Advanced Catalytic Oxidation for High-Purity Tazobactam Intermediates and Commercial Scalability
Introduction to Next-Generation Beta-Lactam Oxidation Technology
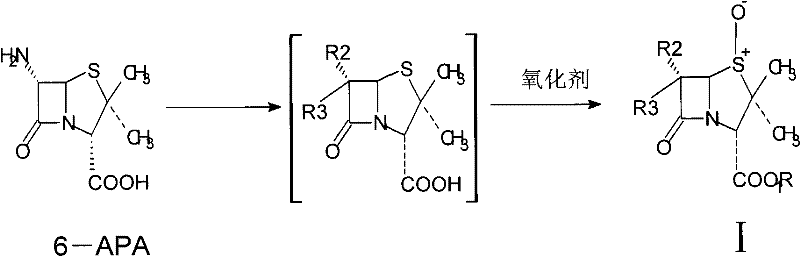
The synthesis of high-value beta-lactamase inhibitors, such as Tazobactam, relies critically on the efficient production of key intermediates like penicillanic acid sulfoxide. A pivotal advancement in this domain is detailed in patent CN101434609B, which introduces a novel catalytic oxidation system utilizing tungstate or molybdate catalysts coupled with hydrogen peroxide. This technology represents a significant departure from traditional, hazardous oxidation methods, offering a pathway that aligns perfectly with modern green chemistry principles. For R&D directors and process engineers, this patent provides a robust framework for achieving high selectivity and yield while mitigating the environmental and safety risks associated with strong oxidants. The ability to convert 6-amino-penicillanic acid (6-APA) into the sulfoxide intermediate with minimal byproduct formation is essential for maintaining the integrity of the subsequent ring-expansion reactions required for cephalosporin and carbacephem synthesis.
Furthermore, the commercial implications of adopting this catalytic system are profound for any organization seeking a reliable pharmaceutical intermediate supplier. The process described not only enhances the purity profile of the final intermediate but also streamlines the downstream processing requirements. By avoiding the use of corrosive and unstable peracids, manufacturers can significantly reduce the complexity of their waste treatment protocols and improve overall plant safety. This report delves deep into the mechanistic advantages, operational parameters, and supply chain benefits of this technology, providing a comprehensive analysis for stakeholders involved in the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the oxidation of penicillanic acid derivatives has been fraught with challenges related to reagent stability, safety, and selectivity. Traditional methods often rely on meta-chloroperbenzoic acid (mCPBA) or high-concentration peracetic acid, both of which present substantial logistical and operational hurdles. The preparation of mCPBA is notoriously difficult and costly, driving up the raw material expenses for large-scale manufacturing. Alternatively, the use of peracetic acid requires handling high-density solutions that are thermally unstable and prone to explosive decomposition if not managed with extreme caution. These conventional oxidants often lack the necessary chemoselectivity, leading to over-oxidation where the desired sulfoxide is further oxidized to the corresponding sulfone, a difficult-to-remove impurity that compromises the quality of the Tazobactam precursor.
Moreover, the reaction conditions for these traditional methods are often unforgiving. For instance, peracetic acid oxidation typically requires strict temperature control near 0°C to prevent decomposition, yet the byproduct acetic acid can lower the freezing point of the mixture, causing stirring difficulties and heterogeneous reaction environments. This inconsistency leads to variable yields and complicates the crystallization process of the penicillanic acid sulfoxide. The presence of residual acetic acid and the need for extensive washing steps to remove acidic byproducts further burden the production workflow. Consequently, reliance on these legacy methods results in higher operational costs, increased safety liabilities, and a larger environmental footprint due to the generation of chlorinated or acidic waste streams.
The Novel Approach
In stark contrast, the catalytic system disclosed in the patent utilizes a tungstate or molybdate catalyst activated by hydrogen peroxide, creating a highly efficient and environmentally benign oxidation environment. This approach fundamentally shifts the paradigm from stoichiometric oxidation using hazardous reagents to a catalytic cycle driven by a safe, green oxidant. The reaction proceeds under mild acidic conditions (pH 1-3) and low temperatures (0-5°C), ensuring the stability of the sensitive beta-lactam ring while facilitating rapid conversion. The use of hydrogen peroxide is particularly advantageous as its reduction product is merely water, eliminating the formation of organic acid waste and simplifying the workup procedure significantly.
The selectivity of this novel system is exceptional, effectively suppressing the formation of sulfone byproducts which are common in non-catalytic oxidations. This high fidelity ensures that the resulting penicillanic acid sulfoxide possesses a sharp melting point (162-164°C) and high purity, as confirmed by HPLC analysis showing no residual starting material or over-oxidized impurities. For procurement teams, this translates to a more consistent supply of high-quality intermediates that require less reprocessing. The ability to recycle the tungstate catalyst further enhances the economic viability of this method, making it a superior choice for cost reduction in API manufacturing compared to single-use oxidant systems.
Mechanistic Insights into Tungstate-Catalyzed Oxidation
The core of this technological breakthrough lies in the formation of an active peroxo-tungstate complex in situ. When sodium tungstate is mixed with hydrogen peroxide in an acidic medium, it generates a reactive species capable of transferring an oxygen atom to the sulfur atom of the thioether moiety in 6-APA. This mechanism is highly specific, targeting the sulfide bond without attacking the vulnerable beta-lactam amide bond or the carboxylic acid group. The acidic environment, maintained typically with hydrochloric or oxalic acid at a pH between 1 and 3, is crucial for stabilizing the peroxo-complex and ensuring optimal reaction kinetics. This precise control over the active species prevents the runaway radical reactions often seen with organic peroxides, thereby safeguarding the structural integrity of the penicillin nucleus.
