Advanced Synthesis of Polyketone Ligands for High-Performance Polymer Manufacturing
The global demand for high-performance green polymers has intensified the search for efficient catalytic systems, particularly for polyketone (POK) production. Patent CN114106048A introduces a groundbreaking synthetic methodology for the critical ligand 3,3-bis-[bis-(2-methoxyphenyl)phosphinomethyl]-1,5-dioxa-spiro[5,5]undecane, which serves as the cornerstone for next-generation palladium-based polymerization catalysts. This technical disclosure represents a significant paradigm shift from traditional multi-step halogenated routes to a streamlined, direct lithiation strategy starting from abundant anisole. For R&D directors and process engineers, this innovation promises not only enhanced reaction kinetics but also a drastic simplification of the purification workflow, addressing long-standing bottlenecks in fine chemical manufacturing. The method leverages precise temperature control and specific depolymerizing agents to achieve exceptional regioselectivity, ensuring that the resulting ligand possesses the structural integrity required for high-turnover catalytic activity in industrial polymer synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of bis-phosphine ligands for polyketone applications has relied heavily on o-bromoanisole as the primary feedstock, necessitating complex Grignard or lithium-halogen exchange reactions that are inherently sensitive to moisture and oxygen. These conventional pathways often require the use of hazardous reagents such as sodium hydride or sodium tert-butoxide in the final coupling stages, introducing severe safety liabilities including pyrophoric risks and difficult exotherm management during scale-up. Furthermore, the reliance on halogenated starting materials creates a substantial burden on waste treatment infrastructure due to the generation of stoichiometric amounts of salt byproducts and halogenated organic residues. From a supply chain perspective, the volatility in the pricing and availability of specialized brominated aromatics can lead to unpredictable production delays, while the multi-solvent systems typically employed complicate solvent recovery and increase the overall carbon footprint of the manufacturing process.
The Novel Approach
The methodology outlined in patent CN114106048A circumvents these challenges by utilizing direct ortho-lithiation of anisole, a cheaper and more accessible commodity chemical, facilitated by n-butyllithium and specific depolymerizing agents like tetramethylethylenediamine (TMEDA). This strategic substitution eliminates the need for pre-functionalized halogenated precursors, thereby reducing raw material costs and simplifying the initial reaction setup. The subsequent reduction step employs trichlorosilane and tertiary amines, a system that offers superior chemoselectivity compared to traditional hydride reductions, minimizing the formation of phosphine oxides and other stubborn impurities. By replacing dangerous sodium hydride with anhydrous solid alkali hydroxides in the final coupling step, the process significantly enhances operational safety and facilitates easier workup procedures, making it highly attractive for large-scale commercial implementation where safety and environmental compliance are paramount concerns for facility managers.
Mechanistic Insights into Lithiation-Phosphorylation and Reduction
The core of this synthetic breakthrough lies in the controlled ortho-lithiation of anisole, where the coordination of n-butyllithium with depolymerizing agents creates a highly reactive superbase capable of selectively deprotonating the aromatic ring adjacent to the methoxy group. This intermediate anisole lithium species then undergoes nucleophilic attack on diethyl phosphite, forming the P-C bond with high fidelity to generate bis(2-methoxyphenyl)phosphine oxide. The precision of this step is critical, as any meta-substitution or over-lithiation would lead to isomeric impurities that are notoriously difficult to separate and can poison the final polymerization catalyst. Following isolation, the phosphine oxide is subjected to a reduction protocol using trichlorosilane in the presence of a tertiary amine, which activates the silicon-hydrogen bond for oxygen abstraction, effectively converting the P=O double bond into a P-H single bond without affecting the sensitive methoxy substituents on the aromatic rings.
