Scalable Synthesis of Novel Cinnamaldehyde-Phenanthroimidazole Derivatives for Oncology Drug Development
Introduction to Advanced Antitumor Intermediate Technology
The pharmaceutical industry is constantly seeking novel scaffolds that can overcome drug resistance and reduce systemic toxicity in cancer therapy. Patent CN114671870B introduces a significant breakthrough in this domain by disclosing a series of cinnamaldehyde-modified phenanthroimidazole derivatives. These compounds represent a strategic fusion of two pharmacologically active motifs: the rigid, planar phenanthroimidazole core known for its intercalation properties and the bioactive cinnamaldehyde side chain. This structural hybridization results in molecules that demonstrate potent inhibitory effects on tumor cell proliferation, specifically targeting lung, liver, and breast cancer lines, while exhibiting remarkably low cytotoxicity against normal cells. The innovation lies not only in the biological profile but also in the accessibility of the chemical structure, which is defined by the general Formula I shown below. 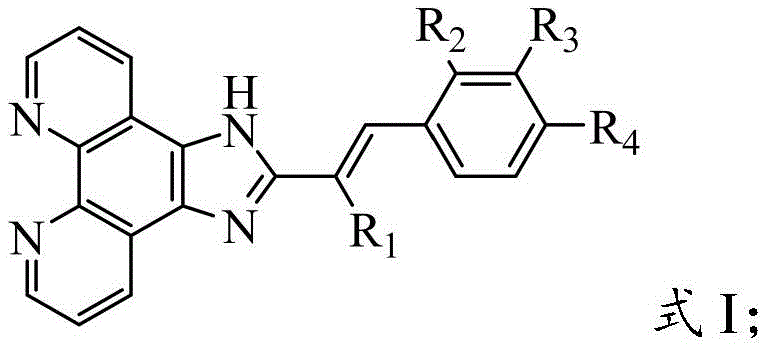 For R&D directors and procurement specialists, understanding the versatility of the R1, R2, R3, and R4 substituents is crucial, as they allow for fine-tuning the physicochemical properties and metabolic stability of the final drug candidate. This patent provides a robust foundation for developing next-generation oncology therapeutics that balance efficacy with safety.
For R&D directors and procurement specialists, understanding the versatility of the R1, R2, R3, and R4 substituents is crucial, as they allow for fine-tuning the physicochemical properties and metabolic stability of the final drug candidate. This patent provides a robust foundation for developing next-generation oncology therapeutics that balance efficacy with safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of complex heterocyclic systems like phenanthroimidazoles often involves multi-step sequences that require harsh reaction conditions, expensive transition metal catalysts, or protection-deprotection strategies that lower overall atom economy. Conventional routes to functionalized imidazoles may suffer from poor regioselectivity, leading to difficult-to-separate isomeric mixtures that complicate purification and reduce final yield. Furthermore, many existing methods rely on solvents or reagents that pose significant environmental and safety hazards, increasing the cost of waste disposal and regulatory compliance. In the context of large-scale API manufacturing, these inefficiencies translate into higher production costs and longer lead times, creating bottlenecks in the supply chain for critical oncology intermediates. The reliance on precious metal catalysts also introduces the risk of heavy metal contamination, necessitating additional purification steps to meet stringent pharmaceutical standards.
The Novel Approach
In contrast, the methodology described in CN114671870B offers a streamlined, one-pot condensation strategy that dramatically simplifies the synthetic landscape. By reacting 1,10-phenanthroline-5,6-dione directly with various cinnamaldehyde derivatives in the presence of ammonium acetate and glacial acetic acid, the process constructs the imidazole ring and installs the functional side chain simultaneously. This approach eliminates the need for pre-functionalized imidazole precursors or complex coupling reactions. The reaction conditions are mild yet effective, typically operating between 30°C and 110°C, which is compatible with standard glass-lined or stainless-steel reactors found in most chemical plants. The use of ammonium acetate as a nitrogen source is particularly advantageous due to its low cost and ease of handling compared to gaseous ammonia or hazardous amines. This novel route not only accelerates the timeline from bench to pilot plant but also enhances the purity profile of the crude product, facilitating easier downstream processing.