From an impurity control perspective, the mechanism inherently limits over-oxidation. The catalytic cycle is tuned such that once the sulfoxide is formed, it is less susceptible to further oxidation under these specific conditions compared to the starting sulfide. This kinetic selectivity is vital for pharmaceutical applications where impurity profiles are strictly regulated. Furthermore, the solubility characteristics of the product in the reaction medium facilitate easy isolation. As the reaction progresses, the penicillanic acid sulfoxide crystallizes out upon acidification and cooling, allowing for a straightforward filtration process. This ease of separation reduces the need for complex extraction solvents, aligning with the principles of green chemistry and reducing the load on solvent recovery systems.
How to Synthesize Penicillanic Acid Sulfoxide Efficiently
The practical implementation of this catalytic oxidation system involves a streamlined sequence of operations designed for reproducibility and safety. The process begins with the preparation of the catalytic solution, followed by the controlled addition of the substrate, and concludes with a simple crystallization workup. This standardized approach minimizes operator error and ensures consistent batch-to-batch quality, which is critical for regulatory compliance in pharmaceutical manufacturing. The detailed procedural steps below outline the specific parameters required to achieve the high yields and purity reported in the patent literature.
- Prepare the catalytic oxidation system by mixing sodium tungstate and 30% hydrogen peroxide, cooling to 0-5°C, and adjusting pH to 2.0 with hydrochloric acid.
- Add 6-amino-penicillanic acid (6-APA) in batches while strictly maintaining the reaction temperature below 5°C for approximately 2 hours.
- Adjust pH to 1.0 using sulfuric acid, cool to 0°C for crystallization, filter, wash with cold water, and dry under vacuum to obtain the white sulfoxide product.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this tungstate-catalyzed oxidation technology offers transformative benefits for the supply chain and procurement sectors of the pharmaceutical industry. By shifting away from hazardous, single-use oxidants like mCPBA and concentrated peracetic acid, manufacturers can drastically simplify their raw material sourcing strategies. Hydrogen peroxide and sodium tungstate are commodity chemicals with stable global supply chains, reducing the risk of shortages that often plague specialized reagents. This stability ensures continuous production schedules and protects against market volatility, providing a secure foundation for long-term supply agreements. Additionally, the elimination of dangerous reagents lowers insurance premiums and reduces the regulatory burden associated with storing and transporting hazardous materials.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the recyclability of the catalyst and the low cost of the oxidant. Unlike traditional methods where the oxidant is consumed stoichiometrically and generates expensive waste, the tungstate catalyst can be recovered from the aqueous phase and reused multiple times without significant loss of activity. This circular usage model significantly lowers the cost of goods sold (COGS) for the intermediate. Furthermore, the simplified workup procedure, which avoids complex extractions and extensive washing to remove organic acids, reduces utility consumption and labor hours, contributing to substantial overall cost savings in the production lifecycle.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions enhances the reliability of the supply chain. The tolerance for mild temperatures and the use of stable reagents mean that the process is less susceptible to disruptions caused by equipment failure or environmental fluctuations. The high yield (approximately 98%) ensures that less starting material is wasted, maximizing the throughput of existing manufacturing assets. For supply chain heads, this efficiency translates to shorter lead times and the ability to respond more agilely to market demand fluctuations for Tazobactam and related beta-lactam antibiotics, securing a competitive edge in the marketplace.
- Scalability and Environmental Compliance: From an environmental standpoint, this process is inherently scalable and compliant with increasingly stringent global regulations. The primary byproduct is water, which eliminates the need for costly treatment of chlorinated or acidic organic waste streams. This "green" profile facilitates easier permitting for plant expansions and reduces the carbon footprint of the manufacturing site. The ability to scale this reaction from laboratory benchtop to multi-ton production without compromising safety or yield makes it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates, ensuring that growth is sustainable and environmentally responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic oxidation system. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the operational feasibility and quality outcomes of the process. Understanding these details is crucial for technical teams evaluating the transition from legacy oxidation methods to this advanced catalytic approach.
Q: What are the primary advantages of using the tungstate-hydrogen peroxide system over traditional peracetic acid methods?
A: The tungstate-hydrogen peroxide system eliminates the need for hazardous, high-concentration peracetic acid or expensive mCPBA. It operates under mild conditions (0-5°C), produces water as the only byproduct, and offers superior chemoselectivity, preventing over-oxidation to sulfones which simplifies purification.
Q: Can the tungstate catalyst be recycled in this oxidation process?
A: Yes, the patent data indicates that the tungstate catalyst can be recovered from the aqueous layer by acidification to precipitate tungstic acid, which can then be reused. The catalyst maintains significant activity even after multiple cycles, reducing raw material consumption.
Q: What is the expected purity and yield of the penicillanic acid sulfoxide produced?
A: According to the experimental examples in the patent, the process achieves yields of approximately 96% to 98%. The resulting product has a melting point of 162-164°C and HPLC analysis confirms the absence of residual 6-APA and sulbactam impurities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Penicillanic Acid Sulfoxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and production of life-saving antibiotics. Our technical team has extensively analyzed the catalytic oxidation pathways described in patent CN101434609B and possesses the expertise to implement this green chemistry approach at an industrial level. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of materials that meet stringent purity specifications. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying the absence of critical impurities such as sulfones and residual starting materials, guaranteeing the integrity of your downstream synthesis.
We invite you to collaborate with us to optimize your supply chain for Tazobactam intermediates. By leveraging our technical capabilities, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us help you achieve a more sustainable, cost-effective, and reliable supply of penicillanic acid sulfoxide, driving value and efficiency across your entire pharmaceutical manufacturing operation.