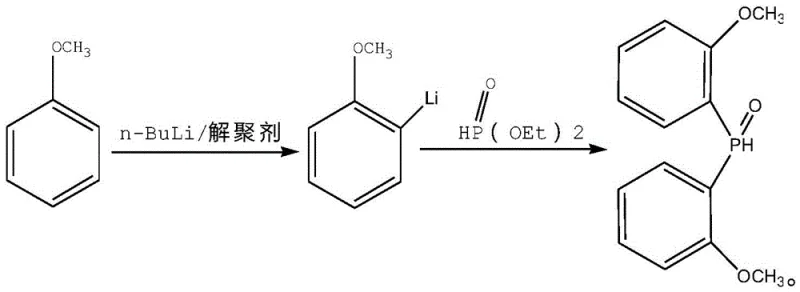
In the final stage, the generated bis(2-methoxyphenyl)phosphine acts as a nucleophile in a substitution reaction with the rigid spiro-bromide intermediate, 5,5-bis(bromomethyl)-1,5-dioxa-spiro[5,5]undecane. The use of anhydrous potassium or sodium hydroxide serves to deprotonate the phosphine, generating a highly nucleophilic phosphide anion in situ that attacks the bromomethyl groups of the spiro-compound. This step constructs the final bidentate ligand architecture, locking the phosphine donors into a specific geometric arrangement that is essential for stabilizing the palladium center during polyketone polymerization. The rigidity of the spiro-cycle prevents unfavorable conformational changes, ensuring consistent catalytic performance, while the mild basic conditions prevent the degradation of the phosphine moiety which is susceptible to oxidation or hydrolysis under harsher alkaline environments.
![Chemical structure of the key intermediate 5,5-bis(bromomethyl)-1,5-dioxa-spiro[5,5]undecane](/insights/img/polyketone-ligand-synthesis-catalyst-supplier-20260315072104-02.png)
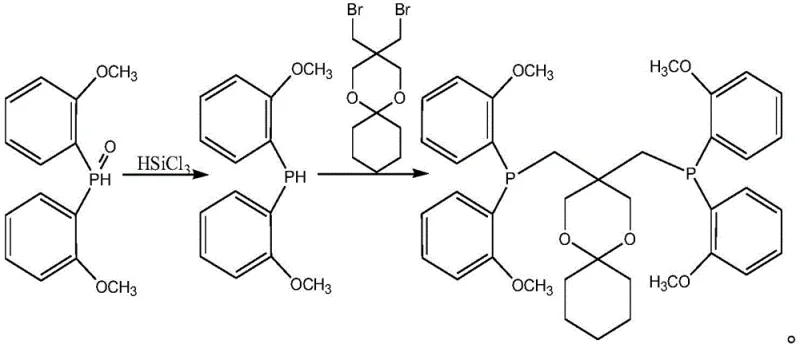
How to Synthesize 3,3-bis-[bis-(2-methoxyphenyl)phosphinomethyl]-1,5-dioxa-spiro[5,5]undecane Efficiently
Implementing this synthesis requires strict adherence to anhydrous and anaerobic conditions, particularly during the lithiation and reduction phases, to prevent the quenching of reactive intermediates by atmospheric moisture or oxygen. The process begins with the preparation of the lithiated anisole solution at cryogenic temperatures, followed by the controlled addition of diethyl phosphite to manage the exotherm and ensure high conversion to the phosphine oxide. Subsequent reduction with trichlorosilane must be monitored closely to avoid over-reduction or side reactions, while the final coupling step benefits from the use of polar aprotic solvents like dimethyl sulfoxide or acetonitrile to enhance the solubility of the inorganic base and facilitate the nucleophilic substitution. Detailed standard operating procedures regarding temperature ramps, addition rates, and quenching protocols are essential for reproducibility and safety.
- React anisole with diethyl phosphite using n-butyllithium and a depolymerizing agent to form bis(2-methoxyphenyl)phosphine oxide.
- Reduce the phosphine oxide intermediate using trichlorosilane and a tertiary amine to obtain bis(2-methoxyphenyl)phosphine.
