Scalable Manufacturing of AQ4N Prodrug via Low-Temperature Oxidation and Purification
The pharmaceutical industry continuously seeks robust manufacturing routes for complex oncology prodrugs, and Patent CN1930117A represents a significant advancement in the synthesis of AQ4N, a non-toxic precursor to the cytotoxic agent AQ4. This compound functions as a bioreductive prodrug, remaining inert until metabolized within the hypoxic environment of solid tumors, where it releases the active DNA-intercalating agent. However, historical manufacturing methods described in prior art such as WO 00/05194 have struggled with critical impurity profiles, specifically the formation of AQMN, a degradation product that poses toxicity risks. The disclosed innovation addresses these challenges by introducing a controlled low-temperature oxidation protocol and improved intermediate purification strategies. By shifting from hazardous solid-state reactions to solvent-mediated processes, this technology offers a pathway to high-purity pharmaceutical intermediates suitable for clinical scale-up. For R&D directors and procurement specialists, understanding these mechanistic improvements is vital for securing a reliable supply chain of high-quality API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
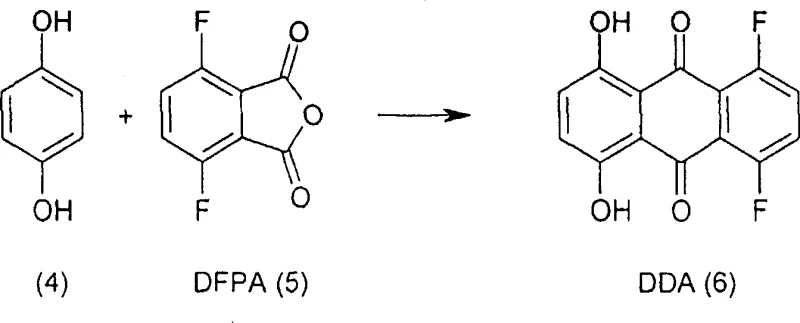
Historically, the synthesis of the key intermediate DDA (1,4-difluoro-5,8-dihydroxy-anthracene) relied on solid-state Friedel-Crafts acylation, a process fraught with engineering and safety challenges for commercial production. In these legacy methods, powdered mixtures of para-hydroquinone, 3,6-difluorophthalic anhydride (DFPA), and aluminum chloride were heated to extreme temperatures reaching 220°C. Such high-temperature solid-phase reactions create severe heat transfer limitations, making it difficult to control exotherms and leading to inconsistent reaction kinetics across large batches. Furthermore, the harsh conditions promote the formation of intractable aluminum-containing by-products that are notoriously difficult to remove from the crude organic matrix. The presence of residual aluminum not only complicates downstream processing but also introduces potential cell toxicants that must be rigorously excluded from pharmaceutical products. Additionally, the subsequent conversion of DDA to AQ4 in prior art often resulted in medium yields of approximately 40%, indicating significant material loss and inefficiency in atom economy.
The Novel Approach
The innovative process detailed in CN1930117A fundamentally re-engineers the synthesis by introducing a solvent-based system that dramatically improves controllability and purity. Instead of heating solid powders, the Friedel-Crafts acylation is conducted in a high-boiling solvent such as tetramethylene sulfone (sulfolane) at reduced temperatures between 155°C and 180°C. The presence of the solvent allows for efficient stirring and uniform heat distribution, which significantly lowers the rate of gas generation and mitigates safety risks associated with thermal runaways. Crucially, this liquid-phase approach enables a novel purification step where the crude DDA is treated with aqueous hydrochloric acid to form a slurry. This acid slurrying effectively solubilizes and removes aluminum impurities, reducing content from thousands of ppm to acceptable levels without requiring complex chromatographic separation. This shift from solid-state to solution chemistry exemplifies a strategic move towards manufacturability, ensuring that the process is robust enough for multi-kilogram to ton-scale production while maintaining stringent quality standards.
