Scalable Electrochemical Synthesis of 4-Sulfonic Acid Isoquinolinone Derivatives for Commercial Pharma Applications
Scalable Electrochemical Synthesis of 4-Sulfonic Acid Isoquinolinone Derivatives for Commercial Pharma Applications
The pharmaceutical and fine chemical industries are constantly seeking sustainable and efficient methodologies for constructing complex heterocyclic scaffolds, particularly isoquinolinone derivatives which serve as critical intermediates in drug discovery. A groundbreaking approach detailed in patent CN110616439B introduces a novel electrochemical oxidation strategy for synthesizing 4-sulfonic acid substituted isoquinolinone derivatives. This technology represents a paradigm shift from traditional stoichiometric oxidations to a greener, electricity-driven process that operates in an undivided cell. By leveraging the power of anodic oxidation, this method effectively couples N-substituted benzamides with sulfonating reagents to form the target heterocycles without the reliance on transition metal catalysts or hazardous chemical oxidants. For R&D directors and procurement managers alike, this innovation promises a pathway to higher purity intermediates with a drastically simplified supply chain footprint.
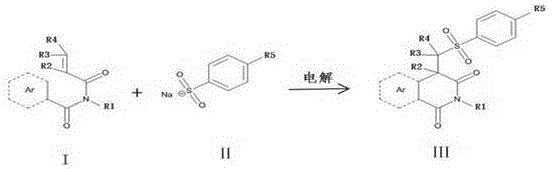
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 4-sulfonic acid substituted isoquinolinone frameworks has relied heavily on methodologies that pose significant challenges for industrial scalability and environmental compliance. Prior art, such as the work by Xiangsheng Xu, utilized tert-butyl hydroperoxide (TBHP) as a stoichiometric oxidant in the presence of tetrabutylammonium iodide, requiring elevated temperatures and generating substantial organic waste. Other approaches, like those reported by Peipei Sun, depended on expensive and scarce iridium-based photoredox catalysts, which not only drive up raw material costs but also introduce risks of heavy metal contamination in the final API intermediate. Furthermore, methods involving silver nitrate catalysis and persulfate co-oxidation often suffer from long reaction times and variable yields, creating bottlenecks in production schedules. These conventional routes typically necessitate rigorous purification steps to remove metal residues, adding complexity and cost to the downstream processing workflow.
The Novel Approach
In stark contrast, the electrochemical oxidation method disclosed in CN110616439B offers a streamlined alternative that directly addresses these pain points through the use of electrons as clean reagents. This novel approach operates in a simple undivided electrolytic bath, eliminating the need for costly proton exchange membranes that often complicate reactor engineering and maintenance. The process utilizes readily available graphite, glassy carbon, or platinum net electrodes as the anode and common metal sheets like iron or aluminum as the cathode, significantly lowering the capital expenditure required for reactor setup. By conducting the reaction at mild temperatures ranging from 25-80°C and avoiding external chemical oxidants, the method minimizes thermal hazards and waste generation. This transition from chemical to electrochemical driving forces ensures a more consistent reaction profile, reducing the batch-to-batch variability that often plagues traditional catalytic systems.
Mechanistic Insights into Electrochemical Oxidative Cyclization
The core mechanism of this transformation involves the anodic oxidation of the sulfonating reagent, typically a sodium sulfinate, to generate a reactive sulfonyl radical species. Upon application of a constant current, the sulfinate anion loses an electron at the anode surface, initiating a radical cascade that targets the electron-rich alkene moiety of the N-substituted benzamide substrate. This radical addition is followed by an intramolecular cyclization and subsequent oxidative aromatization or functionalization to yield the stable isoquinolinone core. The beauty of this electrochemical cycle lies in its self-regulating nature; the cathode simultaneously balances the charge, often facilitating proton reduction, which maintains the overall neutrality of the system without the need for additional base additives in many cases. This precise control over the oxidation potential allows for the selective activation of the sulfinate reagent while preserving other sensitive functional groups on the aromatic rings.
From an impurity control perspective, the absence of transition metal catalysts is a decisive advantage for producing high-purity pharmaceutical intermediates. Traditional methods using silver or iridium often leave trace metal residues that are difficult to purge and can catalyze degradation pathways during storage or subsequent formulation. The electrochemical method inherently avoids this contamination vector, resulting in a cleaner crude reaction mixture that simplifies the isolation process. Furthermore, the avoidance of strong chemical oxidants like persulfates reduces the formation of over-oxidized byproducts and sulfone impurities that can arise from non-selective oxidation. The result is a product profile with a superior impurity spectrum, facilitating easier regulatory approval and reducing the burden on quality control laboratories tasked with validating metal limits.
