Revolutionizing Clavulanic Acid Purification via Stable Diamine Salt Intermediates
Revolutionizing Clavulanic Acid Purification via Stable Diamine Salt Intermediates
The pharmaceutical industry continuously seeks robust methods to enhance the stability and purity of beta-lactam antibiotics, particularly clavulanic acid, a potent beta-lactamase inhibitor. Patent CN1041933C introduces a groundbreaking advancement in this domain by disclosing novel tertiary-tertiary diamine salts of clavulanic acid. These new chemical entities address long-standing challenges associated with the isolation and purification of clavulanic acid, which is notoriously unstable and prone to degradation. By transforming the free acid into specific diamine salts, manufacturers can achieve significantly improved physicochemical properties, facilitating easier handling, storage, and subsequent conversion into final drug products. This technological leap provides a reliable pharmaceutical intermediate supplier with the tools to deliver high-purity materials essential for modern antibiotic formulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the isolation of clavulanic acid has relied heavily on the formation of salts with primary amines or simple tertiary amines like tert-butylamine, as documented in earlier patents such as US 4650795 and European Patent 26044. While these methods provided a means to isolate the acid, they were fraught with significant operational deficiencies. Primary amine salts often exhibited poor crystallinity, tending to form viscous oils or amorphous solids that were extremely difficult to purify further. This lack of defined crystal structure complicated filtration and drying processes, leading to substantial product loss and inconsistent quality. Furthermore, the hygroscopic nature of many conventional salts made them susceptible to moisture uptake, accelerating degradation pathways that compromised the potency of the active pharmaceutical ingredient before it could even be formulated.
The Novel Approach
In stark contrast, the methodology outlined in CN1041933C utilizes specific tertiary-tertiary diamines to generate salts with exceptional crystalline characteristics. The invention specifically highlights salts such as N,N,N',N'-tetramethyl-1,2-diaminoethane clavulanate, which precipitate as large, well-defined crystals rather than intractable oils. This physical transformation is not merely cosmetic; it represents a fundamental improvement in processability. The ability to isolate the intermediate as a free-flowing crystalline solid allows for efficient washing to remove impurities, resulting in a much higher purity profile. This novel approach effectively bypasses the bottlenecks of traditional isolation techniques, offering a streamlined pathway for cost reduction in pharmaceutical intermediate manufacturing.
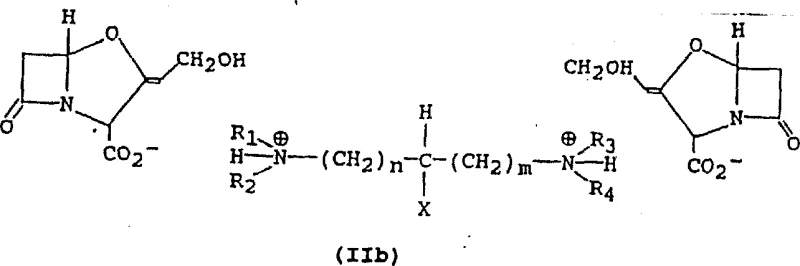
Mechanistic Insights into Diamine Salt Formation and pH Control
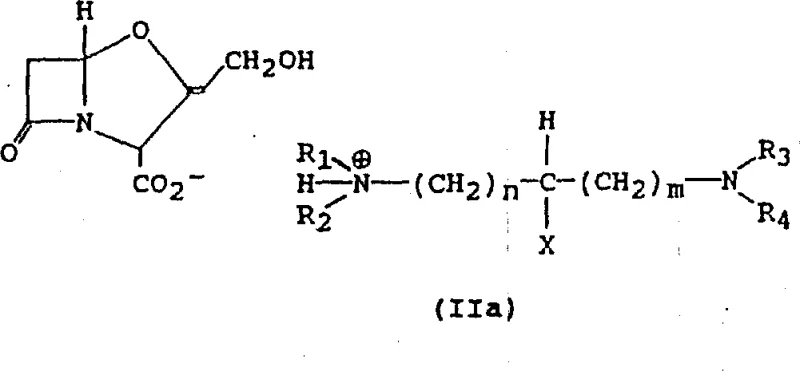
The core of this innovation lies in the precise control of protonation states of the diamine ligand relative to the carboxylic acid group of clavulanic acid. The patent elucidates that the nature of the salt formed—whether a monosalt or a disalt—is governed by the stoichiometry and the pH of the reaction medium. When the concentration of the diamine is high relative to the acid, or at higher pH values, the equilibrium shifts towards the formation of monoprotonated diamine species. These monoprotonated species interact with the clavulanate anion to form the stable monosalt structure depicted in Formula IIa. Conversely, under conditions of lower pH or lower diamine concentration, diprotonated species dominate, leading to the formation of disalts as shown in Formula IIb.
