditions, marking a significant departure from traditional thermal or metal-catalyzed processes. The method operates in an air atmosphere at room temperature, utilizing 12W blue light irradiation to drive the reaction efficiently without the need for expensive noble metal catalysts. This innovation is particularly relevant for the production of high-purity intermediates used in polymer light-emitting diodes and photovoltaic cells, where impurity profiles are strictly controlled. By enabling a greener and more economical pathway, this patent provides a robust foundation for scalable manufacturing of complex fluorene skeletons. 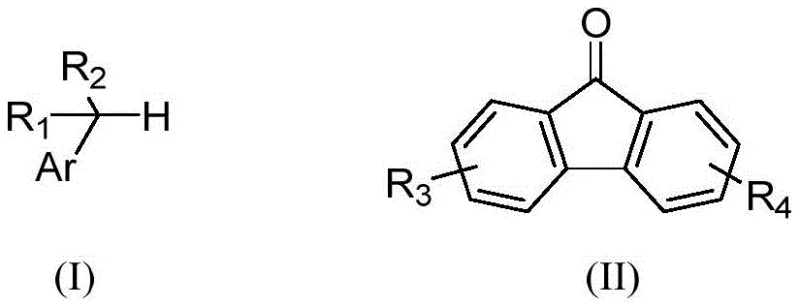
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 9-benzylated fluorenol derivatives has relied heavily on the Grignard reaction, a process known for its stringent operational requirements and significant safety hazards. This traditional approach necessitates strictly anhydrous conditions and the use of reactive organometallic reagents, which generate substantial amounts of metal salt waste that is environmentally detrimental. Furthermore, the sensitivity of Grignard reagents to moisture and oxygen often leads to inconsistent yields and complicates the purification process, increasing overall production costs. Alternative methods such as carboxylic acid decarboxylation radical coupling have been explored, yet these often suffer from moderate yields and the generation of carbon dioxide by-products that require careful management. 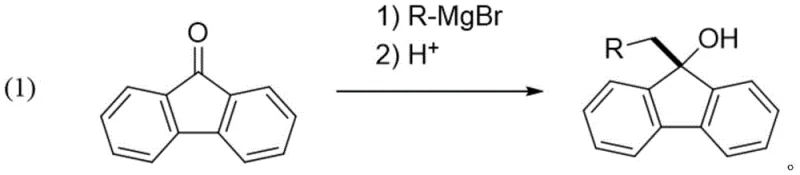
The Novel Approach
In stark contrast, the novel photochemical method disclosed in the patent utilizes renewable blue light to activate the reactants, eliminating the need for harsh thermal conditions or toxic metal catalysts. This approach allows the reaction to proceed smoothly in common organic solvents like acetonitrile under ambient air pressure, drastically simplifying the equipment setup and operational protocol. The absence of noble metals not only reduces raw material costs but also removes the necessity for complex downstream metal scavenging steps, streamlining the entire manufacturing workflow. High conversion rates are achieved with minimal by-product formation, ensuring that the final fluorenol derivatives meet the stringent purity standards required for electronic applications. This shift towards photocatalysis represents a paradigm change in how fine chemical intermediates are produced, prioritizing sustainability and economic efficiency.
Mechanistic Insights into Blue Light Photocatalyzed Coupling
The core of this technological advancement lies in the unique radical mechanism initiated by blue light irradiation, which generates reactive species without external photocatalysts. Upon exposure to 12W blue light, the fluorenone substrate absorbs photons to form a transient diradical species, which is the key active intermediate in this transformation. This excited state species then engages in a hydrogen atom transfer (HAT) process with the toluene derivative, effectively abstracting a hydrogen atom to generate a benzyl radical and a fluorenol alpha-carbon radical simultaneously. The precise control over this radical generation is critical, as it prevents uncontrolled polymerization or side reactions that typically plague radical chemistry in complex organic synthesis. 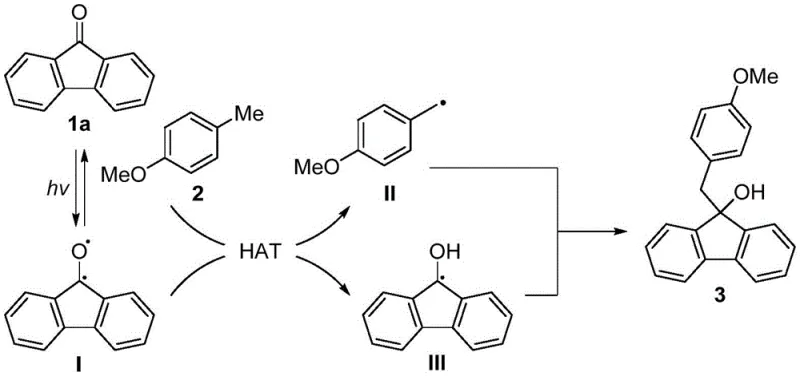
Following the hydrogen atom transfer, the resulting benzyl radical and fluorenol alpha-carbon radical undergo a selective radical-radical coupling to form the desired carbon-carbon bond. This coupling step is highly efficient due to the proximity and reactivity of the generated radical species within the solvent cage, leading to the formation of the 9-benzylated fluorenol skeleton with high regioselectivity. The use of an inorganic base such as cesium fluoride further facilitates the reaction by stabilizing intermediates and promoting the deprotonation steps necessary for the cycle to continue. Understanding this mechanism is vital for R&D teams aiming to optimize reaction parameters for specific substituted derivatives, as the electronic nature of the substituents on the toluene and fluorenone rings can influence the radical stability. This deep mechanistic understanding ensures that the process can be reliably scaled while maintaining the high purity and yield observed in laboratory examples.
