Advanced Green Synthesis of Flumazenil Intermediates for Commercial Scale-up
Introduction to Patent CN112979658B
The pharmaceutical industry continuously seeks robust and environmentally sustainable pathways for the production of critical active pharmaceutical ingredients (APIs) and their precursors. Patent CN112979658B discloses a significant advancement in the synthesis of Flumazenil, a specific benzodiazepine receptor antagonist used clinically for reversing sedation. This intellectual property outlines a novel four-step synthetic route that begins with the commercially accessible 5-fluoro-2-nitrobenzoic acid. Unlike legacy methods that rely on hazardous cyclization agents, this approach leverages a tandem reduction-cyclization strategy mediated by heterogeneous metal catalysts. The technical breakthrough lies in the ability to construct the core 1,4-benzodiazepine-2,5-dione scaffold directly from a nitro-amide precursor, thereby streamlining the process flow. For R&D directors and procurement specialists, this patent represents a viable alternative that addresses both regulatory pressure for greener chemistry and the economic imperative for higher throughput. The method not only simplifies the operational complexity but also enhances the overall safety profile of the manufacturing facility by removing the necessity for handling extremely toxic reagents such as phosgene derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Flumazenil and its key intermediates has been plagued by significant safety and environmental challenges inherent to traditional synthetic designs. Conventional routes, often designated as paths A through D in prior literature, typically necessitate the formation of a 6-fluoroisatinic anhydride intermediate. This transformation frequently employs phosgene or various chloroformate esters, which are classified as highly toxic and corrosive substances requiring stringent containment measures. Furthermore, alternative oxidative pathways utilizing chromium trioxide or peroxyacids introduce heavy metal contamination risks and generate substantial quantities of hazardous waste streams. These legacy processes not only increase the cost of compliance with environmental regulations but also complicate the purification of the final API due to the formation of difficult-to-remove oxidation byproducts. The reliance on such aggressive reagents creates bottlenecks in supply chain continuity, as the sourcing and handling of these controlled substances add layers of logistical friction and liability to the production schedule.
The Novel Approach
In stark contrast, the methodology described in CN112979658B circumvents these historical pitfalls by adopting a convergent strategy centered on nucleophilic substitution and catalytic hydrogenation. The process initiates with the condensation of 5-fluoro-2-nitrobenzoic acid with sarcosine esters, forming a linear nitro-amide chain without the need for anhydride formation. The pivotal innovation occurs in the subsequent step, where the nitro group is reduced in the presence of a catalyst like Raney Nickel, triggering an immediate intramolecular cyclization to form the seven-membered diazepine ring. This "one-pot" style transformation effectively merges two distinct chemical operations into a single unit operation, drastically reducing solvent consumption and processing time. By eliminating the requirement for strong oxidants and virulent chlorinating agents in the early stages, the novel approach offers a cleaner reaction profile. This shift not only mitigates occupational health risks for plant personnel but also results in a crude product with a superior impurity spectrum, facilitating easier downstream purification and ultimately delivering a higher quality pharmaceutical intermediate.
Mechanistic Insights into Raney Nickel-Catalyzed Reductive Cyclization
The cornerstone of this synthetic advancement is the catalytic reductive cyclization step, which transforms the N-(5-fluoro-2-nitrobenzoyl)-N-methylglycine ester into the 7-fluoro-3,4-dihydro-4-methyl-1H-[1,4]benzodiazepine-2,5-dione core. Mechanistically, this reaction proceeds via the surface-mediated hydrogenation of the aromatic nitro group to an aniline intermediate. Under the specific reaction conditions employing solvents like methanol-water mixtures, the newly formed amino group acts as a potent nucleophile. It immediately attacks the adjacent ester carbonyl carbon within the same molecule, displacing the alkoxide leaving group to close the ring. The use of heterogeneous catalysts such as Raney Nickel or Palladium on Carbon is critical here, as they provide the necessary active sites for hydrogen activation while remaining easily separable from the reaction mixture via simple filtration. This heterogeneity is a major advantage for GMP manufacturing, as it prevents metal leaching into the product stream compared to homogeneous counterparts. The patent data indicates that optimizing the water content in the alcohol solvent is crucial; a methanol-water ratio ensures sufficient solubility of the polar intermediates while maintaining the catalytic activity required for high conversion rates.