Mechanistic Insights into Low-Temperature Oxidation and Impurity Control
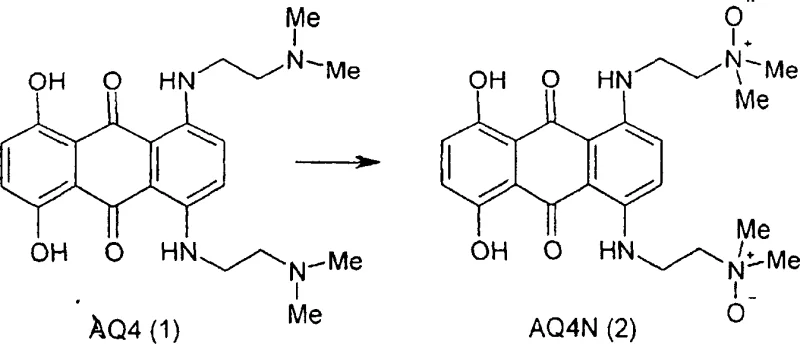
The conversion of the active compound AQ4 to its N-oxide prodrug form, AQ4N, is the most critical step in the synthesis, as it determines the final impurity profile and stability of the drug substance. Conventional oxidation methods often utilize reagents like Davis reagent (2-benzenesulfonyl-3-phenyl-oxaziridine) at room temperature, which lack the specificity required to prevent over-oxidation or degradation. The primary mechanistic challenge is avoiding the formation of AQMN (1-amino-4-{[2-(dimethylamino) ethyl] amino}-5,8-dihydroxyanthraquinone), a degradation product formed when AQ4N breaks down under severe or uncontrolled oxidative conditions. Even under relatively gentle conditions, prior art methods have reported the presence of this undesirable impurity, which compromises the safety profile of the final pharmaceutical product. The degradation pathway is thermally driven, meaning that higher reaction temperatures accelerate the breakdown of the sensitive N-oxide bond, leading to reduced yields and increased purification burdens.
To counteract this, the patented process employs a precise low-temperature oxidation strategy using magnesium monoperphthalate (MMPP) as the oxidant. The reaction is conducted at temperatures strictly not exceeding 10°C, with a preferred operating range around -11°C. At these sub-zero temperatures, the kinetic energy of the molecules is reduced, selectively favoring the formation of the N-oxide bond while suppressing the activation energy required for the degradation pathway leading to AQMN. The choice of MMPP is also strategic; it is air-stable and water-soluble, making it safer and easier to handle on a large scale compared to explosive peracids. The solvent system, typically a mixture of methylene dichloride and aliphatic alkyl alcohols like methanol, is optimized to maintain solubility at these low temperatures while facilitating the reaction. This meticulous control over thermal and chemical parameters ensures that the oxidation proceeds cleanly, minimizing the generation of partial oxidation by-products like AQ4M and preserving the integrity of the final AQ4N molecule.
How to Synthesize AQ4N Efficiently
The synthesis of AQ4N requires a disciplined approach to reaction conditions, particularly regarding temperature control and purification sequences to ensure pharmaceutical-grade quality. The process begins with the preparation of the DDA intermediate via solvent-mediated Friedel-Crafts acylation, followed by amination to form AQ4, and concludes with the critical low-temperature oxidation step. Each stage incorporates specific work-up procedures, such as acid slurrying for metal removal and activated carbon treatment for decolorization, which are essential for meeting regulatory purity specifications. Operators must adhere strictly to the thermal profiles outlined in the patent, especially during the oxidation phase where deviations above 10°C can lead to significant impurity spikes. The following guide summarizes the standardized operational framework derived from the patent examples, providing a roadmap for technical teams to replicate this high-efficiency route.
- Perform Friedel-Crafts acylation of para-hydroquinone with DFPA in sulfolane solvent at 155-180°C, followed by acid slurrying to remove aluminum residues.
- Convert the resulting DDA intermediate to AQ4 using N,N-dimethyl-ethylenediamine, neutralizing acidic by-products with cold ammonium hydroxide.
- Oxidize AQ4 to AQ4N using magnesium monoperphthalate in methanol and methylene dichloride at temperatures strictly below 10°C, preferably around -11°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from legacy solid-state chemistry to this improved solvent-based process offers substantial strategic benefits beyond mere technical elegance. The ability to conduct reactions in a fluid phase significantly enhances process scalability, allowing manufacturers to move from laboratory bench scales to commercial production volumes with greater confidence and reduced risk of batch failure. The elimination of extreme high-temperature solid-state steps reduces the wear and tear on reactor equipment and lowers the energy consumption required for heating, contributing to overall cost reduction in pharmaceutical intermediates manufacturing. Furthermore, the improved impurity profile means that fewer resources are spent on extensive downstream purification, such as multiple recrystallizations or preparative chromatography, which are both time-consuming and costly. This efficiency translates directly into a more reliable supply chain, as the process is less susceptible to the variability that often plagues complex multi-step syntheses.
