Revolutionizing Enamine and Indole Production: A Noble-Metal-Free One-Pot Strategy for Industrial Scale-Up
Revolutionizing Enamine and Indole Production: A Noble-Metal-Free One-Pot Strategy for Industrial Scale-Up
The landscape of organic synthesis for high-value nitrogen-containing heterocycles is undergoing a significant transformation, driven by the urgent need for greener, more cost-effective manufacturing processes. A pivotal advancement in this domain is detailed in patent CN114276286A, which discloses a groundbreaking method for preparing N,N-disubstituted-alpha,beta-unsaturated enamines directly from aromatic ketones and secondary nitrogen compounds. This technology represents a paradigm shift for reliable pharmaceutical intermediate suppliers and chemical manufacturers, as it replaces complex, multi-step transition metal catalysis with a simple, base-mediated one-pot reaction. By leveraging dimethyl sulfoxide (DMSO) as both the reaction medium and a crucial carbon source, this invention addresses long-standing challenges in atom economy and operational simplicity. For R&D directors and procurement strategists alike, understanding the implications of this patent is critical for optimizing supply chains and reducing the total cost of ownership for complex fine chemicals.
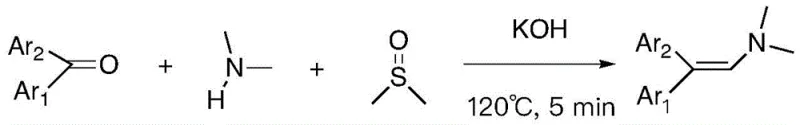
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the N,N-disubstituted-alpha,beta-unsaturated enamine motif has been fraught with synthetic inefficiencies and economic burdens. Traditional pathways often rely on the coupling of terminal alkynes with secondary amines, a process that necessitates the use of dual gold and copper catalytic systems. As illustrated in prior art, this approach frequently results in poor regioselectivity, yielding difficult-to-separate mixtures of N,N-disubstituted-alpha,beta-unsaturated enamines and their beta,beta-unsaturated isomers. Furthermore, alternative strategies involving the reaction of terminal haloalkenes with amines demand the presence of palladium catalysts and specialized halogenated olefin starting materials, which are often unstable and commercially scarce. Another prevalent method utilizes methyl aryl ketone p-toluenesulfonyl hydrazides; however, these precursors are notoriously difficult to source and handle, typically delivering yields below 40 percent. These legacy methods impose severe constraints on cost reduction in API manufacturing due to the high price of noble metals, the complexity of purification, and the instability of raw materials.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the methodology described in CN114276286A offers a streamlined, atom-economical solution that bypasses the need for precious metals entirely. The core innovation lies in the utilization of readily available aromatic ketones and secondary amines reacting in a DMSO solution under basic conditions and an atmospheric oxygen environment. This process effectively converts the carbon-oxygen double bond of the starting ketone into a carbon-carbon double bond, while simultaneously incorporating a carbon atom from the DMSO solvent to extend the chain. The result is a high-yield formation of the target enamine structure with exceptional purity profiles. This approach not only simplifies the reaction setup to a single pot but also utilizes commodity chemicals, thereby enhancing the commercial scale-up of complex polymer additives and pharmaceutical intermediates. The ability to achieve high conversion rates within minutes at moderate temperatures (e.g., 120°C) underscores the robustness of this chemistry for large-scale industrial applications.
Mechanistic Insights into Base-Mediated Oxidative Coupling
To fully appreciate the value of this technology for process chemists, one must delve into the unique mechanistic pathway that enables this transformation without transition metals. The reaction initiates with an Aldol-type condensation between the aromatic ketone and DMSO under alkaline conditions, generating a key 1-methylsulfinyl intermediate. Crucially, this intermediate is then oxidized by molecular oxygen from the air to form a reactive sulfone species. This oxidation step is vital, as it activates the adjacent carbon for nucleophilic attack. Subsequently, the secondary amine acts as a nucleophile, attacking the activated double bond to form a transient adduct where the nitrogen and the methylsulfonyl group are attached to the same carbon. The final step involves the base-mediated elimination of the methylsulfonyl group, which serves as an excellent leaving group, resulting in the formation of the stable N,N-disubstituted-alpha,beta-unsaturated enamine. This elegant cascade ensures that the carbon skeleton is expanded by exactly one carbon atom derived from the solvent.