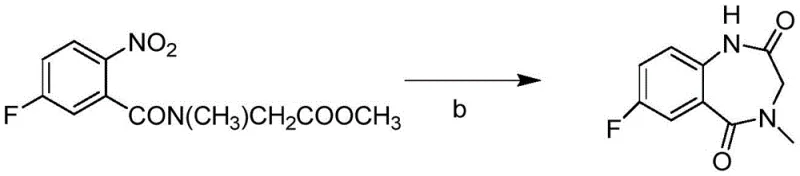
Furthermore, the mechanistic pathway inherently suppresses the formation of side products commonly associated with oxidative cyclization methods. In traditional routes involving chromium or peracid oxidation, over-oxidation of the benzylic positions or the amine nitrogen can lead to complex impurity profiles that are challenging to purge. The reductive nature of this new protocol ensures that the oxidation state of the carbon framework remains controlled throughout the ring-closing event. The patent specifies that yields for this specific transformation can approach quantitative levels, suggesting that the equilibrium strongly favors the cyclic lactam product under the prescribed thermal and pressure conditions. For process chemists, understanding this mechanism allows for precise tuning of hydrogen pressure and temperature to maximize throughput. The ability to drive this reaction to near-completion minimizes the burden on subsequent purification columns, directly translating to cost savings in material usage and waste disposal. This level of control over the reaction trajectory is essential for maintaining batch-to-batch consistency in a commercial setting.
How to Synthesize 7-Fluoro-3,4-dihydro-4-methyl-1H-[1,4]benzodiazepine-2,5-dione Efficiently
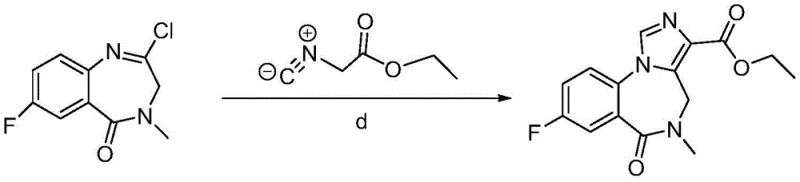
Implementing this synthesis requires careful attention to the activation of the starting carboxylic acid and the management of the catalytic hydrogenation environment. The initial condensation can be achieved using standard peptide coupling reagents like EDCI and HOBt, or alternatively via acid chloride formation using thionyl chloride, offering flexibility based on available infrastructure. Once the nitro-amide precursor is secured, the focus shifts to the high-pressure hydrogenation vessel where the ring closure occurs. The detailed standardized synthesis steps see the guide below, which outlines the specific molar ratios and solvent systems proven to deliver optimal results. Following the cyclization, the resulting dione is subjected to chlorination using reagents such as phosphorus pentachloride to activate the 2-position for the final imidazole ring fusion. This sequence ensures a logical flow of chemistry that balances reactivity with safety.
- Condense 5-fluoro-2-nitrobenzoic acid with sarcosine ester using EDCI/HOBt or thionyl chloride activation to form the nitro-amide intermediate.
- Perform catalytic hydrogenation using Raney Nickel or Pd/C in methanol-water to reduce the nitro group and simultaneously cyclize to the benzodiazepine dione.
- Chlorinate the dione intermediate using phosphorus pentachloride or thionyl chloride to generate the imine chloride species.
- React the chloro-intermediate with ethyl isocyanoacetate under basic conditions to complete the cycloaddition and form Flumazenil.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this patented route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By utilizing 5-fluoro-2-nitrobenzoic acid, a commodity chemical, manufacturers can avoid the volatility and premium pricing associated with specialized anhydrides or isatins required by older methods. The elimination of phosgene and chloroformates removes the need for specialized storage facilities and expensive scrubbing systems, leading to significant capital expenditure reductions for new production lines. Moreover, the high efficiency of the reductive cyclization step means that less raw material is wasted as byproduct, improving the overall mass balance of the process. This efficiency gain is critical for maintaining healthy margins in the competitive generic pharmaceutical market. The robustness of the catalyst system also implies longer campaign runs with less frequent reactor cleaning, enhancing asset utilization rates.
