Advanced Multi-Unit Red Light TADF Materials for High-Performance OLED Device Manufacturing
Advanced Multi-Unit Red Light TADF Materials for High-Performance OLED Device Manufacturing
The rapid evolution of the organic electroluminescence industry demands materials that transcend the limitations of traditional fluorescent and phosphorescent emitters, particularly in the challenging red spectrum. Patent CN113816911A introduces a groundbreaking class of red light materials featuring multiple light-emitting units, specifically designed to address the critical scarcity of high-efficiency, low roll-off red emitters. This technology leverages a sophisticated molecular architecture where aromatic amine derivatives serve as donor units and cyano-substituted nitrogen heterocycles act as acceptor units, all integrated onto a central benzene core. By strategically arranging two or three of these donor-acceptor units, the material achieves superior molar absorptivity and fluorescence quantum efficiency, positioning it as a transformative solution for full-color displays and white light illumination applications.
The significance of this innovation lies in its ability to harness Thermally Activated Delayed Fluorescence (TADF) mechanisms without the reliance on precious heavy metals. Traditional phosphorescent materials, while efficient, suffer from high costs due to iridium and platinum content, alongside stability issues in deep blue and red regions. In contrast, this patent outlines a purely organic approach that maintains high exciton utilization rates through effective intersystem crossing, offering a sustainable and economically viable pathway for next-generation OLED manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of red organic light-emitting diodes has been hindered by the energy gap law, which dictates that non-radiative transition rates increase rapidly as the emission wavelength extends into the red region. Conventional fluorescent dyes are fundamentally limited by spin statistics, utilizing only 25% of singlet excitons, which caps their internal quantum efficiency. While phosphorescent materials utilizing heavy metal complexes like iridium or platinum can theoretically reach 100% efficiency by harvesting triplet excitons, they introduce severe economic and technical bottlenecks. The inclusion of scarce heavy metals drastically inflates raw material costs, and the chemical stability of these complexes, especially in red and deep-red emissions, often proves inadequate for long-term device operation. Furthermore, these heavy metal-based systems frequently exhibit significant efficiency roll-off under high brightness conditions, limiting their utility in high-performance display panels.
The Novel Approach
The methodology disclosed in the patent circumvents these obstacles by employing a multi-unit donor-acceptor strategy that eliminates the need for heavy metals entirely. The core innovation involves constructing a twisted molecular configuration through the combination of specific aromatic amine donors and cyano-substituted nitrogen heterocycle acceptors. This structural twist minimizes the overlap between the highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO), thereby reducing the singlet-triplet energy gap (ΔEST) to facilitate efficient reverse intersystem crossing (RISC). By integrating multiple such emitting units onto a single benzene ring scaffold, the material not only retains the TADF characteristics but also amplifies the molar absorption coefficient. This results in a red light emitter that combines the cost benefits of organic synthesis with the high efficiency previously reserved for metal complexes, effectively solving the problem of efficiency roll-off and stability in red OLEDs.
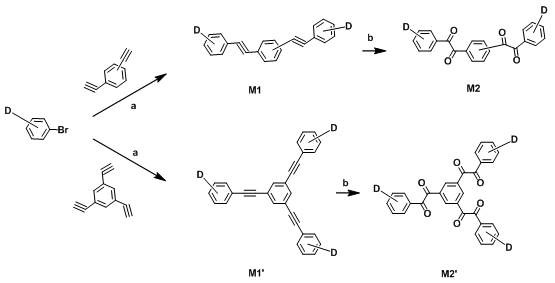
Mechanistic Insights into Multi-Unit TADF Emitter Synthesis
The synthesis of these advanced materials relies on a robust three-stage chemical transformation sequence that ensures high purity and structural integrity. The process initiates with a Sonogashira cross-coupling reaction, where a brominated raw material containing the donor group reacts with an ethynylbenzene derivative. This step, catalyzed by palladium and copper species in a triethylamine solvent under nitrogen reflux, forms the carbon-carbon triple bond linkage essential for extending the conjugation system. Following this, the alkyne groups undergo a controlled oxidation using potassium permanganate in a weakly acidic acetone medium. This critical oxidative cleavage converts the ethynyl groups into vicinal diketones, creating the reactive centers necessary for the final ring-closing step. The precision of this oxidation is vital, as over-oxidation can degrade the delicate donor units, while under-oxidation prevents the subsequent cyclization.
The final stage involves a condensation reaction between the vicinal diketone intermediate and a diamino-nitrile acceptor precursor in acetic acid. This cyclization forms the rigid nitrogen-containing heterocyclic core that defines the acceptor strength of the molecule. For specific variants involving brominated phenanthrene or acenaphthylene derivatives, a subsequent substitution reaction using cuprous cyanide in methyl pyrrolidone replaces bromine atoms with cyano groups, further tuning the electron-withdrawing capability. This multi-step pathway allows for precise modulation of the electronic properties, ensuring that the final product possesses the optimal energy levels for red emission. The rigorous purification protocols, including column chromatography and vacuum sublimation described in the examples, ensure that the final material meets the stringent purity specifications required for commercial electroluminescent devices.
