Advanced Synthesis of Octadecyl Isocyanate: Enhancing Safety and Commercial Scalability
The chemical industry is currently undergoing a significant paradigm shift towards safer, more sustainable manufacturing processes, particularly in the synthesis of high-value isocyanates. Patent CN101245036A introduces a groundbreaking methodology for the production of octadecyl isocyanate, a critical intermediate used in various pharmaceutical and agrochemical applications. This innovation replaces the historically hazardous use of gaseous phosgene with bis(trichloromethyl)carbonate, commonly known as triphosgene, thereby fundamentally altering the safety profile and operational feasibility of the synthesis. By leveraging this solid phosgene equivalent, manufacturers can achieve high reaction yields while mitigating the severe regulatory and safety burdens associated with handling toxic gases. This report analyzes the technical merits of this approach, demonstrating how it serves as a reliable octadecyl isocyanate supplier strategy for modern chemical enterprises seeking to optimize their production capabilities without compromising on safety or quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of long-chain alkyl isocyanates, such as octadecyl isocyanate, relied heavily on the direct reaction of primary amines with gaseous phosgene. As referenced in prior art like GB761590, this traditional route necessitates the use of highly toxic carbon monoxide dichloride gas, which presents monumental challenges for both laboratory-scale research and large-scale commercial manufacturing. The requirement for specialized, hermetically sealed reaction vessels capable of withstanding corrosive gases drives up capital expenditure significantly, creating a high barrier to entry for many producers. Furthermore, the logistical complexity of storing and transporting phosgene gas imposes strict regulatory compliance costs and continuous safety monitoring requirements. From an environmental perspective, the potential for tail gas leakage poses severe ecological risks, requiring extensive scrubbing systems to neutralize unreacted phosgene before release. Additionally, the gaseous nature of the reagent makes precise stoichiometric control difficult, often leading to localized excesses that promote side reactions and reduce overall product purity, thereby complicating downstream purification processes.
The Novel Approach
The novel approach detailed in the patent data utilizes bis(trichloromethyl)carbonate as a safe, solid alternative that effectively mimics the reactivity of phosgene without its inherent dangers. This method allows for the dissolution of the reagent in common organic solvents, facilitating precise metering and homogeneous mixing with the octadecylamine substrate. By operating with a solid reagent, the process eliminates the need for complex gas delivery infrastructure, drastically simplifying the reactor setup and reducing the initial investment required for plant construction. The reaction conditions are remarkably mild yet effective, initiating at low temperatures to control exothermicity and progressing to moderate heating to drive the conversion to completion. This transition from gas-phase to solution-phase chemistry not only enhances operator safety but also improves the reproducibility of the reaction, ensuring consistent batch-to-batch quality. Consequently, this methodology represents a substantial advancement in cost reduction in fine chemical intermediates manufacturing, offering a streamlined pathway that aligns with modern green chemistry principles and rigorous safety standards.
Mechanistic Insights into Triphosgene-Mediated Phosgenation
The core of this synthesis lies in the thermal decomposition of bis(trichloromethyl)carbonate, which acts as a reservoir for phosgene generation within the reaction medium. Upon heating or under catalytic conditions, one mole of triphosgene decomposes to release three moles of phosgene in situ, which immediately reacts with the nucleophilic octadecylamine. This controlled release mechanism prevents the accumulation of high concentrations of free phosgene, thereby minimizing the risk of runaway reactions and enhancing process safety. The reaction proceeds through the formation of a carbamoyl chloride intermediate, which subsequently eliminates hydrogen chloride to form the desired isocyanate functionality. The stoichiometry is precisely balanced, with three equivalents of amine reacting with one equivalent of triphosgene to yield three equivalents of the isocyanate product and six equivalents of hydrogen chloride gas. Understanding this mechanistic pathway is crucial for optimizing reaction parameters, such as temperature ramps and addition rates, to maximize yield while suppressing the formation of urea byproducts that can occur if amine concentration is too high relative to the generated phosgene.
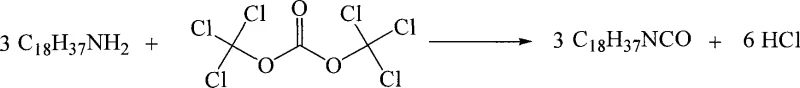
Impurity control is another critical aspect of this mechanism, primarily managed through the efficient removal of the hydrogen chloride byproduct. As the reaction progresses, the evolution of HCl gas can lead to the formation of amine hydrochloride salts if not adequately vented, which would consume the starting material and lower the overall yield. The patent specifies a post-treatment step involving nitrogen purging to sweep out the acidic gas, ensuring that the equilibrium is driven towards the product side. Furthermore, the choice of solvent plays a pivotal role in managing impurities; solvents like toluene or dichloroethane provide an optimal balance of solubility for both reactants and products while allowing for easy separation during the workup phase. The high purity levels reported, often exceeding 98%, indicate that side reactions such as polymerization or over-chlorination are effectively suppressed under these controlled conditions. This level of purity is essential for downstream applications in pharmaceutical synthesis, where trace impurities can have significant impacts on the efficacy and safety of the final active ingredient.
How to Synthesize Octadecyl Isocyanate Efficiently
The synthesis of octadecyl isocyanate via this triphosgene-mediated route offers a robust protocol that balances safety with high efficiency. The process begins with the careful preparation of reactant solutions, ensuring that the solid triphosgene is fully dissolved before the introduction of the amine to prevent localized hot spots. The dropwise addition of the amine solution at low temperatures is a critical control point that manages the exothermic nature of the initial reaction phase. Following the addition, a staged temperature profile is employed, starting with a room temperature hold to allow for intermediate formation, followed by heating to drive the elimination of HCl and complete the cyclization. This systematic approach ensures that the reaction proceeds smoothly without thermal runaway, making it suitable for scale-up. For detailed operational parameters and specific molar ratios, please refer to the standardized synthesis steps outlined below.
