Advanced Manufacturing of Quinazolone Derivatives for Oncology Applications
Advanced Manufacturing of Quinazolone Derivatives for Oncology Applications
The pharmaceutical industry is constantly seeking novel scaffolds that can effectively target oncogenic pathways with high selectivity and low toxicity. Patent CN102382064B introduces a significant advancement in this field by disclosing a series of quinazolone derivatives characterized by a specific structural formula (I). These compounds exhibit potent inhibitory effects on telomerase activity and the expression of proto-oncogenes such as c-kit, making them highly valuable candidates for anticancer drug development. The core innovation lies in the versatile substitution patterns allowed at the R1, R2, R3, and R4 positions, which enable fine-tuning of pharmacological properties. As a leading reliable pharmaceutical intermediate supplier, understanding the structural nuances of these molecules is critical for scaling their production to meet the demands of modern oncology research.
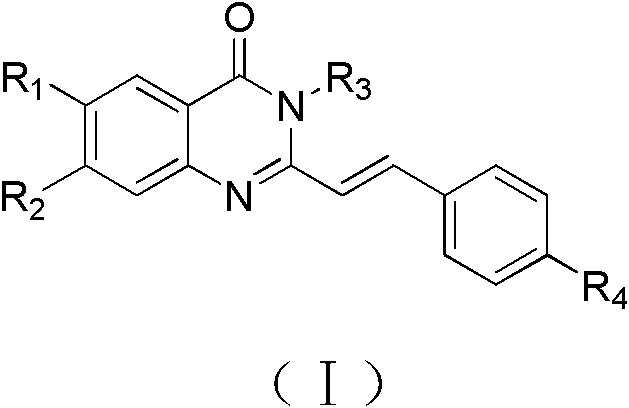
The structural versatility of Formula (I) allows for the incorporation of halogens (F, Cl, Br, I) at the R1 position and various amine-containing side chains at R2 and R4. This flexibility is essential for optimizing binding affinity to G-quadruplex DNA structures. The presence of aromatic rings (Ar) and alkyl chains further enhances the lipophilicity and cell permeability of these agents. For R&D directors focusing on purity and impurity profiles, the defined synthetic pathways provided in the patent offer a clear roadmap for controlling regio-isomers and by-products, ensuring the delivery of high-purity pharmaceutical intermediates suitable for preclinical and clinical evaluation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of quinazolone alkaloids often relies on harsh conditions that can lead to poor yields and difficult purification processes. Many conventional routes struggle with the selective functionalization of the quinazolone core, particularly when introducing complex side chains required for biological activity. The lack of modularity in older methods frequently necessitates lengthy protection and deprotection sequences, which not only increase the overall cost of goods but also generate significant chemical waste. Furthermore, achieving high stereochemical purity and controlling the substitution pattern on the benzene ring can be challenging with non-specific catalysts, leading to heterogeneous product mixtures that complicate downstream drug formulation.
The Novel Approach
The methodology outlined in CN102382064B presents a streamlined and modular strategy that overcomes these historical bottlenecks. By utilizing a stepwise construction starting from substituted 2-aminobenzoic acids, the process achieves high regioselectivity during the initial ring closure. The subsequent nucleophilic substitutions and condensations are performed under controlled conditions that maximize yield while minimizing side reactions. This novel approach significantly simplifies the manufacturing workflow, allowing for the efficient introduction of diverse functional groups such as piperazinyl and morpholinyl moieties. For procurement managers, this translates to cost reduction in pharmaceutical intermediate manufacturing through improved atom economy and reduced solvent consumption.
Mechanistic Insights into Quinazolone Ring Formation and Functionalization
The core mechanistic pathway involves the initial cyclization of 4,5-dihalo-substituted 2-aminobenzoic acid with acetic anhydride. This reaction proceeds via an intramolecular condensation where the amino group attacks the activated carboxylic acid derivative, forming the stable quinazolone ring system. The presence of halogen atoms at the 4 and 5 positions serves a dual purpose: they activate the ring towards subsequent nucleophilic aromatic substitution and provide handles for further cross-coupling reactions if needed. The use of acetic anhydride as both solvent and reagent drives the equilibrium towards product formation, ensuring high conversion rates. This robust cyclization step is fundamental to establishing the rigid planar structure required for intercalation or stacking interactions with DNA targets.
Following ring formation, the mechanism shifts to nucleophilic substitution at the carbonyl carbon or the C2 position, depending on the specific derivative desired. The reaction with aromatic amines (H2NR3) or secondary amines (HR2) exploits the electrophilic nature of the quinazolone scaffold. In the subsequent steps, condensation with nitrobenzaldehyde followed by reduction with sodium sulfide (Na2S) installs the styryl side chain with high E-selectivity. This sequence is crucial for extending the conjugated system, which enhances the molecule's ability to stabilize G-quadruplex structures. Understanding these mechanistic details allows process chemists to optimize reaction parameters such as temperature and stoichiometry to ensure consistent quality across large-scale batches.
