Advanced Synthesis of Crystalline 3-O-Alkyl-Ascorbic Acid for Global Pharmaceutical Supply Chains
The global demand for stable, bioactive vitamin C derivatives continues to surge across the pharmaceutical and cosmetic sectors, driven by the need for ingredients that resist oxidation while maintaining potent antioxidant properties. Traditional L-ascorbic acid, while effective, suffers from significant instability in aqueous phases and poor oil solubility, limiting its formulation potential. Addressing these critical formulation challenges, Patent CN101528722A introduces a groundbreaking methodology for the preparation of crystalline 3-O-alkyl-ascorbic acid derivatives. This innovation leverages a unique catalytic system involving anion exchange resins adsorbed with polyiodide anions, enabling high-yield alkylation under mild conditions. For R&D directors and supply chain managers seeking reliable pharmaceutical intermediates supplier partnerships, this technology represents a paradigm shift from cumbersome, low-yield legacy processes to a streamlined, commercially viable production route that ensures consistent quality and supply continuity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-substituted ascorbic acid derivatives has been plagued by significant chemical and operational inefficiencies that hinder large-scale adoption. Conventional approaches, such as those described in earlier literature, often rely on the reaction of ascorbic acid with sodium methoxide in high-boiling polar aprotic solvents like dimethylsulfoxide (DMSO) or dimethylformamide (DMF). These solvents present severe downstream processing challenges; their high boiling points make removal via vacuum distillation energy-intensive and time-consuming. Furthermore, the resulting 3-substituted derivatives exhibit high solubility in these polar media, necessitating complex purification techniques such as column chromatography, which are impractical for ton-scale manufacturing. Additionally, these harsh alkaline conditions frequently lead to uncontrolled side reactions and the generation of impurities, resulting in suboptimal yields and compromising the purity profile required for high-grade cosmetic active ingredients.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes a sophisticated catalytic strategy that circumvents these traditional bottlenecks. By employing 5,6-O-isopropylidene ascorbic acid as a protected precursor and reacting it with alkyl halides in the presence of a polyiodide-adsorbed anion exchange resin, the process achieves remarkable selectivity and efficiency. The reaction proceeds rapidly at room temperature, typically completing within one hour, which significantly reduces thermal stress on the sensitive ascorbic acid backbone. This mild condition not only preserves the structural integrity of the molecule but also minimizes the formation of degradation by-products. The subsequent deprotection step can be elegantly performed using recoverable solid acid catalysts like Nafion H, further enhancing the green chemistry profile of the synthesis. This holistic approach transforms the production landscape, offering a robust pathway for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Polyiodide Anion Exchange Resin Catalysis
The core innovation of this synthesis lies in the unique role of the polyiodide anions ($I_3^-$, $I_5^-$, $I_7^-$) adsorbed onto the polymer matrix of the anion exchange resin. Mechanistically, these polyiodide species act as potent nucleophilic promoters that facilitate the substitution reaction at the 3-hydroxyl position of the ascorbic acid ring. Unlike traditional strong bases that can induce elimination reactions or ring opening, the polyiodide system provides a controlled environment that favors $S_N2$-type alkylation. The resin support serves a dual function: it concentrates the catalytic species locally and allows for the modulation of nucleophilicity through the specific ratio of iodide salt to elemental iodine (typically 1:1 to 1:5). This precise control over the catalytic microenvironment ensures that the alkylation occurs selectively at the C3 position, avoiding unwanted substitution at the C2 or C5 positions, which is critical for maintaining the biological activity of the final derivative.
Furthermore, the mechanism extends to the deprotection phase, where the choice of catalyst profoundly impacts the impurity profile. When using Nafion H, a perfluorosulfonic acid resin, the hydrolysis of the isopropylidene protecting group occurs via a heterogeneous acid catalysis mechanism. This prevents the accumulation of free mineral acids in the reaction mixture, which could otherwise catalyze the degradation of the newly formed ether linkage or the lactone ring. The ability to simply filter off the Nafion H resin post-reaction means that the crude product is significantly cleaner than those obtained via liquid acid hydrolysis. This mechanistic advantage directly translates to simplified crystallization protocols, allowing manufacturers to achieve high-purity OLED material grade standards (or equivalent pharma grade) without resorting to expensive chromatographic purification steps.
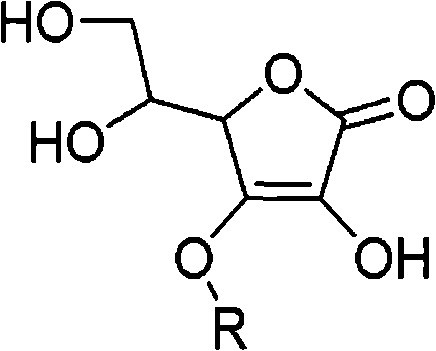
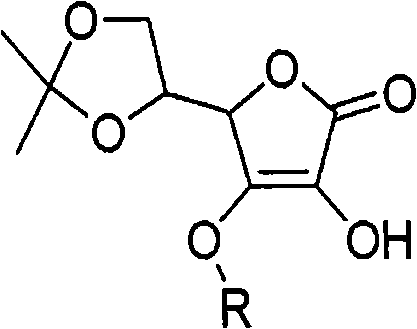
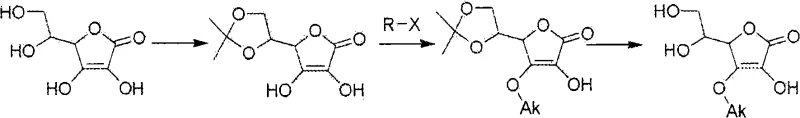
How to Synthesize 3-O-Alkyl-Ascorbic Acid Efficiently
Implementing this advanced synthesis route requires careful attention to the preparation of the catalytic resin and the control of reaction stoichiometry. The process begins with the generation of the polyiodide solution by mixing potassium iodide and iodine in water, followed by the immersion of a macroreticular anion exchange resin (such as Amberlite IRA-402) to adsorb the active species. Once the catalyst is prepared, the alkylation is conducted in an aprotic solvent like DMF or acetonitrile at ambient temperature. The simplicity of the workup—filtration of the resin followed by solvent removal—makes this protocol highly attractive for process chemists looking to optimize throughput. For a detailed breakdown of the specific reagent quantities, reaction times, and purification parameters validated in the patent examples, please refer to the standardized synthesis guide below.
