Advanced CDI-Mediated Synthesis of Benzimidazole Carboxamides for Commercial Scale-up of Complex Pharmaceutical Intermediates
Advanced CDI-Mediated Synthesis of Benzimidazole Carboxamides for Commercial Scale-up of Complex Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust synthetic methodologies that balance high purity with environmental sustainability, particularly for critical antihypertensive intermediates. Patent CN101781256B introduces a transformative approach for preparing 4-methyl-2-propyl-1H-benzimidazole-6-carboxamide compounds, which serve as pivotal scaffolds in the development of Angiotensin II AT1 receptor antagonists. This technology replaces hazardous thionyl chloride activation with a milder N,N'-carbonyldiimidazole (CDI) mediated pathway, addressing long-standing issues regarding solubility, yield, and waste management. By leveraging this innovation, manufacturers can achieve superior process control and product quality, positioning themselves as a reliable pharmaceutical intermediates supplier capable of meeting stringent global regulatory standards. The shift from traditional chlorination to carbodiimide coupling represents a significant leap forward in green chemistry applications within the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzimidazole carboxamides relied heavily on the conversion of carboxylic acids to acid chlorides using thionyl chloride, a process fraught with operational and environmental challenges. The primary drawback involves the poor solubility of the starting carboxylic acid in organic solvents or thionyl chloride itself, leading to heterogeneous reaction mixtures that are difficult to stir and control effectively. This physical limitation often results in incomplete reactions, necessitating extended processing times and complex purification steps to remove unreacted starting materials. Furthermore, the generation of stoichiometric amounts of sulfur dioxide and hydrogen chloride gas poses severe environmental hazards, requiring expensive scrubbing systems and specialized corrosion-resistant equipment. The resulting amide products frequently suffer from lower purity profiles due to side reactions inherent to the highly reactive acid chloride species, typically capping overall yields below 80 percent.
The Novel Approach
The methodology disclosed in the patent utilizes N,N'-carbonyldiimidazole to activate the carboxylic acid moiety under mild conditions, fundamentally altering the reaction landscape for better efficiency and safety. This activation strategy proceeds smoothly at room temperature or with moderate heating, ensuring homogeneous reaction conditions that facilitate complete conversion without the need for aggressive reagents. By avoiding the formation of acid chlorides, the process eliminates the release of toxic acidic gases, thereby drastically simplifying the engineering controls required for industrial production. The resulting acyl imidazole intermediate is highly selective towards nucleophilic attack by amines, leading to cleaner reaction profiles and significantly enhanced product purity. This streamlined approach not only improves the economic viability of the synthesis but also aligns with modern sustainability goals by reducing the chemical footprint of the manufacturing process.
Mechanistic Insights into CDI-Mediated Amidation
The core of this synthetic advancement lies in the formation of an acyl imidazole intermediate through the nucleophilic attack of the carboxylate oxygen on the carbonyl carbon of N,N'-carbonyldiimidazole. This activation step generates a highly electrophilic species that is sufficiently reactive to undergo amidation with various amines yet stable enough to minimize hydrolysis during the reaction window. The mechanism avoids the formation of harsh electrophiles like acid chlorides, which are prone to reacting with sensitive functional groups on the benzimidazole ring or the amine substrate. Consequently, the reaction pathway is highly chemoselective, preserving the integrity of the 4-methyl-2-propyl-1H-benzimidazole core while efficiently forming the desired amide bond. This mechanistic elegance ensures that the final product retains a high degree of structural fidelity, which is critical for downstream biological activity in antihypertensive applications.
Impurity control is inherently superior in this CDI-mediated system due to the absence of chlorinating agents that often lead to halogenated byproducts or ring-chlorinated impurities. The byproduct of the activation step is imidazole, a solid that is easily removed during the aqueous workup or crystallization phases, unlike the gaseous byproducts of thionyl chloride which require complex trapping. The mild reaction conditions, typically ranging from 20°C to 100°C, prevent thermal degradation of the sensitive benzimidazole scaffold, further contributing to a cleaner impurity profile. This high level of control over the reaction environment allows for the synthesis of diverse derivatives with varying R groups, as illustrated in the general structure below, without compromising on yield or quality.
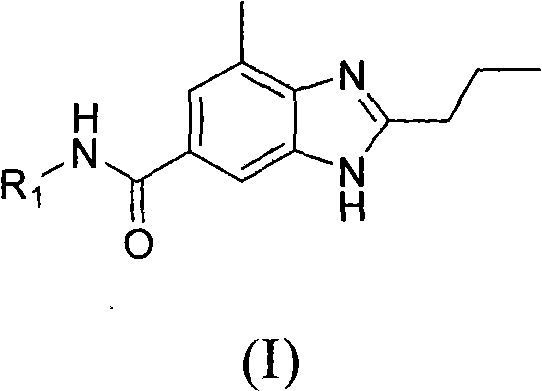
The versatility of this method is demonstrated by its compatibility with a broad spectrum of amine nucleophiles, ranging from simple alkyl amines to complex heterocyclic amines containing piperazine or morpholine rings. The steric and electronic properties of the acyl imidazole intermediate allow it to accommodate bulky amines that might struggle with traditional acid chloride coupling due to steric hindrance. This flexibility is paramount for medicinal chemists exploring structure-activity relationships, as it enables the rapid generation of diverse libraries of analogues for biological screening. The consistent performance across different substrates underscores the robustness of the CDI activation strategy, making it a preferred choice for both laboratory-scale discovery and large-scale commercial production of high-purity pharmaceutical intermediates.
