Advanced Oxidative Cleavage of Phenanthrene for Commercial Scale Production of Diphenic Acid
The chemical industry constantly seeks efficient pathways to transform abundant coal tar derivatives into high-value intermediates, and Patent CN1023117C presents a breakthrough methodology for achieving exactly that. This specific intellectual property outlines a sophisticated phase-transfer catalytic oxidation process that converts phenanthrene directly into 2,2'-biphenyldicarboxylic acid, a critical building block for polyamide fibers and specialized dyes. By leveraging the synergistic effects of ruthenium tetroxide and sodium hypochlorite within a biphasic system, this technology overcomes the historical limitations of harsh oxidative cleavage reactions. The process operates under remarkably mild conditions, utilizing inexpensive bleach as the primary oxygen source while maintaining exceptional control over product selectivity. For global procurement teams and R&D directors, this represents a paradigm shift from stoichiometric oxidation methods to a catalytic, sustainable manufacturing model that aligns with modern green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the oxidative degradation of polycyclic aromatic hydrocarbons like phenanthrene has been plagued by severe economic and environmental inefficiencies. Traditional protocols often rely on stoichiometric amounts of potent oxidants such as potassium permanganate or ozone, which drive up raw material costs exponentially and generate massive quantities of hazardous solid waste. For instance, permanganate oxidation produces manganese dioxide sludge that requires complex and costly disposal procedures, creating a significant burden on waste management infrastructure. Furthermore, older methods frequently demand extreme reaction conditions, including high temperatures or highly acidic environments, which can lead to over-oxidation of the target molecule into unwanted byproducts like phthalic acid or complete mineralization to carbon dioxide. These factors collectively result in low atom economy and inconsistent yields, making conventional routes unsuitable for large-scale commercial production where margin compression is a constant threat.
The Novel Approach
In stark contrast, the methodology described in CN1023117C introduces a catalytic cycle that dramatically improves both the economic and operational profile of the synthesis. By employing sodium hypochlorite—a commodity chemical widely available at low cost—as the terminal oxidant, the process eliminates the need for expensive stoichiometric reagents. The innovation lies in the use of a phase-transfer catalyst, specifically tailored quaternary ammonium salts, which facilitate the transport of the hypochlorite anion into the organic phase where the oxidation occurs. This allows the reaction to proceed in a strongly alkaline medium, which surprisingly stabilizes the biphenyldicarboxylic acid product against further degradation, a common failure point in acidic oxidative systems. The result is a streamlined workflow that achieves high conversion rates with superior selectivity, effectively turning a low-value coal tar fraction into a premium chemical intermediate with minimal environmental footprint.
Mechanistic Insights into Ruthenium-Catalyzed Phase Transfer Oxidation
The core of this technological advancement relies on a dynamic redox cycle involving ruthenium species that shuttle between oxidation states to drive the cleavage of the phenanthrene central ring. In this biphasic system, the active oxidant, ruthenium tetroxide (RuO4), resides primarily in the organic phase or at the interface, where it aggressively attacks the electron-rich double bonds of the phenanthrene substrate. As the oxidation proceeds, the ruthenium is reduced to ruthenium dioxide (RuO2), which precipitates out of the solution. Crucially, this reduced species is not a waste product but a key intermediate that migrates to the aqueous-organic interface. Here, it encounters the hypochlorite ions transported by the phase-transfer catalyst, which re-oxidize the RuO2 back to RuO4, thereby regenerating the active catalyst and closing the loop. This continuous regeneration means that only a catalytic amount of ruthenium is required, rather than a full stoichiometric equivalent, fundamentally altering the cost structure of the reaction.
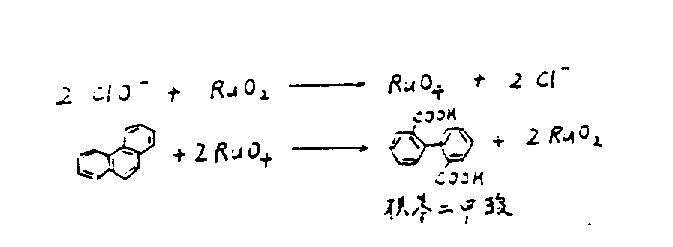
Beyond the catalytic cycle, the control of impurities is meticulously managed through the specific choice of solvent and pH conditions. The patent highlights that maintaining a strongly alkaline environment is critical; in weakly alkaline or acidic media, the target dicarboxylic acid is susceptible to further oxidative decarboxylation, leading to significant yield losses. Additionally, the selection of the organic solvent plays a pivotal role in mass transfer efficiency. While chloroform was initially considered, solvents containing unsaturated linkages or specific chlorinated ethylenes like trichloroethylene were found to accelerate the reaction rate significantly in strong base. The phase-transfer catalyst itself, typically a quaternary ammonium salt with a specific carbon chain length (C15-C20), ensures that the hydrophilic hypochlorite anion can effectively penetrate the lipophilic organic layer where the phenanthrene is dissolved. This precise orchestration of chemical potentials ensures that side reactions are minimized, resulting in a crude product with purity exceeding 99% after simple workup.