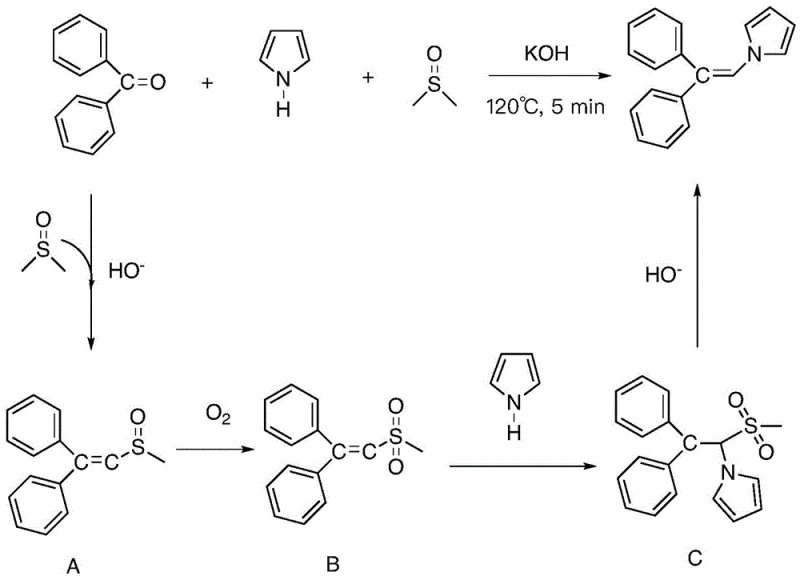
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed routes. Since no heavy metals are introduced, the risk of metal contamination in the final active pharmaceutical ingredient (API) is eliminated, removing the need for expensive scavenging resins or complex recrystallization steps designed to meet strict ppm limits. Furthermore, the reaction demonstrates high chemoselectivity; experiments indicate that aldehydes or primary/tertiary amines do not interfere or produce side products under these specific conditions, and ketones lacking alpha-hydrogens do not undergo self-condensation. This specificity ensures a cleaner crude reaction profile, facilitating easier downstream processing. Additionally, the versatility of this mechanism extends to the synthesis of indole derivatives when 2-acylaniline substrates are employed, where the intramolecular amino group attacks the intermediate to close the ring, showcasing the broad utility of this oxidative coupling strategy for generating diverse heterocyclic scaffolds.
How to Synthesize N,N-Disubstituted-Alpha,Beta-Unsaturated Enamine Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific thermal and stoichiometric parameters to maximize yield and safety. The process is remarkably robust, tolerating a variety of substituents on the aromatic rings of the ketone, including alkyl, halogen, and alkoxy groups. The standard protocol involves heating the reaction mixture to 120°C for a brief period, typically around 5 minutes, which is sufficient to drive the reaction to completion. This short residence time is a significant advantage for continuous flow processing or high-throughput batch manufacturing. For detailed operational parameters, stoichiometry ratios, and workup procedures, please refer to the standardized guide below.
- Combine aromatic ketone, secondary amine compound, and potassium hydroxide (KOH) in a reaction vessel containing dimethyl sulfoxide (DMSO) as the solvent.
- Heat the reaction mixture to 120°C under an air atmosphere and maintain stirring for approximately 5 minutes to facilitate the oxidative coupling and elimination sequence.
