Advanced Synthesis of Heteroaryloxy-Beta-Carbolines for CNS Pharmaceutical Applications
The pharmaceutical landscape for central nervous system (CNS) therapeutics is constantly evolving, driven by the need for compounds with higher specificity and reduced side effect profiles. Patent CN1039906C discloses a sophisticated preparation method for heteroaryloxy-beta-carbolines, a class of compounds demonstrating significant potential as anxiolytic and anticonvulsant agents. Unlike earlier generations of benzodiazepine receptor ligands, these novel structures are engineered to maintain therapeutic efficacy while minimizing ataxia and muscle relaxation, addressing a critical unmet need in neuropharmacology. The synthetic routes detailed in this intellectual property leverage precise etherification and cyclization strategies to construct complex heterocyclic systems with high regioselectivity. For R&D teams evaluating new chemical entities, understanding the underlying process chemistry is vital for assessing feasibility and scalability. This report analyzes the technical merits of this patent, highlighting how its methodologies can be adapted for commercial manufacturing to support the development of next-generation CNS medications. 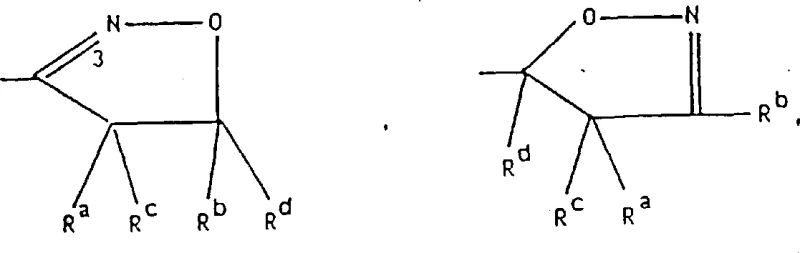
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of beta-carboline derivatives often encounters significant hurdles regarding regioselectivity and functional group tolerance during the substitution of the core scaffold. Conventional etherification protocols frequently require harsh reaction conditions that can lead to the degradation of sensitive ester or ketone functionalities located at the 3-position of the carboline ring. Furthermore, standard methods may struggle to introduce bulky heteroaryl groups at the 5 or 6 positions without generating substantial amounts of regioisomeric impurities that are difficult to separate. The reliance on non-specific catalysts or excessive temperatures in older processes often results in lower overall yields and increased waste generation, which poses challenges for both cost efficiency and environmental compliance. These limitations can severely impact the timeline for process development, as extensive optimization is required to achieve pharmaceutical-grade purity. Consequently, many promising candidates fail to advance due to the inability to establish a robust and scalable manufacturing process that meets strict regulatory standards for impurity control.
The Novel Approach
The methodology presented in CN1039906C offers a transformative solution by utilizing specific polar aprotic solvents and controlled basic conditions to facilitate the nucleophilic substitution of hydroxy-beta-carbolines. This approach allows for the efficient coupling of diverse heteroaryl halides, such as chloroisoquinolines and pyrimidines, with the beta-carboline core under relatively mild thermal conditions. By optimizing the stoichiometry of the base and the addition rate of the electrophile, the process minimizes side reactions and ensures high conversion rates even with sterically hindered substrates. The patent also describes versatile strategies for modifying the 3-position substituent, enabling the synthesis of esters, ketones, and complex heterocycles like isoxazoles through distinct, well-defined pathways. This modularity provides a significant advantage for medicinal chemists seeking to explore structure-activity relationships without being constrained by synthetic bottlenecks. 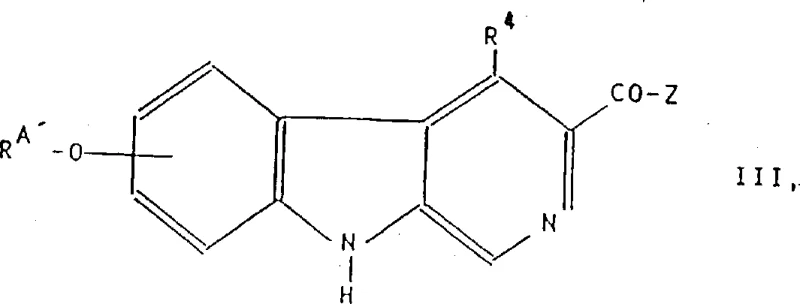
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this synthetic strategy relies on the precise manipulation of electron density within the beta-carboline system to enable selective functionalization. In the etherification step, the deprotonation of the phenolic hydroxyl group generates a highly nucleophilic phenoxide species that attacks the electron-deficient carbon of the heteroaryl halide. The choice of solvent, such as dimethylsulfoxide or dimethylformamide, plays a crucial role in stabilizing the transition state and solvating the cationic counterions, thereby enhancing the reaction kinetics. For the formation of ketone derivatives, the process employs organolithium reagents at cryogenic temperatures to prevent over-addition or attack on other sensitive sites within the molecule. This level of control is essential for maintaining the integrity of the heteroaryloxy linkage while introducing the desired carbonyl functionality. The subsequent cyclization reactions, such as the formation of isoxazoles from nitrile oxides, proceed through concerted mechanisms that are highly dependent on the electronic nature of the dipolarophile. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters for optimal performance and reproducibility.
Impurity control is another critical aspect addressed by the mechanistic understanding of these pathways. The patent highlights the importance of protecting group strategies, particularly at the 9-position of the beta-carboline ring, to prevent unwanted side reactions during lithiation or cyclization steps. By temporarily masking reactive sites, the synthesis ensures that transformations occur exclusively at the intended locations, significantly simplifying the purification process. The removal of these protecting groups is designed to be orthogonal, meaning it can be achieved without affecting the newly formed heteroaryl ether or the 3-position substituent. This strategic use of protection and deprotection sequences minimizes the formation of structurally related impurities that could complicate regulatory filing. Furthermore, the described work-up procedures, involving pH adjustments and specific solvent extractions, are tailored to remove inorganic salts and organic by-products efficiently. This comprehensive approach to impurity management ensures that the final active pharmaceutical ingredient meets the stringent quality requirements necessary for clinical applications.