How to Synthesize 2,2'-Biphenyldicarboxylic Acid Efficiently
Implementing this synthesis route requires careful attention to the molar ratios of the phase-transfer catalyst and the oxidant to ensure optimal kinetics without compromising safety. The process begins with the suspension of industrial-grade phenanthrene in a chlorinated solvent, followed by the addition of the ruthenium precursor and the phase-transfer agent. The reaction is initiated by the slow addition of sodium hypochlorite solution while rigorously maintaining the pH in the strongly basic range using sodium hydroxide. Monitoring is typically conducted via thin-layer chromatography to determine the endpoint, ensuring complete consumption of the starting material.
- Prepare the reaction mixture by combining industrial phenanthrene, a suitable chlorinated solvent (e.g., trichloroethylene), and a quaternary ammonium phase transfer catalyst.
- Add the ruthenium catalyst precursor and maintain a strongly alkaline environment using sodium hydroxide while introducing sodium hypochlorite as the terminal oxidant.
- Monitor reaction progress via TLC, separate phases, recover the ruthenium dioxide precipitate using sodium bisulfite, and acidify the filtrate to isolate the pure dicarboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain managers and procurement officers, the adoption of this phase-transfer catalytic oxidation method offers profound strategic benefits that extend far beyond simple yield improvements. The most immediate impact is seen in the drastic reduction of raw material expenditures, as the process substitutes costly, specialized oxidants with ubiquitous industrial bleach. This shift not only lowers the direct cost of goods sold but also insulates the manufacturing process from volatility in the pricing of niche chemical reagents. Furthermore, the ability to utilize lower-purity industrial phenanthrene feedstocks eliminates the need for expensive upstream purification steps, allowing manufacturers to source raw materials from a broader, more competitive supplier base. This flexibility enhances supply chain resilience, ensuring continuity of production even when high-grade feedstock availability is constrained.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the catalytic nature of the ruthenium species and the use of sodium hypochlorite. Unlike traditional methods that consume vast quantities of oxidant, this system regenerates the active ruthenium species in situ, meaning the expensive metal is not lost but recycled. Additionally, the reduced formation of hazardous sludge translates to significantly lower waste disposal costs, a hidden expense that often erodes margins in fine chemical manufacturing. The overall effect is a leaner production cost structure that allows for more competitive pricing in the global market for pharmaceutical and polymer intermediates.
- Enhanced Supply Chain Reliability: By relying on commodity chemicals like bleach and caustic soda, the process reduces dependency on complex supply chains for specialized reagents. Sodium hypochlorite is produced globally in massive quantities for water treatment and sanitation, ensuring a stable and secure supply line that is unlikely to face disruptions. Moreover, the robustness of the reaction conditions allows for the use of standard stainless steel or glass-lined reactors without the need for exotic corrosion-resistant materials required by highly acidic alternatives. This compatibility with standard industrial equipment simplifies maintenance and reduces capital expenditure requirements for facility upgrades.
- Scalability and Environmental Compliance: The environmental profile of this method aligns perfectly with increasingly stringent global regulations regarding heavy metal discharge and hazardous waste. Since the ruthenium catalyst precipitates as a solid oxide that can be easily filtered and recovered, the risk of heavy metal contamination in the wastewater stream is virtually eliminated. The solvent system, utilizing recoverable chlorinated hydrocarbons, further supports a closed-loop manufacturing approach. This ease of waste management and the potential for solvent recycling make the process highly scalable, enabling seamless transition from pilot plant batches to multi-ton commercial production without encountering the bottlenecks typical of batch-wise stoichiometric oxidations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidation technology, derived directly from the patent specifications and experimental data. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing production lines.
Q: What are the primary advantages of using sodium hypochlorite over potassium permanganate for this oxidation?
A: Sodium hypochlorite serves as a significantly more economical terminal oxidant compared to potassium permanganate, drastically reducing raw material costs while generating less hazardous solid waste (manganese dioxide sludge).
Q: How is the expensive ruthenium catalyst managed to ensure cost-effectiveness?
A: The process utilizes a catalytic cycle where ruthenium tetroxide is regenerated in situ; furthermore, the reduced ruthenium dioxide precipitate can be quantitatively recovered and recycled, minimizing heavy metal loss.
Q: Can this method tolerate lower purity industrial phenanthrene feedstocks?
A: Yes, the patented method is robust enough to process industrial-grade phenanthrene with a content exceeding 75%, eliminating the need for costly pre-purification steps often required by other oxidation protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2'-Biphenyldicarboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the phase-transfer catalytic oxidation route described in CN1023117C for producing high-purity 2,2'-biphenyldicarboxylic acid. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch meets the exacting standards required for downstream applications in polyamide synthesis and dye manufacturing. We are committed to delivering consistent quality while optimizing the process parameters to maximize yield and minimize environmental impact.
We invite forward-thinking partners to collaborate with us to leverage this advanced technology for their supply chains. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your product portfolio and reduce your overall cost of ownership.
