Optimizing Efavirenz Intermediate Production via Novel Salt-Formation Refining Strategies
Optimizing Efavirenz Intermediate Production via Novel Salt-Formation Refining Strategies
The pharmaceutical industry continuously seeks robust methodologies to enhance the efficiency and sustainability of Active Pharmaceutical Ingredient (API) synthesis, particularly for critical antiretroviral therapies. Patent CN113979877A introduces a transformative refining method specifically targeting the purification of 4-chloro-2-trifluoroacetylaniline, a pivotal intermediate in the synthesis of Efavirenz. This non-nucleoside reverse transcriptase inhibitor remains a cornerstone in HIV-1 treatment regimens globally, necessitating supply chains that can deliver high-purity precursors with minimal environmental impact. The disclosed technology addresses the persistent challenge of low-purity mother liquors generated during standard production, offering a pathway to recover valuable materials that would otherwise be discarded as waste. By leveraging a sophisticated sequence of vacuum concentration, hydrochloride salt formation, and strategic recrystallization, this process achieves product quality indistinguishable from primary batch standards.
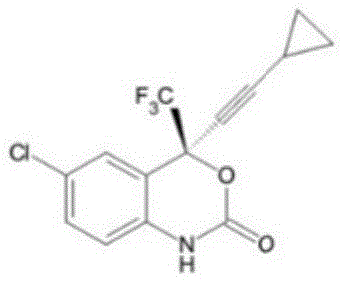
Efavirenz, chemically defined as (S)-6-chloro-4-(cyclopropylethynyl)-1,4-dihydro-4-(trifluoromethyl)-2H-3,1-benzoxazin-2-one, represents a complex molecular architecture where stereochemical integrity and impurity control are paramount. The synthetic route to this molecule is notoriously lengthy, often resulting in accumulated impurities in the mother liquor streams of its intermediates. Traditional approaches to handling these streams have been inefficient, typically yielding products with purities hovering around 85% and failing to adequately remove specific degradation byproducts. The innovation presented in this patent fundamentally shifts the paradigm from waste disposal to resource recovery, providing a reliable pharmaceutical intermediate supplier with the technical capability to maximize yield from every kilogram of raw material input while adhering to stringent regulatory purity specifications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of 4-chloro-2-trifluoroacetylaniline mother liquor has been plagued by significant technical hurdles that compromise both economic efficiency and product quality. Conventional direct crystallization methods often rely on benign solvents to dissolve the crude material, followed by the addition of non-benign solvents to induce precipitation. However, analytical data reveals that this approach is remarkably ineffective at removing critical degradation impurities, particularly those exhibiting relative retention times (RRT) between 0.7 and 0.8. These stubborn impurities co-crystallize with the target molecule, capping the achievable purity at approximately 85% and rendering the recovered material unsuitable for subsequent coupling reactions in the Efavirenz synthesis. Furthermore, the inability to effectively separate N-pivaloyl parachloroaniline (RRT 0.3~0.4) and other high-RRT contaminants means that manufacturers face a difficult choice: either accept substandard intermediates that risk downstream reaction failure or incur the substantial costs associated with hazardous waste disposal. This inefficiency not only inflates the cost of goods sold but also creates a bottleneck in the supply chain, as the low comprehensive yield necessitates larger initial batches to meet final API demand.
The Novel Approach
The patented methodology circumvents these limitations by introducing a chemical modification step—hydrochloride salt formation—that fundamentally alters the physicochemical properties of the intermediate. Instead of attempting to crystallize the free base directly from a contaminated mixture, the process first converts the 4-chloro-2-trifluoroacetylaniline into its hydrochloride salt using concentrated hydrochloric acid in solvents such as isopropyl acetate or ethyl acetate. This transformation exploits the differential solubility between the target hydrochloride and the neutral organic impurities, allowing for a highly selective purification during the subsequent reflux pulping stage. By refining the salt rather than the free base, the process effectively strips away the degradation impurities that previously resisted removal. Following the purification of the salt, a controlled liberation step using liquid caustic soda regenerates the free base, which is then subjected to a final polishing crystallization. This multi-stage strategy ensures that the final product achieves a purity of greater than 99.0%, matching the quality of the original main batch and validating the process as a viable industrial solution for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Hydrochloride Salt Formation and Purification
The core mechanism driving the success of this refining process lies in the thermodynamic and kinetic advantages conferred by the salt formation step. When the concentrated mother liquor residue is treated with concentrated hydrochloric acid at controlled temperatures between 0°C and 10°C, the amine functionality of the 4-chloro-2-trifluoroacetylaniline is protonated to form a stable ammonium chloride salt. This ionic species exhibits distinct solubility characteristics compared to the neutral impurities present in the matrix, such as the N-pivaloyl derivatives and various degradation products. During the reflux pulping phase in alcoholic solvents like ethanol or isopropanol, the hydrochloride salt remains largely in the solid phase or recrystallizes selectively, while the non-ionic impurities remain dissolved in the supernatant. This solid-liquid equilibrium is meticulously managed by maintaining the temperature at 10-15°C during the final stirring period, which promotes the growth of high-purity crystals while minimizing the occlusion of mother liquor containing contaminants. The subsequent liberation step, performed in methyl tert-butyl ether with pH adjustment to 7-8, ensures a clean phase separation where the organic layer retains the purified free base, leaving inorganic salts and water-soluble byproducts in the aqueous phase.