How to Synthesize Heteroaryloxy-Beta-Carboline Efficiently
Implementing this synthesis requires a systematic approach to reagent preparation and reaction monitoring to ensure consistent quality and yield. The process begins with the careful drying of solvents and reagents to prevent moisture-induced side reactions, particularly during the organolithium steps where water can act as a proton source and quench the reactive species. Operators must adhere to strict temperature controls, especially during the addition of electrophiles and the subsequent heating phases, to avoid thermal runaways or decomposition of intermediates. The patent outlines specific protocols for quenching reactions and isolating products, emphasizing the use of chromatography and recrystallization to achieve high purity. Detailed standard operating procedures should be developed based on these guidelines to ensure that the transfer from laboratory to pilot plant is seamless.
- Prepare the beta-carboline core by reacting 6-hydroxy-beta-carboline-3-carboxylic acid esters with heteroaryl halides under basic conditions in polar solvents.
- Introduce ketone substituents at the 3-position by reacting ester derivatives with organolithium compounds at low temperatures followed by hydrolysis.
- Form isoxazole or oxadiazole rings via cycloaddition of nitrile oxides or reaction with acid anhydrides to complete the heteroaryloxy structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the synthetic routes described in this patent offer substantial opportunities for cost optimization and supply chain stabilization. The starting materials, including various heteroaryl halides and beta-carboline precursors, are derived from readily available chemical feedstocks that are produced by multiple suppliers globally. This diversity in the supply base reduces the risk of shortages and provides leverage in price negotiations, ensuring a steady flow of raw materials for continuous manufacturing operations. The elimination of exotic or highly specialized catalysts further simplifies the sourcing process, as standard inorganic bases and common organic solvents can be utilized throughout the synthesis. This reliance on commodity chemicals significantly lowers the overall material costs and reduces the complexity of inventory management. Additionally, the robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, enhancing operational reliability.
- Cost Reduction in Manufacturing: The streamlined synthetic pathway eliminates the need for expensive transition metal catalysts and complex purification steps often associated with traditional methods. By utilizing direct etherification and efficient cyclization reactions, the process reduces the number of unit operations required to produce the final intermediate. This simplification translates directly into lower energy consumption and reduced labor costs per kilogram of product. Furthermore, the high yields achieved through optimized reaction conditions minimize the waste of valuable starting materials, contributing to a more sustainable and cost-effective production model. The ability to recycle solvents and recover by-products adds another layer of economic benefit, making the overall process highly competitive in the global market.
- Enhanced Supply Chain Reliability: The use of stable intermediates and robust reaction protocols ensures that production schedules can be maintained with minimal disruption. Unlike processes that rely on unstable reagents or sensitive conditions, this methodology allows for longer campaign runs and easier scale-up without compromising product quality. The compatibility with standard manufacturing equipment means that production can be easily shifted between different facilities if necessary, providing flexibility in response to market demands. This resilience is crucial for maintaining the continuity of supply for critical pharmaceutical ingredients, especially in times of global logistical challenges. The predictable nature of the synthesis also facilitates better planning and forecasting for procurement teams.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from laboratory glassware to industrial reactors. The absence of hazardous reagents and the use of environmentally benign solvents align with modern green chemistry principles, reducing the environmental footprint of the manufacturing process. Waste streams are easier to treat due to the simpler chemical composition, lowering the costs associated with environmental compliance and disposal. This alignment with regulatory standards not only mitigates risk but also enhances the corporate social responsibility profile of the manufacturing operation. The efficient use of resources and minimization of waste contribute to a more sustainable supply chain that meets the evolving expectations of stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these specialized intermediates. The answers are derived from the detailed experimental data and process descriptions found within the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these aspects is crucial for making informed decisions about integrating this technology into existing production pipelines.
Q: What are the primary therapeutic applications of heteroaryloxy-beta-carbolines?
A: These compounds exhibit high affinity for benzodiazepine receptors, making them potent candidates for anxiolytic and anticonvulsant medications without significant muscle relaxant side effects.
Q: How does the etherification process improve yield compared to traditional methods?
A: The use of specific polar solvents like DMSO and controlled temperature profiles minimizes side reactions and degradation, leading to cleaner reaction profiles and easier purification.
Q: Are the starting materials for this synthesis commercially available?
A: Yes, key precursors such as chloroisoquinolines and beta-carboline esters are accessible through standard chemical supply chains, ensuring reliable production continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Heteroaryloxy-Beta-Carboline Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, offering unparalleled expertise in translating complex patent methodologies into commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from development to market. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of heteroaryloxy-beta-carboline meets the highest industry standards. Our commitment to quality and reliability makes us the ideal partner for pharmaceutical companies seeking a secure and efficient supply of critical intermediates. We understand the critical nature of CNS drug development and are dedicated to supporting your success through technical excellence and operational agility.
We invite you to engage with our technical procurement team to discuss how we can optimize your supply chain for these valuable compounds. Request a Customized Cost-Saving Analysis to understand the potential economic benefits of partnering with us for your manufacturing needs. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your specific requirements. By collaborating with NINGBO INNO PHARMCHEM, you gain access to a wealth of chemical knowledge and production capacity that can accelerate your drug development timeline. Contact us today to initiate a conversation about your next project.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