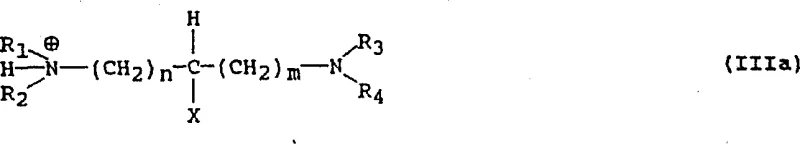
This mechanistic understanding allows for tunable process parameters. By manipulating the pH, typically maintaining it between 8 and 9 for monosalt formation using bases like TMEDA, manufacturers can selectively precipitate the desired crystalline phase. The structural integrity of these salts is reinforced by strong hydrogen bonding networks within the crystal lattice, as evidenced by X-ray diffraction data provided in the patent examples. This robust lattice energy is what prevents the formation of oils and ensures that the material remains stable during downstream processing. For R&D teams, this implies that impurity profiles can be tightly controlled by managing the protonation equilibrium, ensuring that the final API meets stringent regulatory specifications without the need for extensive chromatographic purification.
How to Synthesize Clavulanic Acid Diamine Salts Efficiently
The synthesis of these advanced intermediates involves a straightforward yet precise salting-out procedure performed in organic media. The process begins with the extraction of clavulanic acid into a suitable non-hydroxylic solvent, followed by the controlled addition of the diamine base. The detailed standardized synthesis steps for implementing this technology in a GMP environment are provided in the guide below.
- Prepare a solution of impure clavulanic acid in a non-hydroxylic organic solvent such as ethyl acetate or acetone.
- Adjust the pH of the mixture by adding a specific tertiary-tertiary diamine, controlling the stoichiometry to favor either monosalt or disalt formation.
- Induce crystallization by cooling or solvent adjustment, then separate the pure crystalline salt via filtration and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this diamine salt technology translates into tangible operational efficiencies and risk mitigation. The shift from oily, unstable intermediates to stable crystalline solids fundamentally alters the logistics of production. Crystalline materials are inherently easier to transport, store, and handle in bulk quantities compared to viscous oils, which often require specialized heating or pumping equipment. This physical stability reduces the risk of degradation during warehousing, thereby extending the shelf-life of the intermediate and minimizing waste due to spoilage. Consequently, this leads to substantial cost savings by improving overall material throughput and reducing the frequency of batch failures.
- Cost Reduction in Manufacturing: The elimination of oil formation removes the need for complex and expensive purification steps such as preparative chromatography or repeated extractions that are often necessary to recover product from emulsions. The high yield of crystalline precipitation directly improves the mass balance of the process. Furthermore, the ability to wash the crystals effectively removes residual solvents and impurities in a single unit operation, significantly lowering utility consumption and labor costs associated with multi-step purification protocols.
- Enhanced Supply Chain Reliability: The robustness of the crystalline diamine salts ensures consistent quality across different production batches, which is critical for maintaining uninterrupted supply to downstream API manufacturers. The use of readily available diamines like TMEDA ensures that raw material sourcing is not a bottleneck. This reliability allows for better inventory planning and reduces the safety stock requirements, optimizing working capital for the organization.
- Scalability and Environmental Compliance: The process operates effectively in common organic solvents like ethyl acetate and acetone, which are widely accepted in industrial settings and have established recovery protocols. The simplicity of the filtration and drying steps facilitates easy scale-up from pilot plants to commercial-scale reactors without significant re-engineering. Additionally, the high purity of the isolated salt reduces the burden on wastewater treatment systems by minimizing the discharge of organic impurities, aligning with increasingly strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of the clavulanic acid diamine salt technology described in the patent literature. These insights are derived directly from the experimental data and claims to assist technical teams in evaluating feasibility.
Q: Why are tertiary-tertiary diamine salts preferred over primary amine salts for clavulanic acid?
A: Tertiary-tertiary diamine salts, such as those formed with TMEDA, exhibit superior crystallinity and stability compared to primary amine salts, which often form difficult-to-handle oils or amorphous solids.
Q: How does pH control influence the formation of monosalts versus disalts?
A: Higher pH levels favor the existence of monoprotonated diamines, leading to the precipitation of monosalts, whereas lower pH levels promote diprotonated species and disalt formation.
Q: What solvents are compatible with this salt formation process?
A: The process utilizes non-hydroxylic organic solvents including tetrahydrofuran, dioxane, ethyl acetate, methyl acetate, acetone, and methyl ethyl ketone to ensure optimal solubility and crystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Clavulanic Acid Diamine Salt Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving antibiotics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of clavulanic acid intermediate performs consistently in your downstream synthesis.
We invite global partners to collaborate with us to leverage this advanced purification technology for their supply chains. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production volumes. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized diamine salt process can enhance your operational efficiency and product quality.
