Advanced Green Synthesis of 5-Nitro-2-Chlorobenzenesulfonyl Chloride for Commercial Scale-Up
The chemical industry is constantly evolving towards greener, safer, and more efficient manufacturing processes, a trend vividly exemplified by the technological advancements detailed in patent CN102351751A. This pivotal intellectual property introduces a novel method for the chemical synthesis of 5-nitro-2-chlorobenzenesulfonyl chloride, a critical intermediate widely utilized in the production of acid dyes, fluorescent pigments, and various pharmaceutical and agrochemical compounds. Traditionally, the synthesis of such sulfonyl chlorides has been plagued by significant safety hazards and environmental concerns, primarily due to the reliance on aggressive chlorinating agents. However, this new methodology leverages bis(trichloromethyl)carbonate (BTC), often known as triphosgene, as a superior alternative to traditional reagents. By shifting the paradigm from hazardous gaseous or volatile liquid reagents to a stable solid-state chlorinating agent, the process not only enhances operational safety but also drastically improves the quality of the final product. For R&D directors and procurement specialists alike, understanding this shift is crucial, as it represents a tangible move towards sustainable chemistry that does not compromise on yield or purity, ultimately securing a more reliable supply chain for high-value fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 5-nitro-2-chlorobenzenesulfonyl chloride has relied heavily on a two-step process starting from p-nitrochlorobenzene. As illustrated in the reaction scheme below, the conventional route involves sulfonation followed by chlorination using thionyl chloride (SOCl2). While chemically effective, this traditional approach presents severe engineering and safety challenges that impact both cost and operational continuity. The use of thionyl chloride generates substantial quantities of sulfur dioxide and hydrogen chloride gases as byproducts, which are highly corrosive and toxic. These gases necessitate complex and expensive scrubbing systems to prevent environmental release and equipment degradation. Furthermore, the exothermic nature of the reaction and the volatility of the reagents create significant safety risks for plant operators, requiring stringent containment protocols that increase capital expenditure. The corrosion caused by the acidic off-gases often leads to frequent maintenance downtime and shortened equipment lifespans, creating hidden costs that erode profit margins in large-scale manufacturing scenarios.
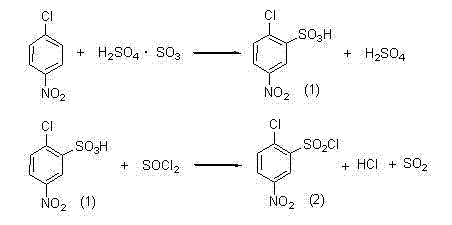
The Novel Approach
In stark contrast to the legacy methods, the innovative technique described in the patent utilizes bis(trichloromethyl)carbonate (BTC) in the presence of an organic base to effect the chlorination of 5-nitro-2-chlorobenzenesulfonic acid. This approach fundamentally alters the reaction profile by replacing the evolution of toxic gases with a cleaner transformation that occurs under much milder conditions. The reaction typically proceeds in organic solvents such as tetrahydrofuran, dichloromethane, or toluene at temperatures ranging from 25°C to 80°C, eliminating the need for extreme thermal inputs. The use of BTC, a solid reagent, simplifies logistics and storage compared to handling pressurized cylinders of hazardous gases or volatile liquids. Moreover, the byproduct profile is significantly cleaner, minimizing the burden on waste treatment facilities and reducing the overall environmental footprint of the manufacturing process. This transition not only aligns with modern green chemistry principles but also offers a robust pathway for producing high-purity intermediates essential for sensitive downstream applications in the pharmaceutical and agrochemical sectors.
Mechanistic Insights into BTC-Mediated Chlorination
The core of this technological breakthrough lies in the unique reactivity of bis(trichloromethyl)carbonate, which serves as a safe and equivalent source of phosgene without the associated handling dangers. Mechanistically, the reaction begins with the nucleophilic attack of the sulfonic acid oxygen on the carbonyl carbon of the BTC molecule, facilitated by the presence of an organic base such as triethylamine or pyridine. The base plays a dual role: it acts as a catalyst to activate the sulfonic acid and simultaneously scavenges the hydrochloric acid generated during the formation of the chlorosulfite intermediate. This intermediate subsequently decomposes to release the desired sulfonyl chloride functionality while generating carbon dioxide and chloride ions as benign byproducts. The controlled release of the chlorinating species ensures that the reaction proceeds smoothly without the violent exotherms often observed with thionyl chloride. This precise control over the reaction kinetics is vital for maintaining the integrity of the nitro and chloro substituents on the aromatic ring, preventing unwanted side reactions or decomposition that could lead to difficult-to-remove impurities.
From an impurity control perspective, this mechanism offers distinct advantages for ensuring high product quality. The absence of sulfur dioxide gas eliminates the risk of sulfonation side reactions or the formation of sulfite esters that can contaminate the final product. Additionally, the use of specific organic bases allows for fine-tuning the pH of the reaction medium, which is critical for suppressing the hydrolysis of the sensitive sulfonyl chloride group. The patent data indicates that through careful optimization of the molar ratios between the sulfonic acid, BTC, and the organic base, yields exceeding 90% can be consistently achieved. Post-reaction processing is also streamlined; the solid byproducts and excess reagents can often be removed via simple filtration or aqueous workup, followed by recrystallization from solvents like ethanol. This results in a final product with purity levels reaching up to 99.1%, a specification that is paramount for customers in the fine chemical and pharmaceutical industries who require rigorous control over their raw material quality.