How to Synthesize 9-Benzylated Fluorenol Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry of reactants and the intensity of the light source to maximize efficiency. The standard protocol involves dissolving the toluene derivative and fluorenone in a polar aprotic solvent like acetonitrile, followed by the addition of one equivalent of an inorganic base such as cesium fluoride. The reaction mixture is then subjected to continuous stirring under 12W blue light irradiation for approximately 24 hours at room temperature, ensuring complete conversion of the starting materials. Detailed standardized synthesis steps see the guide below.
- Dissolve toluene derivatives, fluorenone, and an equivalent of inorganic base in an organic solvent such as acetonitrile.
- React the mixture under 12W blue light irradiation in an air atmosphere at 25°C for 24 hours.
- Perform post-treatment via reduced pressure concentration and column chromatography to isolate the pure 9-benzylated fluorenol derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement and supply chain professionals, this patent offers a compelling value proposition by addressing several critical pain points associated with traditional fine chemical manufacturing. The elimination of noble metal catalysts directly translates to significant cost reductions, as there is no need to purchase expensive palladium or rhodium complexes or invest in specialized metal removal resins. Furthermore, the ability to run the reaction in air at room temperature reduces energy consumption associated with heating and inert gas purging, leading to lower utility costs per kilogram of product. The simplicity of the equipment, requiring only standard glassware and commercial blue light reactors, minimizes capital expenditure for new production lines and reduces maintenance overheads. These factors combine to create a more resilient and cost-effective supply chain for high-value optoelectronic intermediates.
- Cost Reduction in Manufacturing: The removal of noble metal catalysts from the synthesis route eliminates a major cost driver, as these metals are not only expensive to purchase but also require costly recovery or disposal processes. By utilizing simple inorganic bases and organic solvents, the raw material bill is significantly optimized, allowing for better margin management in competitive markets. Additionally, the mild reaction conditions reduce the energy load on the facility, as there is no need for high-temperature heating or cryogenic cooling systems. This cumulative effect results in substantial cost savings that can be passed down the supply chain or reinvested into further R&D initiatives.
- Enhanced Supply Chain Reliability: The use of readily available toluene derivatives and fluorenone as starting materials ensures a stable supply of raw inputs, reducing the risk of shortages associated with specialized reagents. The robustness of the reaction in an air atmosphere means that production is less susceptible to disruptions caused by inert gas supply failures or moisture control issues. This reliability is crucial for maintaining consistent delivery schedules to downstream customers in the electronics and pharmaceutical sectors. Consequently, supply chain managers can plan inventory levels with greater confidence, knowing that the production process is less prone to unexpected technical stoppages.
- Scalability and Environmental Compliance: The environmentally friendly nature of this process, which avoids toxic metal waste and harsh reagents, simplifies compliance with increasingly strict environmental regulations. The absence of heavy metal by-products reduces the burden on wastewater treatment facilities and lowers the costs associated with hazardous waste disposal. Moreover, the simplicity of the reaction setup facilitates easy scale-up from laboratory to commercial production volumes without the need for complex engineering modifications. This scalability ensures that the supply can grow in tandem with market demand for fluorene-based optoelectronic materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photochemical synthesis method. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation to ensure accuracy. Understanding these details is essential for stakeholders evaluating the feasibility of adopting this new technology for their specific production needs.
Q: What are the primary advantages of this method over conventional Grignard reactions?
A: This photochemical method operates under mild conditions at 25°C in air, eliminating the need for strict anhydrous environments and expensive noble metal catalysts required by Grignard processes, thereby significantly reducing operational complexity and waste.
Q: Does this synthesis route require expensive transition metal catalysts?
A: No, the process utilizes renewable blue light irradiation to drive the reaction without any noble metal catalysts, which removes the need for costly metal removal steps and reduces heavy metal contamination risks in the final product.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method features simple equipment requirements such as standard glass reactors and 12W blue light sources, along with high conversion rates and environmentally friendly profiles, making it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9-Benzylated Fluorenol Supplier
NINGBO INNO PHARMCHEM stands ready to leverage this advanced photochemical technology to deliver high-quality 9-benzylated fluorenol derivatives to the global market. As a specialized CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch meets the exacting standards required for electronic material applications. We understand the critical nature of supply continuity in the optoelectronic industry and are committed to providing a reliable source for these complex intermediates.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits of switching to this metal-free process. We encourage you to contact us for specific COA data and route feasibility assessments to validate the performance of our materials in your downstream applications. Together, we can drive efficiency and innovation in the production of next-generation fluorene derivatives.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