- Couple the phosphine with 5,5-bis(bromomethyl)-1,5-dioxa-spiro[5,5]undecane under basic conditions to yield the final ligand.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers compelling economic and logistical benefits that extend beyond simple yield improvements. By shifting the raw material base from specialized brominated aromatics to bulk commodity anisole, manufacturers can insulate their production costs from the volatility of the halogenated intermediate market, ensuring more stable pricing structures for long-term contracts. The elimination of hazardous reagents like sodium hydride not only reduces the cost of specialized safety equipment and training but also lowers insurance premiums and regulatory compliance burdens associated with handling pyrophoric materials. Furthermore, the simplified workup procedures, which avoid complex distillation of tert-butanol or extensive washing to remove sodium salts, translate directly into reduced utility consumption and shorter batch cycle times, enhancing overall plant throughput and asset utilization rates.
- Cost Reduction in Manufacturing: The substitution of expensive o-bromoanisole with cost-effective anisole significantly lowers the Bill of Materials (BOM), while the avoidance of sodium hydride eliminates the need for expensive mineral oil disposal and specialized inert atmosphere handling equipment. The streamlined purification process reduces solvent usage and energy consumption during distillation, leading to substantial operational expenditure savings without compromising product quality. Additionally, the high selectivity of the reaction minimizes the formation of hard-to-remove impurities, reducing the loss of valuable product during recrystallization and chromatography steps.
- Enhanced Supply Chain Reliability: Sourcing anisole and diethyl phosphite is far more reliable than procuring specialized brominated intermediates, as these chemicals are produced at massive scales for diverse industries, ensuring continuous availability even during market disruptions. The robustness of the synthetic route against minor variations in reaction conditions means that production schedules are less likely to be impacted by batch failures or off-spec results, guaranteeing consistent delivery timelines to downstream polymer manufacturers. This reliability is crucial for maintaining just-in-time inventory levels and preventing production stoppages in the high-value polyketone sector.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing common reactor materials and avoiding extreme pressure or temperature conditions that would require specialized vessel certification. The reduction in hazardous waste generation, particularly the absence of halogenated byproducts and pyrophoric residues, simplifies wastewater treatment and aligns with increasingly stringent global environmental regulations. This green chemistry profile enhances the corporate sustainability rating of the manufacturer, making the supply chain more resilient to future regulatory changes and appealing to eco-conscious end-users in the polymer industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel ligand synthesis technology. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on safety, purity, and scalability. Understanding these aspects is vital for technical teams evaluating the feasibility of integrating this route into existing manufacturing facilities or for procurement specialists assessing the long-term viability of the supply source.
Q: What are the safety advantages of this new synthesis method compared to traditional routes?
A: The novel method eliminates the use of hazardous sodium hydride and tert-butoxide, replacing them with safer anhydrous alkali hydroxides and trichlorosilane reduction, significantly reducing explosion risks and simplifying waste treatment.
Q: How does the purity of the ligand produced via patent CN114106048A compare to market standards?
A: Experimental data indicates the process consistently achieves product purity exceeding 99% with yields over 90%, meeting the stringent requirements for high-activity polyketone polymerization catalysts.
Q: Why is anisole preferred over o-bromoanisole as the starting material?
A: Anisole is a more cost-effective and readily available commodity chemical compared to o-bromoanisole. Direct lithiation avoids the supply chain volatility and higher costs associated with halogenated aromatic precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3-bis-[bis-(2-methoxyphenyl)phosphinomethyl]-1,5-dioxa-spiro[5,5]undecane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity ligands play in the efficiency of polyketone production, and we possess the technical expertise to replicate and optimize the advanced synthesis routes described in patent CN114106048A. Our R&D team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and robust. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify that every batch of ligand meets the exacting standards required for high-activity palladium catalysts, thereby guaranteeing consistent polymer quality for our clients.
We invite forward-thinking organizations to collaborate with us on Customized Cost-Saving Analysis to evaluate how switching to this optimized synthesis route can impact your overall production economics. Our technical procurement team is ready to provide specific COA data and route feasibility assessments tailored to your volume requirements, helping you secure a stable and cost-effective supply of this essential specialty chemical intermediate. Contact us today to discuss how we can support your polymer manufacturing goals with reliable, high-performance ligand solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