- Cost Reduction in Manufacturing: The implementation of solvent-based Friedel-Crafts acylation eliminates the need for handling large quantities of solid aluminum chloride at extreme temperatures, which reduces safety containment costs and waste disposal fees associated with hazardous solid residues. By enabling the removal of aluminum impurities through simple acid slurrying rather than complex extraction or chromatography, the process significantly lowers the consumption of solvents and consumables. Additionally, the higher yields achieved in the conversion of DDA to AQ4, improving from approximately 40% in prior art to over 70% in this method, mean that less raw material is required to produce the same amount of final product. These cumulative efficiencies drive down the cost of goods sold (COGS) without compromising on the stringent quality requirements necessary for oncology drugs.
- Enhanced Supply Chain Reliability: Scalability is a primary concern for supply chain leaders, and the shift to a stirred liquid-phase reaction system inherently supports larger batch sizes compared to solid-state mixing. The use of stable oxidants like magnesium monoperphthalate reduces the dependency on hazardous reagents that may have restricted shipping or storage regulations, thereby smoothing out logistics and reducing lead time for high-purity pharmaceutical intermediates. The robustness of the purification steps, including the effective activated carbon treatment, ensures consistent batch-to-batch quality, minimizing the risk of rejected shipments due to out-of-specification impurity levels. This reliability allows pharmaceutical partners to plan their clinical and commercial timelines with greater certainty, knowing that the supply of this critical prodrug intermediate will remain uninterrupted.
- Scalability and Environmental Compliance: Operating at lower temperatures (155-180°C vs 220°C) and utilizing closed solvent systems significantly reduces the emission of volatile organic compounds and particulate matter, aligning the process with modern environmental health and safety (EHS) standards. The ability to recycle solvents like sulfolane and methylene dichloride further minimizes the environmental footprint of the manufacturing process. Moreover, the reduction in aluminum waste and the avoidance of heavy metal catalysts in the oxidation step simplify the wastewater treatment requirements. These factors make the facility more compliant with increasingly strict global environmental regulations, ensuring long-term operational sustainability and reducing the risk of regulatory shutdowns or fines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the manufacturing of AQ4N, derived directly from the specific innovations and data presented in the patent literature. These insights are intended to clarify the advantages of this specific synthetic route over traditional methods, focusing on impurity control, scalability, and purification efficiency. Understanding these details is crucial for technical evaluators assessing the feasibility of integrating this intermediate into their broader drug development pipelines.
Q: How does the new process reduce AQMN impurity levels compared to conventional methods?
A: Conventional oxidation methods often operate at room temperature or use harsh reagents like Davis reagent, which promote the degradation of AQ4N into the toxic impurity AQMN. The patented process strictly controls the oxidation temperature to below 10°C, preferably around -11°C, using magnesium monoperphthalate. This低温 condition kinetically favors the formation of the N-oxide while suppressing the degradation pathway that leads to AQMN, ensuring higher pharmaceutical purity.
Q: What are the scalability advantages of using solvent-based Friedel-Crafts acylation over solid-state reactions?
A: Traditional solid-state Friedel-Crafts reactions require heating powdered mixtures to extreme temperatures like 220°C, which creates significant heat transfer challenges and safety risks on a large scale. The improved method utilizes a solvent system, specifically tetramethylene sulfone (sulfolane), allowing the reaction to proceed at lower temperatures between 155°C and 180°C under stirring. This fluid phase ensures uniform heat distribution, reduces gas generation rates, and facilitates the subsequent removal of aluminum catalyst residues through acid slurrying.
Q: Why is activated carbon treatment preferred for purifying AQ4N salts?
A: Colored impurities and trace organic by-products are common in anthraquinone synthesis. While recrystallization is a standard purification technique, it can lead to significant yield loss. The patent describes a specific purification step where the AQ4N salt solution is treated with activated carbon. This method effectively adsorbs colored foreign pigments and trace contaminants without the need for aggressive recrystallization cycles, thereby maintaining high mass recovery while achieving the stringent purity specifications required for clinical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable AQ4N Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of oncology therapeutics depends on the availability of high-quality, consistently manufactured intermediates like AQ4N. As a specialized CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our facilities are equipped with rigorous QC labs capable of detecting trace impurities at ppm levels, guaranteeing that every batch meets stringent purity specifications required for clinical trials and market approval. We understand the critical nature of hypoxia-activated prodrugs and are committed to maintaining the integrity of the low-temperature oxidation process to prevent degradation and ensure maximum therapeutic efficacy.
We invite you to collaborate with our technical procurement team to discuss how our advanced manufacturing capabilities can support your project goals. By partnering with us, you gain access to a Customized Cost-Saving Analysis that evaluates how our optimized synthesis route can reduce your overall development costs. We encourage you to contact us today to request specific COA data and route feasibility assessments tailored to your unique requirements, ensuring a seamless transition from development to commercial supply.
