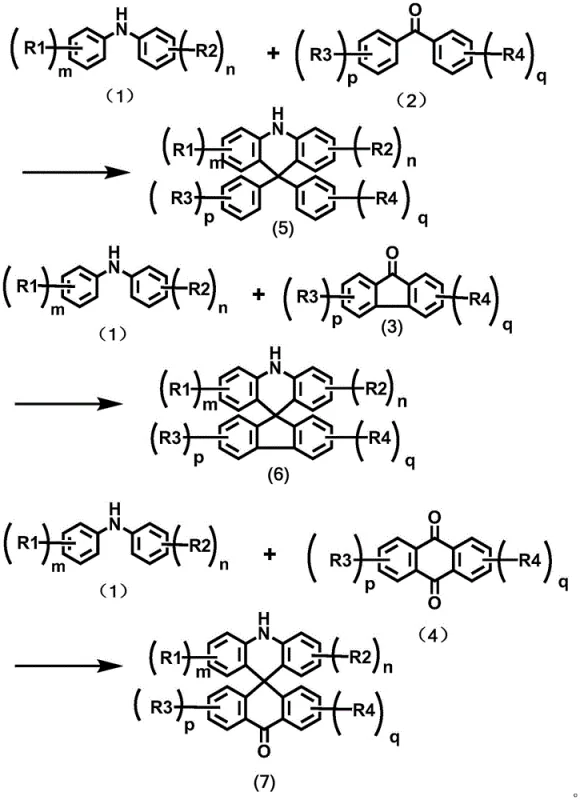
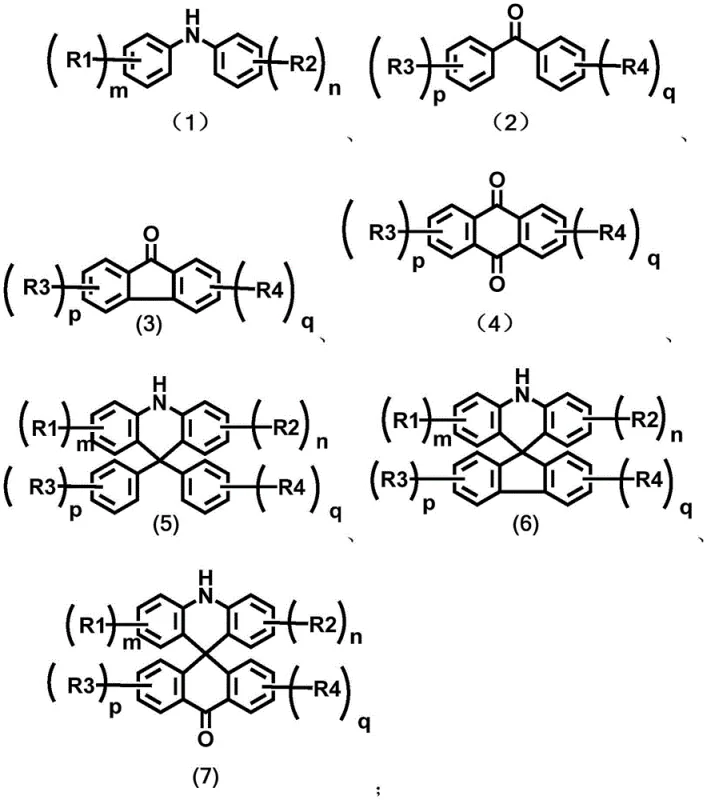
Advanced One-Step Synthesis of Spiro-Substituted Acridines for Commercial OLED Manufacturing
The rapid evolution of organic light-emitting diode (OLED) technology has placed immense pressure on the supply chain to deliver high-performance emissive materials with consistent quality and scalability. Patent CN115477612A introduces a transformative synthesis method for spiro-substituted acridine compounds, which serve as critical donor units in Thermally Activated Delayed Fluorescence (TADF) materials. This intellectual property addresses the longstanding bottlenecks in organic photoelectric material preparation by replacing cumbersome multi-step sequences with a direct, acid-catalyzed cyclization process. For R&D directors and procurement strategists, this innovation represents a pivotal shift towards more efficient manufacturing paradigms that align with the rigorous demands of the display industry. The ability to construct complex spiro-structures in a single operational step not only accelerates development timelines but also fundamentally alters the cost structure of producing high-purity OLED material intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing 9,9'-spiro-substituted acridine derivatives are notoriously inefficient, often relying on hazardous organometallic chemistry that poses significant safety and scalability challenges. Conventional methodologies typically necessitate the protection of aromatic amine precursors followed by lithium-halogen exchange reactions that must be conducted at cryogenic temperatures to maintain control over reactivity. These processes are not only time-consuming, frequently requiring at least two days to complete, but they also involve the use of highly flammable reagents that complicate industrial safety protocols. Furthermore, the subsequent acidification ring closure and deprotection steps introduce additional opportunities for yield loss and impurity generation, making the overall process economically unviable for large-scale commercial production. The reliance on multiple isolation and purification stages between each transformation further exacerbates waste generation and increases the total cost of ownership for manufacturers seeking reliable agrochemical intermediate supplier standards in the electronic sector.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent utilizes a direct condensation strategy that bypasses the need for protective groups and cryogenic conditions entirely. By reacting a diarylamine compound with an aryl ketone compound in the presence of a strong acid catalyst, the synthesis achieves cyclization in a single step with remarkable efficiency. This methodology operates under solvent-free conditions during the critical reaction phase, which dramatically simplifies the workup procedure and reduces the environmental burden associated with volatile organic compound emissions. The reaction temperatures, ranging from 100°C to 220°C, are easily achievable in standard industrial reactors, facilitating a seamless transition from laboratory discovery to commercial scale-up of complex polymer additives and electronic chemicals. This streamlined workflow effectively overcomes the defects of prior art by minimizing operational time and eliminating the need for hazardous reagents, thereby offering a robust pathway for cost reduction in electronic chemical manufacturing.