- Cost Reduction in Manufacturing: The economic model of this synthesis is strengthened by the removal of expensive and hazardous reagents. Traditional methods often incur high costs related to the safe disposal of toxic waste generated by chromium oxidants or phosgene residues. By switching to a hydrogenation-based protocol, the waste stream becomes significantly more benign, primarily consisting of aqueous alcohol and spent catalyst which can often be recycled or disposed of at a lower cost. Additionally, the high yields reported in the patent, particularly in the cyclization step, mean that the effective cost per kilogram of the intermediate is lowered. There is no need for complex transition metal scavenging resins often required for homogeneous catalysts, further reducing the bill of materials. These factors combine to create a leaner cost structure that provides a buffer against raw material price fluctuations.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials. 5-fluoro-2-nitrobenzoic acid and sarcosine esters are produced by multiple global suppliers, reducing the risk of single-source bottlenecks. In contrast, custom-synthesized anhydrides or specific oxidized intermediates often have limited supplier bases, making the supply chain vulnerable to disruptions. The simplified process flow also reduces the lead time for production cycles. With fewer distinct isolation steps and simpler workups, the turnaround time from raw material intake to finished intermediate is shortened. This agility allows manufacturers to respond more quickly to changes in market demand, ensuring consistent availability for downstream API producers. The stability of the intermediates also allows for potential stockpiling strategies without significant degradation concerns.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden inefficiencies, but this route is designed with scalability in mind. The heterogeneous catalysis step is inherently scalable, as hydrogenation reactors are standard equipment in most fine chemical plants. The absence of highly exothermic oxidation steps reduces the thermal load on the reactor cooling systems, making scale-up safer and more predictable. From an environmental standpoint, the process aligns with green chemistry principles by reducing the E-factor (mass of waste per mass of product). The avoidance of heavy metals like chromium eliminates the risk of soil and water contamination, ensuring compliance with increasingly strict environmental regulations globally. This proactive approach to environmental stewardship minimizes the risk of regulatory shutdowns and enhances the corporate sustainability profile, which is increasingly important for partnerships with major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific operational parameters and beneficial effects detailed in the patent documentation. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer. The answers provided reflect the empirical data and logical deductions drawn from the comparative analysis of this method against established industry standards.
Q: How does this new route improve safety compared to traditional Flumazenil synthesis?
A: Traditional routes often rely on highly toxic reagents like phosgene or ethyl chloroformate for ring closure, posing significant safety and environmental risks. This patented method eliminates the need for these hazardous chemicals by using a direct reductive cyclization strategy, significantly lowering the toxicity profile of the manufacturing process.
Q: What are the yield advantages of the Raney Nickel catalyzed step?
A: The reductive cyclization step using Raney Nickel in a methanol-water solvent system demonstrates exceptionally high efficiency, with patent data indicating yields approaching 100% under optimized conditions. This contrasts sharply with older oxidation-based methods which often suffer from lower conversion and higher impurity loads.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. It utilizes readily available starting materials like 5-fluoro-2-nitrobenzoic acid and avoids complex, sensitive reagents. The simplified workup procedures, such as simple filtration to remove the heterogeneous catalyst, facilitate easier scale-up from pilot to commercial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Flumazenil Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our technical team has extensively analyzed the pathway described in CN112979658B and possesses the expertise to implement this green synthesis at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. Our facilities are equipped with state-of-the-art high-pressure hydrogenation units and rigorous QC labs capable of meeting stringent purity specifications required for CNS drug intermediates. We are committed to delivering high-purity Flumazenil intermediates that adhere to the highest quality standards, leveraging our deep understanding of catalytic processes to optimize yield and minimize impurities.
We invite potential partners to engage with our technical procurement team to discuss how this innovative route can be integrated into your supply chain. By collaborating with us, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to request specific COA data and route feasibility assessments to verify the compatibility of this method with your existing quality frameworks. Let us help you secure a sustainable and cost-effective supply of this critical pharmaceutical building block.