How to Synthesize 4-methyl-2-propyl-1H-benzimidazole-6-carboxamide Efficiently
Implementing this synthesis requires careful attention to stoichiometry and solvent selection to maximize the efficiency of the CDI activation step. The process begins with the dissolution of the carboxylic acid precursor in a polar aprotic solvent such as DMF or DCM, followed by the addition of CDI to generate the active species. Once activation is complete, the amine component is introduced, and the mixture is heated to drive the amidation to completion. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and optimal yield.
- Dissolve 4-methyl-2-propyl-1H-benzimidazole-6-carboxylic acid in a suitable organic solvent such as DMF or DCM at room temperature.
- Activate the carboxylic acid by adding N,N'-carbonyldiimidazole (CDI) and stirring for at least 30 minutes to form the reactive acyl imidazole intermediate.
- Introduce the desired amine compound to the mixture and heat between 20-100°C for 2-24 hours to complete the amidation, followed by aqueous workup to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this CDI-based route offers substantial strategic benefits by mitigating risks associated with hazardous reagent handling and waste disposal. The elimination of thionyl chloride removes the need for specialized storage and handling protocols for corrosive liquids, thereby lowering operational overhead and insurance costs associated with dangerous goods. Additionally, the simplified workup procedure, which often involves simple precipitation and filtration rather than complex distillation or extraction, reduces the consumption of energy and auxiliary solvents. These operational efficiencies translate directly into a more resilient supply chain, as the process is less susceptible to disruptions caused by environmental compliance audits or raw material shortages of specialized chlorinating agents.
- Cost Reduction in Manufacturing: The transition to N,N'-carbonyldiimidazole activation eliminates the capital expenditure required for corrosion-resistant reactors and extensive gas scrubbing systems needed for thionyl chloride processes. By removing the generation of hazardous acidic gases, the facility can operate with standard stainless steel equipment, significantly lowering maintenance costs and extending asset life. Furthermore, the higher yields achieved with this method mean that less raw material is wasted per unit of product, optimizing the cost of goods sold. The reduction in waste treatment costs, specifically regarding the neutralization and disposal of acidic effluents, provides an additional layer of financial savings that enhances overall profitability.
- Enhanced Supply Chain Reliability: N,N'-carbonyldiimidazole is a commercially available reagent with a stable global supply chain, reducing the risk of production stoppages due to reagent unavailability. The robustness of the reaction conditions allows for greater flexibility in scheduling and batch sizing, enabling manufacturers to respond more agilely to fluctuating market demands. The simplified purification process shortens the overall cycle time from raw material intake to finished goods, effectively reducing lead time for high-purity pharmaceutical intermediates. This reliability is crucial for maintaining continuous supply to downstream API manufacturers who depend on just-in-time delivery models.
- Scalability and Environmental Compliance: The absence of toxic gas evolution makes this process inherently safer and easier to scale from pilot plant to multi-ton commercial production without significant engineering redesigns. Regulatory compliance is streamlined as the process generates less hazardous waste, aligning with increasingly strict environmental protection laws and corporate sustainability mandates. The ability to produce high-quality intermediates with a lower environmental footprint enhances the brand reputation of the manufacturer as a responsible partner in the pharmaceutical value chain. This alignment with green chemistry principles future-proofs the production capability against tightening global regulations on chemical manufacturing emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic route, derived directly from the patent specifications and experimental data. Understanding these nuances is essential for process engineers and quality assurance teams looking to integrate this technology into their existing manufacturing workflows. The answers provide clarity on solvent choices, reaction parameters, and expected outcomes to facilitate informed decision-making.
Q: What are the advantages of using CDI over thionyl chloride for this synthesis?
A: Using N,N'-carbonyldiimidazole eliminates the generation of corrosive gases like SO2 and HCl, significantly reducing environmental pollution and equipment corrosion risks while improving product purity by avoiding acid chloride side reactions.
Q: What solvents are compatible with this CDI-mediated amidation process?
A: The patent specifies a wide range of compatible solvents including dichloromethane, chloroform, tetrahydrofuran, acetone, ethyl acetate, and N,N-dimethylformamide, offering flexibility for process optimization.
Q: What yields can be expected from this novel synthetic route?
A: Experimental data indicates consistently high yields ranging from 85% to 96% across various amine substrates, surpassing the typical sub-80% yields associated with conventional acid chloride methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-methyl-2-propyl-1H-benzimidazole-6-carboxamide Supplier
At NINGBO INNO PHARMCHEM, we leverage advanced synthetic technologies like the CDI-mediated amidation described in CN101781256B to deliver superior quality intermediates for the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 4-methyl-2-propyl-1H-benzimidazole-6-carboxamide meets the highest industry standards for impurity profiles and physicochemical properties. Our commitment to process excellence allows us to offer a stable and secure source of critical building blocks for antihypertensive drug development.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through our innovative process solutions. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your project timelines and quality objectives. Let us be your partner in bringing high-quality pharmaceutical intermediates to market efficiently and sustainably.