Mechanistic Insights into Acid-Catalyzed Cyclization
The core of this technological breakthrough lies in the activation of the carbonyl group within the aryl ketone substrate by a strong Brønsted acid catalyst, such as trifluoromethanesulfonic acid or p-toluenesulfonic acid. Upon protonation, the electrophilicity of the carbonyl carbon is significantly enhanced, facilitating a nucleophilic attack by the electron-rich aromatic rings of the diarylamine component. This intramolecular electrophilic aromatic substitution proceeds through a cationic intermediate that subsequently undergoes dehydration to restore aromaticity and lock the spiro-configuration. The rigidity imparted by the spiro-center forces the acridine donor and the acceptor unit into a mutually perpendicular orientation, which is crucial for minimizing the overlap of frontier molecular orbitals. This spatial arrangement effectively reduces the singlet-triplet energy gap (ΔEst), thereby accelerating the reverse intersystem crossing rate that is fundamental to high-efficiency TADF emission. Understanding this mechanistic pathway is essential for optimizing reaction parameters to ensure maximum conversion and minimal formation of regioisomeric impurities.
Impurity control in this synthesis is inherently superior due to the chemoselectivity of the acid-catalyzed mechanism, which avoids the non-specific reactivity often seen with organolithium reagents. The absence of halogen-lithium exchange eliminates the risk of metal-halogen coupling byproducts that are difficult to remove and can act as quenching sites in final OLED devices. Moreover, the high thermal stability of the intermediates allows the reaction to proceed to completion without the degradation issues common in lower-temperature protocols. The resulting crude products typically exhibit high purity profiles, requiring only standard silica gel column chromatography to meet the stringent specifications demanded by the semiconductor and display industries. This level of chemical precision ensures that the final high-purity OLED material maintains consistent photophysical properties across different production batches, a critical factor for maintaining yield in downstream device fabrication.
How to Synthesize Spiro-Substituted Acridine Efficiently
Implementing this synthesis requires precise control over thermal gradients and catalyst loading to maximize yield while maintaining safety. The process begins by heating the diarylamine precursor with the acid catalyst to an initial temperature range of 100-180°C, ensuring complete mixing and activation before the introduction of the ketone substrate. Once the aryl ketone is added, the reaction mixture is ramped to a second heating temperature between 160-220°C, where the cyclization occurs rapidly within 20 to 80 minutes. Detailed standardized synthesis steps see the guide below for specific molar ratios and workup procedures tailored to different substrate variations.