- Upon completion, cool the mixture, perform liquid-liquid extraction using ethyl acetate and saturated brine, dry the organic layer, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this DMSO-based synthesis route translates into tangible strategic benefits that go beyond simple yield improvements. The primary driver of value is the complete elimination of noble metal catalysts such as palladium, gold, and copper, which are subject to volatile market pricing and supply chain disruptions. By replacing these expensive inputs with inexpensive inorganic bases like potassium hydroxide, manufacturers can achieve substantial cost savings in raw material procurement. Moreover, the removal of heavy metals from the process stream significantly reduces the environmental burden associated with waste treatment and regulatory compliance, aligning with modern green chemistry initiatives. The use of DMSO, a common and low-cost industrial solvent, further enhances the economic viability of the process, making it an ideal candidate for reducing lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the catalyst system. Traditional methods relying on palladium or gold complexes incur high licensing and material costs, alongside the downstream expense of metal removal to meet regulatory standards (often <10 ppm). This novel base-catalyzed approach eradicates these costs entirely. Additionally, the reaction utilizes DMSO as a reactant, meaning the solvent consumption is integrated into the product structure, improving atom economy and reducing the volume of waste solvent that requires disposal or recycling. The simplicity of the workup, involving standard extraction and chromatography, further lowers operational expenditures compared to the complex purification trains needed for metal-catalyzed byproducts.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity feedstocks. Aromatic ketones and secondary amines are widely available bulk chemicals with established global supply networks, unlike the specialized terminal haloalkenes or unstable hydrazide precursors required by older methods. This availability ensures consistent production scheduling and mitigates the risk of raw material shortages. Furthermore, the reaction operates under atmospheric conditions (air) rather than requiring inert gas blankets or high-pressure hydrogenation equipment, simplifying the infrastructure requirements for manufacturing facilities and allowing for more flexible production planning across different sites.
- Scalability and Environmental Compliance: The one-pot nature of this synthesis significantly reduces the number of unit operations, minimizing the potential for yield loss during intermediate isolation and transfer. The short reaction time (minutes rather than hours or days) allows for higher throughput in existing reactor vessels, effectively increasing capacity without capital investment. From an environmental standpoint, the process avoids toxic reagents and generates less hazardous waste. The absence of heavy metal residues simplifies effluent treatment, ensuring compliance with increasingly stringent environmental regulations regarding industrial discharge. This green profile makes the technology highly attractive for companies aiming to reduce their carbon footprint and improve their sustainability metrics.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity for technical teams evaluating this route for potential integration into their manufacturing portfolios. Understanding these nuances is essential for accurate process modeling and feasibility assessments.
Q: What is the source of the additional carbon atom in the enamine product?
A: Unlike traditional methods where the carbon skeleton remains unchanged, this novel process utilizes dimethyl sulfoxide (DMSO) not just as a solvent but as a reactant. The methyl group from DMSO provides the extra carbon atom that extends the ketone structure into the alpha,beta-unsaturated system.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, this method eliminates the need for costly noble metals such as Palladium (Pd), Gold (Au), or Copper (Cu). The reaction is driven solely by an inorganic base, typically potassium hydroxide (KOH), under aerobic conditions, which drastically lowers catalyst costs and simplifies metal removal protocols.
Q: Can this methodology be applied to synthesize indole derivatives?
A: Yes, the technology is highly versatile. By utilizing 2-acylaniline derivatives (where the ketone and amine functionalities are within the same molecule), the reaction undergoes an intramolecular cyclization to efficiently produce substituted indole derivatives with high yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N-Disubstituted-Alpha,Beta-Unsaturated Enamine Supplier
As the global demand for complex nitrogenous heterocycles continues to rise, partnering with a technically proficient CDMO is essential for navigating the transition from bench-scale discovery to commercial production. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly through development phases. Our state-of-the-art facilities are equipped to handle the specific thermal and solvent requirements of this DMSO-based chemistry, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest industry standards. We understand that consistency and quality are non-negotiable in the pharmaceutical supply chain, and we are committed to delivering excellence.
We invite you to leverage our technical expertise to optimize your synthesis of enamines and indole derivatives. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you unlock the full potential of this innovative chemistry, driving efficiency and value for your organization.
