Advanced Catalytic Oxidation for High-Purity Fluorosulfone Agrochemical Intermediates
Advanced Catalytic Oxidation for High-Purity Fluorosulfone Agrochemical Intermediates
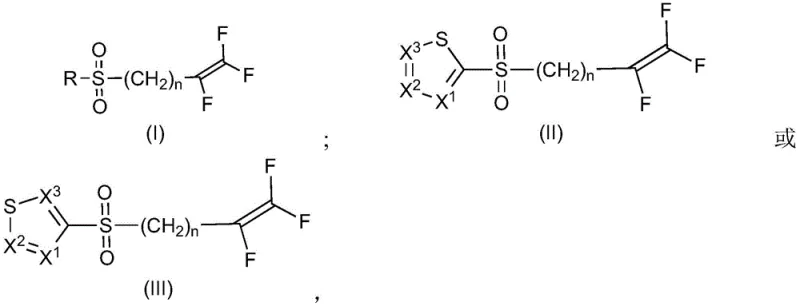
The global demand for effective nematicides in modern agriculture has driven significant innovation in the synthesis of fluorinated sulfone intermediates. Patent CN114174274A introduces a transformative methodology for the selective oxidation of 5-chloro-2-((3,4,4-trifluorobut-3-en-1-yl)thio)-1λ3,3λ2-thiazole and its analogs into their corresponding sulfones. This technical breakthrough addresses long-standing challenges in agrochemical manufacturing, specifically regarding impurity profiles and environmental impact. By utilizing a metal oxide-based catalyst system in conjunction with hydrogen peroxide within an acidic aqueous phase, the process achieves exceptional selectivity while minimizing hazardous waste generation. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, this technology represents a pivotal shift towards greener, more cost-effective production of critical crop protection agents.
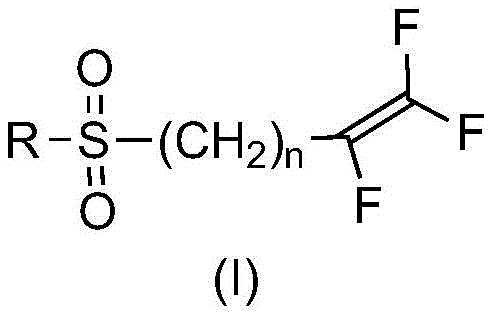
The core innovation lies in the precise control of reaction conditions to favor sulfone formation over oxidative cleavage. Traditional methods often struggle with the electrophilic nature of the trifluoroolefin group adjacent to the sulfur atom, leading to degradation. The disclosed method mitigates this by modulating the reactivity of the oxidant through pH control and catalysis. This ensures that the valuable fluorinated double bond remains intact, preserving the biological activity of the final nematicide. As the industry moves towards stricter environmental regulations, adopting such catalytic oxidation processes becomes not just a technical preference but a strategic necessity for sustainable supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the oxidation of sulfides to sulfones in complex fluorinated systems has been plagued by significant technical hurdles. Prior art, such as WO/2001/002378, describes oxidation using peroxygen reagents in the presence of monocarboxylic acids like acetic acid. While functional, these methods often require elevated temperatures, such as 60°C, and result in moderate yields around 76%. More critically, the use of stoichiometric oxidants like Oxone or Caroat introduces large quantities of environmentally harmful sulfates into the waste stream. Furthermore, non-catalytic oxidation with hydrogen peroxide often leads to the formation of sulfoxide intermediates that are difficult to oxidize further, requiring prolonged reaction times and higher temperatures that degrade the sensitive fluorinated alkene moiety.
The Novel Approach
The novel approach detailed in the patent overcomes these deficiencies by employing a biphasic reaction system with a metal oxide catalyst. By conducting the reaction in an acidic aqueous phase, the nucleophilicity of the hydrogen peroxide is effectively suppressed. This suppression is crucial because it prevents the oxidant from attacking the electrophilic carbon-carbon double bond of the trifluoroolefin group. Consequently, the reaction proceeds selectively to the sulfone stage without generating significant amounts of cleavage by-products. This method allows for operation at milder temperatures, typically between 20°C and 40°C, and utilizes catalytic amounts of inexpensive metal salts like sodium tungstate, drastically reducing raw material costs and waste disposal burdens compared to stoichiometric oxidants.
Mechanistic Insights into Metal Oxide Catalyzed Oxidation
The mechanistic elegance of this process relies on the interplay between the metal oxide catalyst and the acidic medium. In the absence of acid, hydrogen peroxide acts as a strong nucleophile, prone to attacking the electron-deficient double bond of the trifluoroolefin group, especially when activated by an adjacent sulfone or sulfoxide group. However, under acidic conditions (e.g., using 4M sulfuric acid), the protonation equilibrium shifts, reducing the concentration of the reactive hydroperoxide anion. The metal oxide catalyst, such as sodium tungstate (Na2WO4), then facilitates the oxygen transfer specifically to the sulfur atom. This catalytic cycle ensures rapid conversion of the sulfide to the sulfoxide and subsequently to the sulfone, bypassing the kinetic barriers associated with uncatalyzed pathways.