Mechanistic Insights into Acid-Catalyzed Condensation Cyclization
The core of this synthetic transformation is a variation of the Debus-Radziszewski imidazole synthesis, adapted for a fused polycyclic system. The mechanism initiates with the activation of the carbonyl groups on the 1,10-phenanthroline-5,6-dione by the acidic medium (glacial acetic acid). The ammonium acetate serves as an ammonia equivalent, attacking one of the carbonyls to form an imine intermediate. Subsequently, the nucleophilic attack by the amine nitrogen on the aldehyde carbon of the cinnamaldehyde derivative, followed by cyclization and dehydration, closes the imidazole ring. 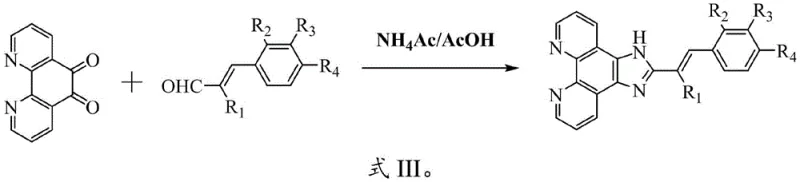 This cascade reaction is highly efficient because the aromatic stabilization of the newly formed phenanthroimidazole system drives the equilibrium towards the product. The presence of electron-withdrawing or electron-donating groups on the cinnamaldehyde phenyl ring (R2, R3, R4) can influence the electrophilicity of the aldehyde, but the robust nature of the reaction conditions ensures high conversion across a broad substrate scope. From a process chemistry perspective, understanding this mechanism allows for precise control over reaction kinetics; for instance, maintaining the temperature at 110°C ensures complete consumption of the starting dione, minimizing the formation of half-reacted intermediates that could act as difficult-to-remove impurities.
This cascade reaction is highly efficient because the aromatic stabilization of the newly formed phenanthroimidazole system drives the equilibrium towards the product. The presence of electron-withdrawing or electron-donating groups on the cinnamaldehyde phenyl ring (R2, R3, R4) can influence the electrophilicity of the aldehyde, but the robust nature of the reaction conditions ensures high conversion across a broad substrate scope. From a process chemistry perspective, understanding this mechanism allows for precise control over reaction kinetics; for instance, maintaining the temperature at 110°C ensures complete consumption of the starting dione, minimizing the formation of half-reacted intermediates that could act as difficult-to-remove impurities.
Impurity control is inherently built into this mechanism through the precipitation workup. Upon completion of the reaction, the mixture is cooled and diluted with water. The addition of concentrated ammonia water to adjust the pH to neutral (pH 7) causes the basic phenanthroimidazole product to precipitate out of the acidic solution, while many polar byproducts and excess ammonium salts remain in the aqueous phase. This pH-switch isolation technique is a powerful tool for bulk purification, effectively removing the majority of non-basic impurities before any chromatographic steps are considered. For R&D teams, this means that the crude solid obtained after filtration is already of significant purity, reducing the load on final purification columns and improving the overall mass balance of the process. The ability to isolate the product simply by filtration rather than extraction also reduces solvent consumption, aligning with green chemistry principles.
How to Synthesize Phenanthroimidazole Derivatives Efficiently
To implement this synthesis in a laboratory or pilot setting, operators must adhere to specific stoichiometric and thermal parameters to maximize yield and reproducibility. The patent outlines a clear protocol where the molar ratio of the phenanthroline dione to the cinnamaldehyde derivative is optimized, typically favoring a slight excess of the aldehyde to drive the reaction to completion. The detailed standardized synthesis steps, including precise weighing, addition rates, and temperature ramping profiles, are critical for ensuring batch-to-b consistency.
- Mix 1,10-phenanthroline-5,6-dione, specific cinnamaldehyde compounds, and ammonium acetate in glacial acetic acid solvent.