How to Synthesize Multi-Unit Red Light TADF Material Efficiently
The preparation of these high-performance emitters follows a standardized protocol that balances yield optimization with operational simplicity. The process begins with the careful selection of donor precursors, such as 4-bromotriphenylamine or carbazole derivatives, which are coupled with diethynylbenzene isomers to establish the molecular backbone. Subsequent oxidation and cyclization steps are performed under reflux conditions to drive the reactions to completion, followed by meticulous workup procedures involving ice water quenching and solvent extraction. The detailed operational parameters, including specific molar ratios, solvent volumes, and temperature controls, are critical for reproducing the high yields reported in the patent examples, such as the 59.8% yield achieved for Compound 1.
- Perform Sonogashira coupling between brominated donor precursors and ethynylbenzene derivatives using Pd/Cu catalysts in triethylamine.
- Oxidize the resulting alkyne intermediate to a vicinal diketone using potassium permanganate in a weakly acidic acetone solvent.
- Condense the diketone intermediate with diamino-nitrile acceptors in acetic acid under reflux to form the final heterocyclic TADF emitter.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement strategists and supply chain managers, the adoption of this multi-unit red light material offers substantial advantages rooted in raw material accessibility and process economics. The elimination of iridium and platinum from the supply chain removes exposure to the volatile pricing and geopolitical risks associated with precious metal mining. Instead, the synthesis relies on abundant organic feedstocks and standard catalysts like palladium and copper, which are widely available in the global chemical market. This shift significantly reduces the cost of goods sold (COGS) and stabilizes long-term supply contracts, ensuring consistent production schedules without the risk of metal shortages. Furthermore, the synthetic route utilizes common solvents such as triethylamine, acetone, and acetic acid, simplifying waste management and solvent recovery processes compared to specialized organometallic syntheses.
- Cost Reduction in Manufacturing: The removal of expensive heavy metal precursors represents a direct and significant reduction in raw material expenditure. Additionally, the synthetic pathway avoids the need for complex ligand synthesis often required for phosphorescent complexes, streamlining the overall production timeline. The use of standard purification techniques like sublimation, rather than resource-intensive preparative HPLC for every batch, further lowers operational costs. These factors combine to create a highly cost-competitive material suitable for mass-market display applications where margin pressure is intense.
- Enhanced Supply Chain Reliability: By relying on a fully organic synthesis tree, manufacturers can diversify their supplier base for key intermediates, reducing dependency on single-source metal providers. The robustness of the Sonogashira coupling and oxidative cyclization steps ensures high reproducibility across different production scales, minimizing batch-to-batch variability. This reliability translates to shorter lead times for high-purity OLED material delivery, as the production cycle is not bottlenecked by the slow turnover of precious metal refining or complex coordination chemistry steps.
- Scalability and Environmental Compliance: The reaction conditions described, such as refluxing in acetic acid or triethylamine, are readily adaptable to large-scale reactors without requiring exotic high-pressure or cryogenic equipment. The waste streams generated are primarily organic solvents and inorganic salts, which are easier to treat and recycle compared to heavy metal-contaminated waste. This alignment with green chemistry principles facilitates regulatory compliance and reduces the environmental footprint of the manufacturing facility, a key metric for modern sustainable supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this red light TADF technology. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, offering clarity on performance metrics and synthesis feasibility. Understanding these details is crucial for R&D teams evaluating the material for integration into existing device architectures.
Q: What distinguishes this multi-unit red TADF material from traditional phosphorescent emitters?
A: Unlike traditional phosphorescent materials that rely on expensive and scarce heavy metals like iridium or platinum, this invention utilizes purely organic small molecules with a twisted donor-acceptor configuration. This design enables Thermally Activated Delayed Fluorescence (TADF), achieving theoretical 100% internal quantum efficiency without the cost and stability issues associated with heavy metal complexes.
Q: How does the multi-emitting unit structure improve device performance?
A: By introducing two or three donor-acceptor luminescent units onto a single central benzene ring, the material exhibits significantly higher molar absorptivity and fluorescence quantum efficiency compared to single-unit analogues. This structural enhancement leads to more efficient red light emission and reduces efficiency roll-off at high brightness levels.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the preparation method relies on standard organic transformations such as Sonogashira coupling and oxidative cyclization using commercially available reagents like potassium permanganate and cuprous cyanide. The process avoids exotic catalysts and utilizes standard purification techniques like column chromatography and vacuum sublimation, facilitating commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Red Light TADF Material Supplier
As the demand for high-efficiency red emitters grows, partnering with an experienced CDMO becomes essential for translating laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including trace metal analysis and thermal stability testing, which are critical for the longevity of OLED devices. We understand the nuances of handling sensitive organic intermediates and have optimized our workflows to minimize degradation during scale-up.
We invite you to engage with our technical procurement team to discuss how this multi-unit red light material can enhance your product portfolio. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching from phosphorescent to TADF-based systems. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your specific device requirements, ensuring a seamless transition to this next-generation lighting technology.