How to Synthesize 5-Nitro-2-Chlorobenzenesulfonyl Chloride Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and temperature control to maximize efficiency and safety. The process generally involves dissolving the sulfonic acid starting material in a suitable aprotic solvent, followed by the addition of the organic base to form a homogeneous mixture. The BTC is then introduced, typically as a solution, to ensure even distribution and controlled reaction rates. Maintaining the temperature within the optimal window of 25°C to 70°C is crucial; too low a temperature may result in incomplete conversion, while excessive heat could promote decomposition. Following the reaction period, usually lasting between 2 to 8 hours, the mixture is processed to isolate the crude product, which is then purified to meet stringent quality standards. For a detailed breakdown of the specific operational parameters and safety protocols required for this synthesis, please refer to the standardized guide below.
- Dissolve 5-nitro-2-chlorobenzenesulfonic acid in an organic solvent such as tetrahydrofuran or dichloromethane under mechanical stirring.
- Add an organic base like triethylamine or pyridine to the mixture to act as an acid scavenger and catalyst.
- Slowly add bis(trichloromethyl)carbonate (BTC) solution while maintaining temperature between 25-80°C, then filter and purify the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this BTC-based synthesis route translates into significant strategic advantages beyond mere chemical efficiency. The primary benefit lies in the drastic reduction of operational risks and associated costs. By eliminating the need for specialized corrosion-resistant equipment designed to handle aggressive acidic gases, manufacturers can utilize standard glass-lined or stainless steel reactors, thereby lowering capital investment requirements. The stability of BTC as a solid reagent also simplifies inventory management and transportation logistics, removing the regulatory burdens and safety hazards associated with shipping volatile chlorinating agents. This reliability ensures a more consistent supply of the intermediate, reducing the risk of production stoppages due to reagent shortages or safety incidents. Furthermore, the simplified waste stream reduces the cost and complexity of environmental compliance, allowing for a more streamlined and cost-effective manufacturing operation that is resilient to regulatory changes.
- Cost Reduction in Manufacturing: The transition to this green chemistry protocol offers substantial economic benefits by removing the need for expensive gas scrubbing infrastructure and heavy-duty corrosion protection. Since the process does not generate large volumes of toxic SO2 and HCl gases, the engineering controls required are far less intensive, leading to lower maintenance costs and extended equipment life. Additionally, the high selectivity of the reaction minimizes the loss of valuable starting materials to side products, improving the overall atom economy. The ability to achieve high yields with simpler purification steps further reduces the consumption of solvents and energy, contributing to a leaner and more profitable production model that enhances competitiveness in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: Supply chain stability is significantly bolstered by the use of bis(trichloromethyl)carbonate, which is easier to source, store, and handle than thionyl chloride. The solid nature of BTC eliminates the risks of leakage and evaporation during storage, ensuring that reagent quality remains consistent over time. This stability allows for larger batch sizes and longer production runs without the interruption of frequent reagent replenishment or safety checks required for volatile liquids. Consequently, manufacturers can offer more reliable lead times to their customers, fostering stronger long-term partnerships. The reduced dependency on complex waste treatment facilities also means that production is less susceptible to disruptions caused by environmental inspections or waste disposal bottlenecks, ensuring a continuous flow of high-quality intermediates to downstream users.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is inherently safer and more straightforward due to the mild reaction conditions and manageable exotherm. The absence of hazardous gas evolution simplifies the design of large-scale reactors, making it easier to expand capacity to meet growing market demand without prohibitive engineering challenges. From an environmental standpoint, the process aligns perfectly with increasingly strict global regulations regarding emissions and waste disposal. By generating minimal hazardous waste and avoiding the release of ozone-depleting or toxic substances, the manufacturer demonstrates a strong commitment to sustainability. This compliance not only mitigates legal risks but also enhances the brand reputation of the supplier, appealing to multinational corporations that prioritize environmentally responsible sourcing in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of 5-nitro-2-chlorobenzenesulfonyl chloride. These insights are derived directly from the technical specifications and experimental data provided in the relevant patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of integrating this intermediate into your specific manufacturing workflows. We encourage technical teams to review these points to gain a comprehensive understanding of the process capabilities and limitations.
Q: Why is Bis(trichloromethyl)carbonate (BTC) preferred over Thionyl Chloride?
A: BTC is a solid crystalline substance that is significantly safer to transport and store compared to volatile liquid thionyl chloride. It eliminates the generation of large volumes of corrosive SO2 and HCl gases during the reaction, reducing equipment corrosion and environmental hazards.
Q: What purity levels can be achieved with this synthetic route?
A: According to patent data, recrystallization of the crude product can achieve purity levels exceeding 99%, with specific examples demonstrating 99.1% purity, making it suitable for high-grade pharmaceutical and agrochemical applications.
Q: Is this process scalable for industrial production?
A: Yes, the process operates at mild temperatures (25-80°C) and uses common organic solvents like THF or toluene. The elimination of aggressive gas byproducts simplifies the engineering requirements for large-scale reactors, facilitating easier commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Nitro-2-Chlorobenzenesulfonyl Chloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global chemical market. Our expertise extends beyond simple manufacturing; we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 5-nitro-2-chlorobenzenesulfonyl chloride meets the highest international standards. We are committed to leveraging green chemistry technologies, such as the BTC-mediated synthesis described herein, to deliver products that are not only high in quality but also produced with a minimal environmental footprint, aligning with the sustainability goals of our partners.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. Whether you are developing new pharmaceutical APIs or expanding your agrochemical portfolio, we encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us be your strategic partner in navigating the complexities of fine chemical sourcing, ensuring reliability, quality, and innovation in every delivery.