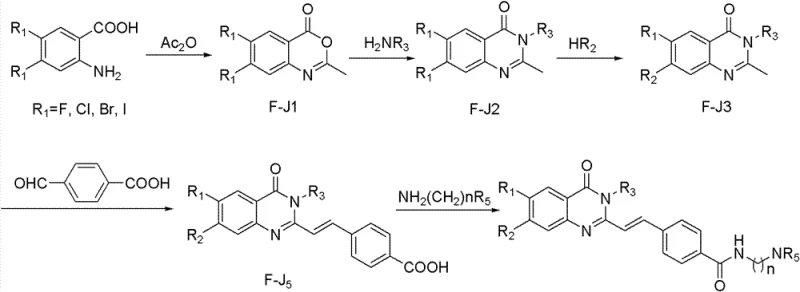
How to Synthesize Quinazolone Derivatives Efficiently
The synthesis of these bioactive molecules requires precise control over reaction conditions to maintain the integrity of the sensitive functional groups. The patented process outlines a logical progression from simple starting materials to complex final products, emphasizing the importance of purification at each stage. Detailed operational procedures involve refluxing conditions for cyclization, followed by careful temperature control during the reduction and acylation steps. For technical teams looking to implement this chemistry, it is essential to adhere to the specified molar ratios and solvent systems to achieve the reported yields. The detailed standardized synthesis steps see the guide below for a comprehensive breakdown of the operational protocol.
- Cyclization of substituted 2-aminobenzoic acid with acetic anhydride to form the core quinazolone ring.
- Nucleophilic substitution at the carbonyl or ring position using aromatic amines or secondary amines.
- Condensation with substituted benzaldehydes followed by reduction or acylation to install the final side chains.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the synthetic route described in this patent offers substantial advantages for supply chain stability and cost management. The reliance on commodity chemicals such as substituted benzoic acids, acetic anhydride, and common amines ensures that raw material sourcing is not a bottleneck. Unlike processes requiring rare earth metals or exotic catalysts, this method utilizes standard organic reagents that are readily available in the global market. This accessibility significantly reduces the risk of supply disruptions and allows for flexible procurement strategies. For supply chain heads, the ability to source materials from multiple vendors provides a strategic buffer against market volatility and price fluctuations.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and complex purification steps leads to a direct reduction in manufacturing costs. The process utilizes simple workup procedures such as filtration and recrystallization, which are far more economical than chromatographic separations on a large scale. By minimizing the number of unit operations and reducing solvent usage, the overall production cost is significantly lowered. This economic efficiency makes the commercial scale-up of complex pharmaceutical intermediates more viable, allowing for competitive pricing in the generic drug market.
- Enhanced Supply Chain Reliability: The use of robust and well-understood chemical transformations ensures high process reliability. The reactions are tolerant to minor variations in input quality, which reduces the rejection rate of raw materials. This robustness translates to consistent production schedules and reliable delivery times for customers. Furthermore, the scalability of the process from gram to kilogram scales has been demonstrated in the examples, providing confidence in the ability to meet increasing demand without compromising quality or lead time.
- Scalability and Environmental Compliance: The synthetic pathway is designed with scalability in mind, avoiding hazardous reagents that would complicate waste treatment. The by-products generated are primarily organic salts and aqueous waste, which can be treated using standard effluent processing methods. This alignment with green chemistry principles facilitates regulatory compliance and reduces the environmental footprint of the manufacturing facility. The ability to scale up safely and sustainably is a key factor for long-term partnerships with major pharmaceutical companies focused on corporate social responsibility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these quinazolone derivatives. The answers are derived directly from the experimental data and structural analysis provided in the patent documentation. Understanding these aspects is vital for stakeholders evaluating the feasibility of integrating these intermediates into their drug discovery pipelines. The information covers biological activity, synthetic feasibility, and potential applications in oncology.
Q: What is the primary biological target of these quinazolone derivatives?
A: These derivatives are designed to inhibit proto-oncogene DNA expression, specifically targeting telomere DNA and c-kit G-quadruplex structures, showing significant antitumor activity.
Q: Are the starting materials for this synthesis commercially available?
A: Yes, the synthesis utilizes readily available commodity chemicals such as substituted 2-aminobenzoic acids, acetic anhydride, and various aromatic amines, ensuring supply chain stability.
Q: How does this method improve upon traditional quinazolone synthesis?
A: The patented route offers a modular approach allowing for diverse substitution at the R1, R2, and R4 positions, facilitating the rapid generation of analog libraries for SAR studies without complex protection/deprotection steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinazolone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications for every batch of quinazolone derivatives produced. We understand the critical nature of oncology intermediates and are committed to delivering materials that meet the highest international standards for safety and efficacy. Our team of expert chemists is ready to assist in optimizing the synthesis route to further enhance yield and reduce costs.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. By partnering with us, you gain access to specific COA data and route feasibility assessments that will accelerate your development timeline. Let us support your journey from early-stage research to commercial manufacturing with our reliable supply chain and technical expertise. Reach out today to discuss how we can collaborate on the next generation of anticancer therapeutics.
