Scalable Synthesis of Neuroprotective Steroid Intermediates for Commercial API Production
Introduction to Advanced Steroid Intermediate Manufacturing
The pharmaceutical industry constantly seeks reliable sources for complex bioactive molecules, particularly those with demonstrated neuroprotective properties. Patent CN100436474C introduces a robust synthetic methodology for producing cholestan-3β, 5α, 6β, 19-tetrahydroxy-24-one and its derivatives, such as 24-methylene-cholesta-3β, 5α, 6β, 19-tetraol. Historically, obtaining these polyhydroxylated steroids required extraction from rare marine soft corals like Nephthea albida, a process plagued by extremely low natural abundance and ecological sustainability concerns. This patented approach shifts the paradigm from unpredictable biological sourcing to precise chemical synthesis, utilizing hyodeoxycholic acid as a readily available starting material. By establishing a clear, step-wise chemical pathway, this technology enables the production of high-purity pharmaceutical intermediates essential for developing treatments against neuronal apoptosis induced by low potassium levels. For R&D teams and procurement specialists, understanding this transition from extraction to synthesis is critical for securing long-term supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for acquiring bioactive steroids like 24-methylene-cholesta-3β, 5α, 6β, 19-tetraol relied heavily on direct isolation from marine organisms. This biological extraction strategy suffers from inherent inefficiencies, including seasonal variability, geographical limitations, and the destruction of marine ecosystems. Furthermore, the concentration of these target compounds in coral tissues is infinitesimally low, necessitating the processing of massive quantities of biomass to obtain mere milligrams of product. From a commercial perspective, this creates an untenable cost structure and significant supply chain volatility. Purification from complex biological matrices also introduces challenges in removing trace impurities and potential toxins, complicating regulatory approval for pharmaceutical applications. Consequently, relying on natural extraction restricts the clinical development of these promising neuroprotective agents due to the inability to secure sufficient quantities for preclinical and clinical trials.
The Novel Approach
The synthetic route disclosed in the patent offers a transformative solution by constructing the complex steroid skeleton through controlled organic reactions. Starting from hyodeoxycholic acid, the method employs a sequence of functional group transformations that are highly reproducible and scalable. Key innovations include the use of ultrasound irradiation during the organocadmium reaction to enhance yield and the strategic use of diacetoxyiodobenzene for oxidative ring closure. This chemical approach bypasses the bottlenecks of natural extraction, allowing for the deliberate installation of hydroxyl groups at the 3β, 5α, 6β, and 19 positions with high stereoselectivity. By converting a cheap, abundant bile acid derivative into a high-value neuroprotective intermediate, the process drastically simplifies the manufacturing landscape. This shift not only ensures a consistent supply of material but also allows for the synthesis of structural analogues that do not exist in nature, expanding the chemical space available for drug discovery.
Mechanistic Insights into Ultrasound-Assisted Organometallic Synthesis
The core of this synthetic strategy lies in the efficient construction of the side-chain ketone and the subsequent functionalization of the steroid nucleus. The conversion of the C-24 carboxylic acid to the corresponding ketone is achieved via an acid chloride intermediate reacting with an isopropyl cadmium reagent. Uniquely, this step is performed under ultrasonic radiation, which promotes the formation of the organometallic species and enhances mass transfer, leading to superior reaction kinetics compared to conventional stirring. Following ketone formation, the introduction of oxygen functionality at C-6 and C-19 is executed through a tandem bromohydroxylation and oxidative cyclization sequence. Treatment with N-bromosuccinimide in the presence of perchloric acid generates a bromohydrin intermediate, which is subsequently subjected to UV irradiation with diacetoxyiodobenzene. This photo-induced oxidation facilitates the formation of the strained 6β,19-epoxy ether bridge, a critical structural motif for the biological activity of the final molecule.
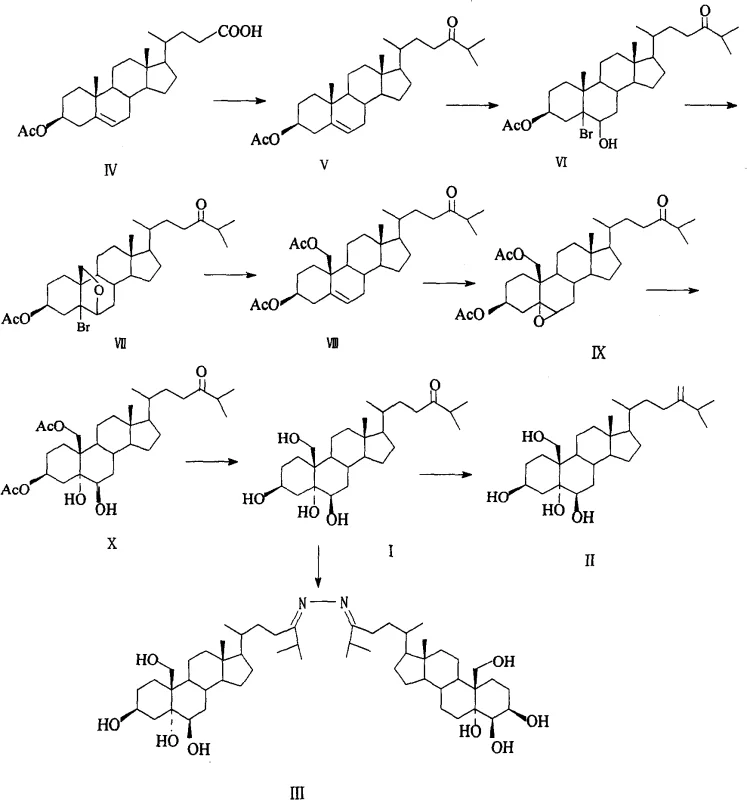
Following the epoxide formation, the synthesis proceeds with a reductive elimination using zinc powder in glacial acetic acid to regenerate the C-5 double bond while retaining the C-19 oxygenation. The final stages involve the oxidation of the 5,6-double bond to install the 5α,6β-diol system, followed by global hydrolysis of the acetyl protecting groups. This mechanistic pathway demonstrates exceptional control over regioselectivity and stereochemistry, ensuring that the final product matches the natural isolate precisely. For process chemists, the ability to tune oxidation conditions—using either potassium permanganate/copper sulfate or m-chloroperbenzoic acid—provides flexibility in optimizing impurity profiles. The rigorous control over each transformation minimizes the formation of by-products, thereby reducing the burden on downstream purification processes and enhancing the overall economic viability of the manufacturing campaign.