- Preparation of the Catalyst: Mix basic iodide and iodine to form polyiodide anions, then adsorb onto an anion exchange resin.
- Alkylation Reaction: React 5,6-O-isopropylidene ascorbic acid with alkyl halides in the presence of the resin at room temperature.
- Deprotection and Purification: Remove the isopropylidene group using Nafion H or acid hydrolysis, followed by recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this polyiodide-resin mediated synthesis offers tangible strategic benefits that extend beyond mere chemical yield. The elimination of high-boiling solvents like DMSO and the avoidance of column chromatography represent a fundamental restructuring of the cost base for producing these valuable antioxidants. By shifting to a process that relies on filtration and recrystallization, manufacturers can drastically reduce solvent consumption and waste disposal costs, aligning with increasingly stringent environmental regulations. Moreover, the ability to run reactions at room temperature eliminates the need for extensive heating or cooling infrastructure, lowering the energy footprint of the manufacturing facility and reducing the risk of thermal runaway incidents.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the simplification of the purification train. Traditional methods often require multiple extraction steps and chromatographic columns to remove trace metal salts and solvent residues, which are cost-prohibitive at scale. In this novel process, the solid-supported catalyst is removed via simple filtration, and the excess alkyl halide is removed by vacuum distillation. This streamlined workflow significantly reduces labor hours and consumable costs. Additionally, the high selectivity of the reaction minimizes the loss of starting material to by-products, effectively improving the overall mass balance and reducing the cost of goods sold (COGS) for the final active ingredient.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on specialized reagents or complex processing equipment. This method utilizes commodity chemicals such as potassium iodide, iodine, and standard anion exchange resins, which are readily available from multiple global suppliers, mitigating the risk of raw material shortages. The robustness of the reaction conditions—specifically the tolerance for room temperature operation—means that production is less susceptible to utility failures or equipment malfunctions related to temperature control systems. This reliability ensures that delivery schedules for critical pharmaceutical intermediates can be met consistently, even during periods of high market demand.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new safety and environmental challenges, but this technology is inherently designed for expansion. The use of heterogeneous catalysts (both the polyiodide resin and Nafion H) facilitates easy separation and potential regeneration, reducing the volume of hazardous chemical waste generated per kilogram of product. The process avoids the use of heavy metal catalysts, simplifying the regulatory compliance burden regarding residual metal limits in the final drug substance. This 'green' profile not only appeals to environmentally conscious partners but also future-proofs the supply chain against tightening global regulations on industrial effluents and solvent emissions.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its integration into their supply chains. The following questions address common inquiries regarding the catalyst lifecycle, solvent selection, and product specifications. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for decision-making.
Q: Why is the polyiodide anion exchange resin superior to traditional sodium methoxide methods?
A: Traditional methods using sodium methoxide in polar solvents like DMSO often result in difficult purification due to high solvent boiling points and significant by-product formation. The polyiodide resin method operates at room temperature, minimizes side reactions, and allows for simple filtration removal of the catalyst, drastically simplifying downstream processing.
Q: What are the purity advantages of using Nafion H for deprotection?
A: Using Nafion H, a perfluorosulfonic acid resin, for deprotection offers significant advantages over liquid hydrochloric acid. It acts as a heterogeneous catalyst that can be easily recovered by filtration, preventing acid contamination in the final product and ensuring higher purity crystalline 3-O-alkyl-ascorbic acid suitable for sensitive pharmaceutical applications.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the method is highly scalable. The reaction completes within a short time at room temperature, reducing energy consumption. Furthermore, the use of solid-supported catalysts (resins) eliminates the need for complex aqueous workups typically associated with homogeneous catalysts, making it ideal for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-O-Alkyl-Ascorbic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of a patent must be translated into practical, industrial reality to create value. Our team of process engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results of laboratory-scale polyiodide catalysis are successfully replicated in our GMP-compliant facilities. We understand that consistency is key; therefore, our rigorous QC labs enforce stringent purity specifications for every batch of 3-O-alkyl-ascorbic acid produced, utilizing advanced analytical techniques to verify the absence of residual solvents and catalyst traces. Our commitment to technical excellence ensures that your formulations receive ingredients of the highest quality, supporting your own product efficacy and brand reputation.
We invite you to collaborate with us to optimize your sourcing strategy for stable vitamin C derivatives. Whether you require custom synthesis of specific alkyl chain lengths or bulk supply of standard ethyl or methyl derivatives, our technical procurement team is ready to assist. Contact us today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments to determine how our advanced manufacturing capabilities can enhance your supply chain resilience and product performance.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
