Revolutionizing 3-Benzofuranone Production: A Low-Cost Mercury-Catalyzed Strategy for Commercial Scale-Up
Revolutionizing 3-Benzofuranone Production: A Low-Cost Mercury-Catalyzed Strategy for Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking efficient, cost-effective pathways to synthesize high-value heterocyclic scaffolds. A recent breakthrough detailed in patent CN111100101A introduces a novel catalytic synthesis method for 3-benzofuranone compounds that fundamentally shifts the economic landscape of producing these critical intermediates. Unlike traditional methods that rely on prohibitively expensive noble metals, this innovation utilizes a mercury-based catalytic system that operates under remarkably mild conditions. For R&D directors and procurement managers alike, this technology represents a pivotal opportunity to optimize supply chains and reduce the cost of goods sold (COGS) for downstream APIs and fine chemicals. The method achieves exceptional yields between 91.0% and 96.0% while drastically simplifying the operational workflow, positioning it as a superior alternative for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-benzofuranone derivatives has been plagued by significant economic and operational bottlenecks. Prior art, such as methods utilizing o-chlorophenyl diketone and phenylboronic acid, often necessitates the use of rhodium complexes or palladium acetate as catalysts. These noble metal catalysts are not only astronomically expensive, with market prices frequently exceeding 1000 yuan per gram, but they also introduce severe supply chain vulnerabilities due to their scarcity and geopolitical sensitivity. Furthermore, these conventional routes typically demand harsh reaction conditions, including elevated temperatures and extended reaction times, which increase energy consumption and complicate process safety. The complexity of removing trace heavy metal residues from the final product to meet stringent pharmaceutical purity specifications adds another layer of cost and time, often requiring specialized scavenging resins or multiple purification steps that erode overall process efficiency.
The Novel Approach
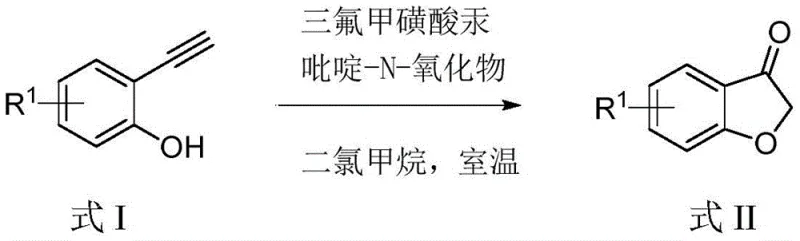
In stark contrast, the methodology disclosed in CN111100101A leverages mercury trifluoromethanesulfonate as a highly effective and economically viable catalyst. With a market price of less than 30 yuan per gram, this catalyst offers a dramatic reduction in raw material costs compared to rhodium or palladium alternatives. The process operates at room temperature and normal pressure, eliminating the need for energy-intensive heating or pressurized reactors. By employing pyridine-N-oxide as a stoichiometric oxidant in a dichloromethane solvent system, the reaction proceeds rapidly to completion within just 1 hour. This streamlined approach not only accelerates throughput but also minimizes the formation of complex by-products, thereby simplifying downstream purification. The versatility of this method is evidenced by its compatibility with a wide range of substituents, including methyl, methoxy, fluoro, chloro, and nitro groups, ensuring broad applicability across diverse chemical libraries.
Mechanistic Insights into Mercury-Catalyzed Cyclization
The core of this technological advancement lies in the unique activation mode of the alkyne moiety by the mercury catalyst. The mechanism initiates with the coordination of the mercury trifluoromethanesulfonate to the triple bond of the 2-ethynylphenol derivative, significantly increasing the electrophilicity of the alkyne carbons. This activation facilitates an intramolecular nucleophilic attack by the adjacent phenolic hydroxyl group, leading to the formation of a cyclic vinyl-mercury intermediate. Subsequently, the presence of pyridine-N-oxide acts as a crucial oxidant, promoting the oxidative cleavage or transformation of the organomercury species to generate the final carbonyl functionality of the 3-benzofuranone ring. This tandem cyclization-oxidation sequence is highly efficient, avoiding the need for separate oxidation steps that are common in other synthetic routes. The mildness of the conditions preserves sensitive functional groups on the aromatic ring, allowing for the synthesis of diverse derivatives without protecting group strategies.
From an impurity control perspective, this mechanism offers distinct advantages for maintaining high product purity. The rapid kinetics of the reaction, completing within 1 hour, limits the timeframe for side reactions such as polymerization of the alkyne or over-oxidation of the phenol ring. The use of dichloromethane as a solvent ensures good solubility of both the organic substrates and the ionic mercury catalyst, promoting homogeneous reaction conditions that minimize localized hot spots or concentration gradients which often lead to impurity formation. Furthermore, the stoichiometric use of pyridine-N-oxide (120% molar amount) ensures complete conversion of the intermediate without excessive oxidant that could degrade the product. The resulting crude reaction mixtures are typically clean, facilitating straightforward isolation via silica gel column chromatography or crystallization, which is critical for meeting the rigorous quality standards required for reliable pharmaceutical intermediate suppliers.