How to Synthesize Cholestan-Tetrahydroxy-Ketone Efficiently
Implementing this synthesis requires careful attention to reaction conditions, particularly regarding temperature control and reagent stoichiometry during the oxidation steps. The process begins with the activation of the carboxylic acid and proceeds through several isolation and purification stages, typically involving silica gel column chromatography to ensure high purity at each intermediate stage. Operators must maintain strict anhydrous conditions during the organometallic step and control the pH carefully during the hydrolysis phases to prevent degradation of the sensitive polyol structure. The detailed standardized operating procedures for scaling this route from laboratory to pilot plant are outlined below, providing a roadmap for technical teams to replicate the high yields reported in the patent literature.
- Convert 3β-acetoxy-cholest-5-en-24-oic acid to the corresponding acid chloride using thionyl chloride, followed by reaction with isopropyl cadmium reagent under ultrasonic irradiation to form the 24-ketone.
- Perform stereoselective bromohydroxylation using N-bromosuccinimide and perchloric acid, followed by oxidative ring closure with diacetoxyiodobenzene under UV light to form the 6β,19-epoxy intermediate.
- Reduce the epoxy intermediate with zinc powder in acetic acid to restore the double bond, then oxidize the 5,6-position and hydrolyze acetyl groups to yield the final tetrahydroxy ketone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from marine extraction to total synthesis represents a significant de-risking of the supply base. The reliance on wild-harvested marine resources introduces uncontrollable variables such as environmental regulations, seasonal availability, and geopolitical instability in sourcing regions. By adopting this synthetic route, companies can secure a stable, year-round supply of critical intermediates produced in controlled GMP-compliant facilities. The use of commodity chemicals like hyodeoxycholic acid, thionyl chloride, and zinc powder ensures that raw material costs remain predictable and low. Furthermore, the elimination of complex biological extraction processes removes the need for expensive solvent recovery systems associated with large-volume biomass processing, leading to substantial operational cost savings. This stability allows for better long-term planning and inventory management, crucial for maintaining continuous production lines for downstream API manufacturing.
- Cost Reduction in Manufacturing: The synthetic route eliminates the exorbitant costs associated with collecting and processing tons of marine coral to extract milligrams of product. By utilizing inexpensive starting materials and standard reagents, the cost of goods sold is drastically reduced. The high yield of each step, facilitated by ultrasound assistance and optimized oxidation conditions, minimizes material waste and maximizes output per batch. Additionally, the avoidance of transition metal catalysts that require complex removal steps further lowers processing costs. These factors combine to create a highly economical manufacturing process that makes the final therapeutic agents more accessible to the market.
- Enhanced Supply Chain Reliability: Chemical synthesis decouples production from biological cycles, ensuring that supply is not interrupted by ecological changes or harvesting bans. The scalability of the process means that production capacity can be ramped up quickly to meet surging demand without the lead times associated with biological cultivation. Sourcing of raw materials is simplified as they are widely available from multiple global suppliers, reducing the risk of single-source dependency. This reliability is paramount for pharmaceutical companies aiming to maintain consistent drug supplies for clinical trials and eventual commercial launch.
- Scalability and Environmental Compliance: The process is designed for scale-up, utilizing reactors and conditions common in the fine chemical industry. Waste streams are primarily organic solvents and inorganic salts, which are easier to treat and dispose of compared to the complex organic waste generated by biomass extraction. The use of zinc and acetic acid generates manageable by-products, and the overall atom economy is improved by the high selectivity of the reactions. This aligns with modern green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations while maintaining high production efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of these neuroprotective steroid intermediates. The answers are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and advantages of this method. Understanding these details helps stakeholders make informed decisions about integrating this technology into their development pipelines.
Q: What are the key advantages of this synthetic route over marine extraction?
A: Marine extraction yields are extremely low and variable. This chemical synthesis provides a consistent, scalable supply of high-purity intermediates independent of biological sources.
Q: How is stereochemistry controlled at the C-19 position?
A: The process utilizes a specific oxidative ring closure using diacetoxyiodobenzene under UV irradiation to form the 6β,19-epoxy bridge, ensuring precise stereochemical configuration.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the route uses standard reagents like NBS, zinc powder, and common oxidants, avoiding exotic catalysts, which facilitates easy scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cholestan-Tetrahydroxy-Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of cholestan-3β, 5α, 6β, 19-tetrahydroxy-24-one derivatives in the field of neuroprotection and oncology. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle the specific requirements of steroid chemistry, including moisture-sensitive organometallic reactions and precise oxidation steps. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest quality standards required for pharmaceutical applications. Our commitment to technical excellence ensures that the complex stereochemistry of these molecules is preserved throughout the production process.
We invite you to collaborate with us to leverage this patented synthetic technology for your drug development programs. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a secure, cost-effective supply chain for high-value steroid intermediates, accelerating your path to market.