- Dissolve bis(trichloromethyl)carbonate in an organic solvent such as toluene or dichloroethane and cool the mixture to 0-5°C.
- Slowly add a solution of octadecylamine in the same solvent dropwise while maintaining the low temperature to control exothermicity.
- Stir at room temperature followed by heating to 75-120°C to complete the reaction, then remove HCl gas and distill the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this triphosgene-based synthesis offers compelling advantages that extend beyond mere technical feasibility. The shift away from gaseous phosgene fundamentally alters the cost structure of production by removing the need for specialized gas containment systems and the associated maintenance and inspection regimes. This reduction in capital intensity allows for more flexible manufacturing setups and lowers the barrier for scaling production capacity to meet market demand. Moreover, the use of solid triphosgene simplifies logistics and storage, as it does not require the high-pressure cylinders or dedicated storage bunkers needed for toxic gases. This ease of handling translates into reduced insurance premiums and lower regulatory compliance costs, contributing to a more favorable overall cost of goods sold. The ability to source raw materials that are commercially available and stable further enhances supply chain resilience, reducing the risk of production stoppages due to reagent shortages or transportation restrictions on hazardous materials.
- Cost Reduction in Manufacturing: The elimination of gaseous phosgene infrastructure results in significant savings on equipment investment and operational overhead. By utilizing standard glass-lined or stainless steel reactors without the need for exotic alloys or complex sealing mechanisms, manufacturers can achieve substantial cost savings. The simplified workup procedure, which involves solvent recovery and distillation, minimizes waste treatment costs and maximizes resource efficiency. Additionally, the high atom economy of the reaction ensures that raw material costs are optimized, as the majority of the reagent mass is converted into the desired product or recoverable byproducts. These factors collectively contribute to a more competitive pricing structure for the final octadecyl isocyanate product, enabling better margin management in volatile market conditions.
- Enhanced Supply Chain Reliability: Sourcing solid triphosgene is generally more straightforward and less regulated than sourcing gaseous phosgene, leading to improved supply continuity. The stability of the solid reagent allows for longer shelf life and bulk purchasing strategies, which can buffer against market fluctuations and supply disruptions. Furthermore, the reduced safety risks associated with the process minimize the likelihood of unplanned shutdowns due to safety incidents or regulatory inspections. This reliability is crucial for maintaining consistent delivery schedules to downstream customers, particularly in the pharmaceutical sector where supply interruptions can have cascading effects on drug development timelines. The robustness of the supply chain is further strengthened by the availability of multiple solvent options, providing flexibility to adapt to regional solvent availability or price variations.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory benchtop to commercial production without significant re-engineering. The manageable exotherm and the use of common solvents facilitate safe scale-up, allowing manufacturers to increase batch sizes to meet growing demand efficiently. From an environmental standpoint, the process generates minimal liquid waste, with the primary byproduct being hydrogen chloride gas which can be captured and utilized in other processes or neutralized safely. This alignment with green chemistry principles enhances the company's environmental profile and ensures compliance with increasingly stringent global environmental regulations. The ability to demonstrate a clean, safe production process is a valuable asset in today's market, appealing to eco-conscious clients and stakeholders who prioritize sustainability in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of octadecyl isocyanate using the triphosgene method. These answers are derived directly from the experimental data and process descriptions found in the relevant patent literature, providing accurate and actionable insights for technical decision-makers. Understanding these details is essential for evaluating the feasibility of adopting this technology within your existing manufacturing framework. The responses cover aspects ranging from reaction optimization to safety protocols, ensuring a comprehensive understanding of the process capabilities.
Q: Why is triphosgene preferred over gaseous phosgene for octadecyl isocyanate production?
A: Triphosgene is a stable crystalline solid that decomposes in situ to generate phosgene, eliminating the need for complex gas handling infrastructure and significantly reducing safety risks associated with toxic gas leaks.
Q: What are the typical purity levels achievable with this synthesis method?
A: According to patent data, this method consistently achieves product purity exceeding 97%, with optimized examples reaching up to 98.4% as determined by gas chromatography.
Q: How does this process impact waste management compared to traditional methods?
A: The process generates hydrogen chloride gas as a byproduct which can be easily captured and recycled, resulting in minimal liquid waste discharge and improved environmental compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Octadecyl Isocyanate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global chemical market. Our expertise in scaling diverse pathways from 100 kgs to 100 MT/annual commercial production positions us as a strategic partner for companies seeking high-purity octadecyl isocyanate. We leverage state-of-the-art facilities and rigorous QC labs to ensure that every batch meets stringent purity specifications, consistently delivering product quality that exceeds industry standards. Our commitment to safety and environmental responsibility is reflected in our adoption of cutting-edge technologies like the triphosgene-mediated synthesis, which minimizes risk while maximizing efficiency. By partnering with us, you gain access to a supply chain that is not only reliable but also aligned with the highest standards of chemical manufacturing excellence.
We invite you to engage with our technical procurement team to discuss how our capabilities can support your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data to validate our product quality, we are ready to provide the necessary documentation and support. Our team can also offer route feasibility assessments to help you integrate this advanced synthesis method into your broader production strategy. Contact us today to explore how NINGBO INNO PHARMCHEM can become your trusted partner in delivering high-quality chemical intermediates for your business growth.