- Heat the mixture to reflux temperatures between 30°C and 110°C for 0.5 to 4 hours to facilitate condensation.
- Cool the reaction, adjust pH to 7 with ammonia water to precipitate the product, then filter, wash, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of this patent are substantial. The shift towards a catalyst-free, one-pot synthesis directly addresses several pain points in the sourcing of complex pharmaceutical intermediates. By eliminating the need for expensive palladium or copper catalysts, the bill of materials (BOM) is significantly reduced, and the supply chain becomes less vulnerable to fluctuations in precious metal prices. Furthermore, the simplicity of the reaction setup means that production can be outsourced to a wider range of CDMOs that possess standard reflux capabilities, rather than being restricted to facilities with specialized high-pressure or inert atmosphere equipment. This flexibility enhances supply security and reduces the risk of production delays due to equipment bottlenecks.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the necessity for costly metal scavenging resins and extensive analytical testing for residual metals, which are mandatory for API release. Additionally, the use of glacial acetic acid as both solvent and catalyst is economically favorable; it is a commodity chemical with a stable global supply, and its recovery and recycling are well-established industrial processes. The high yields reported (up to 90% in examples) mean that less raw material is wasted, directly lowering the cost of goods sold (COGS) and improving the margin profile for the final drug product.
- Enhanced Supply Chain Reliability: The starting materials, 1,10-phenanthroline-5,6-dione and various substituted cinnamaldehydes, are commercially available from multiple global suppliers, preventing single-source dependency. The robustness of the reaction conditions (tolerance to air and moisture compared to organometallic couplings) reduces the risk of batch failures due to minor operational deviations. This reliability translates to more predictable lead times and the ability to scale production volumes rapidly in response to clinical trial demands without requiring extensive process re-validation.
- Scalability and Environmental Compliance: The workup procedure relies primarily on aqueous quenching and pH adjustment, generating waste streams that are easier to treat compared to those containing heavy metals or halogenated solvents. The precipitation step allows for the isolation of large quantities of product without the need for energy-intensive distillation of high-boiling solvents. This environmental friendliness simplifies regulatory filings regarding environmental impact and aligns with the increasing corporate mandates for sustainable manufacturing practices in the pharmaceutical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel derivatives. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy for decision-makers evaluating this technology for their pipeline.
Q: What are the primary advantages of this synthesis route for commercial production?
A: The process utilizes readily available raw materials like phenanthroline dione and cinnamaldehydes, operates under standard reflux conditions without expensive transition metal catalysts, and achieves high yields (up to 90%), significantly simplifying downstream processing and reducing overall manufacturing costs.
Q: How does the structural modification affect biological activity compared to parent compounds?
A: By modifying the phenanthroimidazole scaffold with various cinnamaldehyde groups, the resulting derivatives exhibit enhanced selectivity and potency against tumor cells (such as lung and liver cancer lines) while maintaining low toxicity towards normal human cells, addressing a critical challenge in oncology drug design.
Q: Is the purification process scalable for industrial applications?
A: Yes, the workup involves a straightforward pH adjustment to induce precipitation, followed by filtration and washing. While lab-scale examples use silica gel, the initial precipitation step allows for bulk isolation, making the route highly amenable to scale-up with appropriate crystallization optimization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenanthroimidazole Derivatives Supplier
As the demand for targeted oncology therapies grows, the need for high-quality, scalable intermediates becomes paramount. NINGBO INNO PHARMCHEM stands ready to support your drug development programs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing condensation reactions like the one described in CN114671870B, ensuring that stringent purity specifications are met through our rigorous QC labs and advanced analytical capabilities. We understand the critical nature of timeline adherence in oncology research and are committed to providing a seamless supply of these complex heterocyclic building blocks.
We invite you to engage with our technical procurement team to discuss your specific requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how adopting this synthetic route can optimize your budget. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your project's unique needs, ensuring a successful transition from bench-scale discovery to commercial manufacturing.