How to Synthesize 4-Sulfonic Acid Isoquinolinone Derivatives Efficiently
Implementing this electrochemical protocol requires careful attention to electrode configuration and electrolyte composition to maximize Faradaic efficiency. The standard procedure involves dissolving the N-substituted benzamide and sodium sulfinate in a mixed solvent system of dioxane and water, with a supporting electrolyte such as ammonium bromide to ensure sufficient conductivity. The reaction is conducted under constant current electrolysis, monitoring progress via liquid chromatography to determine the optimal endpoint. While the specific parameters may vary depending on the substrate's electronic nature, the robustness of the graphite electrode system allows for a wide operating window. For detailed operational parameters and specific workup procedures tailored to your specific derivative, please refer to the standardized synthesis guide below.
- Prepare the reaction mixture by combining N-substituted benzamide, sodium sulfinate, and ammonium bromide in a dioxane and water solvent system within an undivided electrolytic cell.
- Insert graphite sheet electrodes (anode and cathode) into the solution and connect them to a constant current power supply, maintaining a temperature between 25-80°C.
- Electrolyze the mixture at a constant current until LC detection confirms reaction completion, then proceed with solvent removal, extraction, and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical technology translates into tangible strategic benefits regarding cost stability and operational resilience. The elimination of precious metal catalysts removes exposure to the volatile pricing markets associated with elements like Iridium and Palladium, ensuring more predictable raw material costs over the long term. Additionally, the simplified reactor design, which foregoes the need for specialized divided cells or proton membranes, lowers the barrier to entry for contract manufacturing organizations (CMOs) and allows for faster technology transfer between sites. This flexibility enhances supply chain reliability by enabling multi-vendor sourcing strategies without the need for highly specialized equipment that might be limited to a single supplier.
- Cost Reduction in Manufacturing: The economic impact of removing stoichiometric chemical oxidants and expensive metal catalysts is profound, as it reduces both the direct material costs and the indirect costs associated with waste treatment. Without the need to quench and dispose of large quantities of oxidizing agents or recover trace metals, the overall cost of goods sold (COGS) is significantly optimized. Furthermore, the milder reaction conditions reduce energy consumption related to heating and cooling, contributing to a leaner manufacturing budget. These cumulative savings allow for more competitive pricing structures in the final supply of pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as graphite electrodes and common salts like ammonium bromide ensures that the supply chain is not vulnerable to geopolitical disruptions affecting rare earth or precious metal mining. The robustness of the undivided cell setup means that production can be scaled across multiple facilities with standard electrochemical equipment, mitigating the risk of single-point failures. This decentralization capability is crucial for maintaining continuous supply lines for critical drug intermediates, especially in times of global logistical constraints.
- Scalability and Environmental Compliance: Scaling electrochemical reactions is increasingly straightforward with modern flow chemistry and plate-and-frame reactor technologies, making this method highly suitable for ton-scale production. The inherent green chemistry attributes, such as the reduction of hazardous waste and the use of electricity as a renewable reagent, align perfectly with stringent environmental regulations and corporate sustainability goals. This compliance reduces the regulatory burden and potential fines associated with hazardous waste disposal, smoothing the path for long-term commercial operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of isoquinolinone derivatives, based on the data provided in patent CN110616439B. These insights are designed to clarify the feasibility of adopting this technology for your specific project needs and to highlight the operational advantages over legacy synthetic routes. Understanding these nuances is essential for making informed decisions about process development and vendor selection.
Q: What are the primary advantages of this electrochemical method over traditional metal-catalyzed routes?
A: The electrochemical method described in CN110616439B eliminates the need for expensive and toxic transition metal catalysts like Iridium or Silver, and avoids hazardous chemical oxidants such as tert-butyl hydroperoxide. This results in a cleaner product profile with significantly reduced metal ion residues and lower waste disposal costs.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes an undivided electrolytic cell which simplifies reactor design and reduces equipment costs by removing the need for proton exchange membranes. The use of robust graphite electrodes and mild reaction conditions (25-80°C) makes it highly adaptable for commercial scale-up.
Q: What types of substrates are compatible with this synthesis route?
A: The method demonstrates broad substrate scope, successfully accommodating various N-substituted benzamides with different electronic properties (electron-donating or withdrawing groups) and diverse sodium sulfinate reagents, yielding the corresponding 4-sulfonic acid substituted isoquinolinone derivatives efficiently.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Sulfonic Acid Isoquinolinone Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modernizing the production of complex pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN110616439B can be seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest standards of quality and consistency required by global regulatory bodies.
We invite you to collaborate with our technical team to explore how this metal-free electrochemical route can optimize your specific supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits tailored to your volume requirements. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and sustainable chemistry available.