- Mix diarylamine compound and acid catalyst, heating to 100-180°C for 5-30 minutes to activate the system.
- Add aryl ketone compound to the mixture and raise temperature to 160-220°C for 20-50 minutes to complete cyclization.
- Cool the reaction to 40-60°C, dissolve in dichloromethane, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this solvent-free, one-step synthesis offers profound strategic advantages that extend beyond simple unit cost metrics. The elimination of multiple reaction steps and the removal of expensive, hazardous reagents directly translate into a significantly reduced bill of materials and lower operational expenditure. By avoiding the use of cryogenic cooling and complex protection-deprotection sequences, manufacturers can utilize standard reactor infrastructure, thereby decreasing capital investment requirements and increasing asset utilization rates. This process intensification allows for shorter production cycles, which enhances the responsiveness of the supply chain to fluctuating market demands for display and optoelectronic materials. Furthermore, the simplified waste profile resulting from solvent-free operations reduces the regulatory burden and costs associated with environmental compliance and hazardous waste disposal.
- Cost Reduction in Manufacturing: The removal of organic solvents from the reaction phase eliminates the substantial costs associated with solvent procurement, storage, recovery, and distillation. Additionally, the one-step nature of the synthesis reduces labor hours and energy consumption per kilogram of product, leading to substantial cost savings compared to multi-step legacy processes. The use of commercially available acid catalysts rather than specialized organometallic reagents further drives down raw material expenses, making the production of complex spiro-structures economically feasible for high-volume applications.
- Enhanced Supply Chain Reliability: The starting materials, primarily diarylamines and aryl ketones, are readily available from established chemical suppliers, reducing the risk of raw material shortages that often plague specialized synthetic routes. The robustness of the reaction conditions means that production is less susceptible to minor variations in utility supply or equipment performance, ensuring consistent output quality. This reliability is critical for maintaining continuous manufacturing operations and meeting the tight delivery schedules required by major panel manufacturers and electronics integrators.
- Scalability and Environmental Compliance: The solvent-free design inherently minimizes the generation of volatile organic compounds (VOCs), simplifying the permitting process and reducing the need for extensive abatement equipment. The high thermal stability of the process allows for easy scale-up from gram-scale laboratory synthesis to ton-scale commercial production without significant re-engineering of the reaction parameters. This scalability ensures that supply can grow in tandem with market demand for next-generation TADF materials, securing long-term supply continuity for downstream partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process feasibility and material performance. Understanding these details is vital for technical teams evaluating the integration of this route into existing manufacturing portfolios.
Q: What are the primary advantages of this acid-catalyzed method over traditional lithiation routes?
A: This method eliminates the need for cryogenic conditions, flammable organolithium reagents, and multi-step protection/deprotection sequences, significantly simplifying the operational complexity and enhancing safety profiles for large-scale production.
Q: Does this synthesis require organic solvents during the reaction phase?
A: No, the core cyclization reaction proceeds under solvent-free conditions, which drastically reduces waste generation and lowers the environmental footprint associated with solvent recovery and disposal.
Q: What is the typical purity profile achievable with this synthetic route?
A: The one-step cyclization minimizes side reactions and byproduct formation, allowing for high-purity specifications suitable for electronic applications after standard purification processes like column chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiro-Substituted Acridine Supplier
As the demand for high-efficiency OLED materials continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthetic technologies and robust manufacturing capabilities. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every batch of spiro-substituted acridine meets the exacting standards required for advanced electronic applications. We are committed to delivering value through technical excellence and operational reliability.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your material costs and supply security. Contact us today to request a Customized Cost-Saving Analysis tailored to your specific production volumes. Our experts are ready to provide specific COA data and route feasibility assessments to support your R&D and sourcing strategies, ensuring a seamless transition to this superior manufacturing technology.