Impurity control is a paramount concern for pharmaceutical and agrochemical manufacturers. A major side reaction in this chemistry is the oxidative cleavage of the fluorinated double bond, which yields 3-((5-chloro-1λ3,3λ2-thiazol-2-yl)sulfonyl)propanoic acid and hydrogen fluoride. As illustrated in the structural analysis of by-products, this degradation pathway is highly undesirable as it destroys the active pharmacophore. The patented process successfully limits the formation of such carboxylic acid by-products to less than 2% by weight, and in optimized examples, down to 1%. This high level of purity (>97%) significantly simplifies downstream purification, eliminating the need for complex chromatography or recrystallization steps that often erode overall process yield.
How to Synthesize 5-chloro-2-((3,4,4-trifluorobut-3-en-1-yl)sulfonyl)-1λ3,3λ2-thiazole Efficiently
Implementing this synthesis route requires careful attention to phase separation and catalyst loading. The process is designed to be robust and scalable, utilizing commercially available reagents like 60% hydrogen peroxide and standard mineral acids. The key to success lies in maintaining the biphasic nature of the system, which allows for the easy recovery of the catalyst. Detailed operational parameters, including specific addition rates and temperature profiles, are critical to maintaining the high selectivity observed in the patent examples. For process chemists looking to replicate this efficiency, the following standardized guide outlines the essential steps derived from the experimental data.
- Prepare a reaction mixture containing the sulfide substrate (Formula Ia), a metal oxide catalyst such as sodium tungstate (Na2WO4), and an acidic aqueous solution (e.g., 4M sulfuric acid).
- Add a peroxygen oxidizing agent, specifically aqueous hydrogen peroxide (H2O2), dropwise to the mixture while maintaining a temperature between 20°C and 40°C.
- Stir the biphasic reaction mixture until total conversion is achieved, then separate the organic phase containing the high-purity sulfone product from the aqueous catalyst phase for reuse.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this catalytic oxidation technology offers substantial strategic benefits beyond mere technical performance. The primary advantage is the drastic simplification of the waste management profile. By replacing stoichiometric oxidants that generate massive salt loads with a catalytic system based on recoverable metal oxides, manufacturers can significantly reduce the volume of hazardous waste requiring treatment. This reduction directly translates to lower operational expenditures related to waste disposal and environmental compliance. Furthermore, the ability to recycle the aqueous catalyst phase means that the consumption of the metal catalyst is minimized, stabilizing raw material costs against market fluctuations.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric oxidants and the reduction in waste treatment costs lead to a leaner manufacturing process. Since the catalyst can be reused multiple times without significant loss of activity, the effective cost per kilogram of the active ingredient is lowered. Additionally, the high selectivity of the reaction minimizes the loss of valuable fluorinated starting materials to side reactions, ensuring that every gram of input contributes to the final yield. This efficiency is crucial for maintaining competitiveness in the high-volume agrochemical market.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as hydrogen peroxide, sulfuric acid, and sodium tungstate ensures a stable and secure supply chain. Unlike specialized reagents that may face sourcing bottlenecks, these materials are globally available in bulk quantities. The robustness of the reaction conditions, which tolerate a range of temperatures and concentrations, further enhances reliability by reducing the risk of batch failures due to minor process deviations. This stability allows for consistent production schedules and reliable delivery timelines to downstream formulators.
- Scalability and Environmental Compliance: The biphasic nature of the reaction makes it inherently scalable from laboratory benchtop to industrial multi-ton reactors. The ease of phase separation simplifies the work-up procedure, facilitating continuous or semi-continuous processing modes. From an environmental perspective, the process aligns with green chemistry principles by using water as a solvent component and generating minimal hazardous by-products. This compliance with stringent environmental standards future-proofs the manufacturing site against tightening regulations, ensuring long-term operational continuity without the need for costly retrofits.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this fluorosulfone synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on reaction scope and limitations. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: How does the acidic aqueous phase prevent double bond cleavage in fluorosulfone synthesis?
A: The acidic environment reduces the nucleophilicity of the hydrogen peroxide oxidant. This prevents non-catalytic nucleophilic attacks on the electrophilic trifluoroolefin moiety, thereby inhibiting the formation of unwanted carboxylic acid by-products like 3-((5-chloro-1λ3,3λ2-thiazol-2-yl)sulfonyl)propanoic acid.
Q: What catalyst loading is required for efficient sulfide to sulfone oxidation?
A: The patent demonstrates high efficiency with low catalyst loading, typically between 1 mol% and 5 mol% of metal oxide salts such as sodium tungstate (Na2WO4), achieving selectivities up to 96%.
Q: Can the catalyst system be recycled in this fluorosulfone production process?
A: Yes, the process utilizes a biphasic system where the catalyst remains in the aqueous phase. After separating the organic product phase, the aqueous catalyst phase can be replenished with fresh oxidant and acid for reuse without significant loss in activity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorosulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of next-generation nematicides. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab scale to full manufacturing is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of fluorosulfone intermediate meets the exacting standards required by global agrochemical leaders. Our commitment to quality ensures that your downstream synthesis remains uninterrupted and efficient.
We invite you to collaborate with us to leverage this advanced catalytic oxidation technology for your specific product needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can enhance your supply chain resilience and profitability.