How to Synthesize 3-Benzofuranone Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting is straightforward and requires standard equipment. The process begins by dissolving the specific 2-ethynylphenol derivative in dichloromethane at a precise molar volume ratio of 1:5 mmol/mL to ensure optimal concentration for reaction kinetics. Following this, the catalyst mercury trifluoromethanesulfonate is added at a loading of 5 mol%, followed by the addition of pyridine-N-oxide at 120 mol%. The mixture is then stirred vigorously at room temperature, with stirring speeds optimized between 300 to 1000 r/min to maintain suspension and mass transfer. Detailed standardized operating procedures and safety protocols for handling mercury salts and scaling this reaction are provided in the technical guide below.
- Dissolve the 2-ethynylphenol derivative in dichloromethane at a molar volume ratio of 1: 5 mmol/mL under normal temperature and pressure.
- Add mercury trifluoromethanesulfonate (5 mol%) and pyridine-N-oxide (120 mol%) to the solution.
- Stir the mixture at room temperature (300-1000 r/min) for 1 hour to complete the cyclization and oxidation reaction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this mercury-catalyzed protocol translates directly into tangible bottom-line improvements and risk mitigation. The most immediate impact is the drastic reduction in catalyst expenditure; shifting from a rhodium-based system costing over 1000 yuan/g to a mercury-based system under 30 yuan/g represents an order-of-magnitude decrease in direct material costs. This cost structure allows for more competitive pricing of the final 3-benzofuranone intermediates, enhancing margin potential for downstream drug manufacturers. Additionally, the simplicity of the operation—requiring only ambient temperature and pressure—reduces the capital expenditure (CAPEX) associated with specialized high-pressure or high-temperature reactors, making it accessible for a wider range of manufacturing facilities. The short reaction time of 1 hour significantly boosts asset utilization rates, allowing existing production lines to achieve higher throughput without additional infrastructure investment.
- Cost Reduction in Manufacturing: The substitution of expensive noble metal catalysts with affordable mercury trifluoromethanesulfonate eliminates a major cost driver in the synthesis of benzofuranone derivatives. This change not only lowers the bill of materials but also reduces the financial burden associated with the recovery and recycling of precious metals, which is often technically challenging and costly. The high yields observed (91.0-96.0%) further contribute to cost efficiency by maximizing the output per unit of raw material input, minimizing waste disposal costs and improving overall atom economy. Consequently, this process enables substantial cost savings in API manufacturing, making it an attractive option for generic drug producers and contract manufacturing organizations aiming to optimize their production budgets.
- Enhanced Supply Chain Reliability: Relying on scarce noble metals like rhodium and palladium exposes supply chains to volatility driven by mining outputs and geopolitical tensions. By transitioning to a mercury-based catalyst, which is more abundant and stable in pricing, manufacturers can secure a more predictable and resilient supply of critical reagents. The mild reaction conditions also reduce the dependency on complex utility systems (e.g., high-pressure steam or cryogenic cooling), ensuring that production can continue uninterrupted even during utility fluctuations. This robustness is essential for maintaining consistent delivery schedules to global clients, reinforcing the status of the manufacturer as a reliable pharmaceutical intermediate supplier capable of meeting demanding Just-In-Time (JIT) requirements.
- Scalability and Environmental Compliance: While mercury handling requires strict safety protocols, the overall process design supports easy scalability from gram to ton scales due to its exothermic neutrality and lack of gas evolution. The use of dichloromethane, a common industrial solvent, allows for established recovery and recycling protocols to be implemented, minimizing solvent waste. Although mercury is a heavy metal, the low loading (5 mol%) and the potential for efficient aqueous workups to sequester mercury ions allow for compliant waste management strategies that meet international environmental standards. The simplified workflow reduces the number of unit operations, thereby lowering the overall environmental footprint of the manufacturing process compared to multi-step conventional routes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic synthesis method. These insights are derived directly from the experimental data and comparative analysis presented in patent CN111100101A, providing clarity on the feasibility and benefits of adopting this technology for industrial applications. Understanding these details is crucial for technical teams evaluating the integration of this route into their existing manufacturing portfolios.
Q: What are the cost advantages of this mercury-catalyzed method compared to traditional rhodium catalysts?
A: The patent highlights that mercury trifluoromethanesulfonate costs less than 30 yuan/gram, whereas traditional rhodium catalysts often exceed 1000 yuan/gram, representing a massive reduction in raw material expenditure.
Q: What are the typical reaction yields for this synthesis?
A: The process demonstrates high efficiency with yields ranging from 91.0% to 96.0% across various substituted 2-ethynylphenol derivatives.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the reaction occurs at room temperature and normal pressure with a short reaction time of only 1 hour, making it highly scalable and operationally simple for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Benzofuranone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the catalytic synthesis method described in CN111100101A for the production of high-purity 3-benzofuranone compounds. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this low-cost mercury-catalyzed route are fully realized in a GMP-compliant environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of 3-benzofuranone intermediate meets the exacting standards required for pharmaceutical and agrochemical applications. We are committed to leveraging such innovative technologies to deliver superior value to our global partners, combining technical excellence with operational reliability.
We invite R&D directors and procurement leaders to collaborate with us to evaluate the feasibility of this route for your specific projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this novel synthesis method. We encourage you to contact us today to obtain specific COA data for our existing benzofuranone library and to discuss route feasibility assessments tailored to your target molecules. Let us help you optimize your supply chain and accelerate your time-to-market with our advanced manufacturing capabilities.