Impurity control is further enhanced by the specific selection of solvents for the final recrystallization of the liberated crude product. The patent specifies the use of n-hexane or n-heptane, which act as anti-solvents to precisely tune the saturation point of the 4-chloro-2-trifluoroacetylaniline. Heating the mixture to 50-60°C ensures complete dissolution of the desired compound while keeping high-molecular-weight impurities insoluble or easily filterable. As the solution cools to 10-20°C, the target molecule precipitates in a highly ordered crystalline lattice that inherently excludes structurally dissimilar impurities. This final polishing step is critical for reducing the levels of trace contaminants with RRT values of 1.5~1.6 and 1.8~2.1, which might have survived the earlier salt refinement. The result is a comprehensive impurity profile that meets the rigorous standards required for GMP manufacturing of antiretroviral drugs, demonstrating how mechanistic understanding of solubility and phase behavior can be leveraged to solve complex purification challenges in fine chemical synthesis.
How to Synthesize 4-Chloro-2-trifluoroacetylaniline Efficiently
The implementation of this refining protocol requires precise adherence to thermal and stoichiometric parameters to ensure consistent recovery of high-purity material from waste streams. The process begins with the concentration of the refined mother liquor under reduced pressure, a step that removes bulk solvents without exposing the heat-sensitive intermediate to excessive thermal stress. Following the formation and refinement of the hydrochloride salt, the liberation and final crystallization steps must be executed with careful pH control and temperature ramping to maximize yield. For detailed operational procedures, including specific solvent ratios and agitation speeds, please refer to the standardized synthesis guide below which outlines the exact execution of this patented technology.
- Concentrate the refined mother liquor under reduced pressure at 30-70°C and -0.08 to -0.09 MPa to obtain a dry residue.
- Dissolve the residue in a solvent like isopropyl acetate, cool to 0-10°C, and add concentrated hydrochloric acid to form the hydrochloride salt.
- Refine the crude hydrochloride by reflux pulping in ethanol or isopropanol, followed by cooling and filtration to remove impurities.
- Liberate the free base by dissolving the refined salt in methyl tert-butyl ether and adjusting pH to 7-8 with liquid caustic soda.
- Perform a final recrystallization of the crude product using n-hexane or n-heptane to achieve purity greater than 99.0%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this refining technology translates directly into enhanced operational resilience and significant financial optimization. By converting what was previously classified as hazardous waste into a saleable, high-value intermediate, manufacturers can drastically reduce the volume of solid waste requiring disposal, thereby lowering environmental compliance costs and mitigating regulatory risks. The simplicity of the process, which utilizes common industrial reagents like hydrochloric acid and standard solvents, ensures that the technology can be scaled up rapidly without the need for exotic catalysts or specialized equipment, facilitating a seamless integration into existing production facilities. This capability to recover value from mother liquors effectively increases the overall throughput of the plant without increasing the consumption of raw starting materials, providing a buffer against supply volatility and price fluctuations in the global chemical market.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the elimination of raw material loss associated with discarding mother liquor. By recovering 4-chloro-2-trifluoroacetylaniline with purity levels comparable to the main batch, the effective yield of the entire synthetic route is substantially improved. This reduces the cost per kilogram of the final API, as the expense of the initial reactants is amortized over a larger quantity of usable product. Furthermore, the avoidance of expensive chromatographic purification or complex enzymatic resolutions in favor of simple crystallization and salt formation keeps capital expenditure and operating costs low, ensuring a competitive pricing structure for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Implementing a robust mother liquor recovery process adds a layer of security to the supply chain by creating an internal buffer of critical intermediates. In scenarios where upstream supply of raw anilines or trifluoroacetylating agents is disrupted, the ability to recycle and refine existing inventory ensures continuity of production. The use of widely available solvents and reagents means that the process is not dependent on single-source suppliers for niche chemicals, reducing the risk of procurement bottlenecks. This reliability is crucial for maintaining the steady supply of antiretroviral medications, where interruptions can have severe public health consequences.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial production in mind, utilizing unit operations such as vacuum concentration and filtration that are standard in large-scale chemical plants. From an environmental perspective, the reduction in solid waste generation aligns with green chemistry principles and increasingly strict environmental regulations regarding pharmaceutical manufacturing. By minimizing the discharge of organic contaminants and maximizing atom economy through recycling, manufacturers can improve their sustainability metrics and corporate social responsibility profiles, which are becoming key differentiators in B2B procurement decisions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this refining method, derived directly from the patent specifications and practical application scenarios. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into their current manufacturing workflows to ensure optimal outcomes.
Q: Why is salt formation preferred over direct crystallization for this intermediate?
A: Direct crystallization often fails to effectively remove degradation impurities with relative retention times (RRT) between 0.7 and 0.8. Converting the intermediate into its hydrochloride salt alters its solubility profile, allowing for the selective precipitation of the target compound while leaving key impurities in the mother liquor, thereby significantly enhancing final purity.
Q: What are the critical process parameters for the hydrochloride formation step?
A: Temperature control is vital; the dissolution should occur at 0-10°C before adding concentrated hydrochloric acid. The reaction mixture must then be maintained at 20-25°C with stirring for approximately one hour to ensure complete salt formation and optimal crystal growth before filtration.
Q: Can this method handle mother liquor with low initial purity?
A: Yes, the method is specifically designed to upgrade mother liquor with initial purities around 85%. Through the multi-stage process of concentration, salt formation, refinement, and liberation, the process consistently yields product with purity exceeding 99.0%, effectively turning waste streams into valuable assets.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Chloro-2-trifluoroacetylaniline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the efficient production of Efavirenz intermediates requires not just chemical expertise but a deep commitment to process optimization and quality assurance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory patent data to full-scale manufacturing is smooth and compliant. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect and quantify trace impurities, guaranteeing that every batch of 4-chloro-2-trifluoroacetylaniline meets the exacting standards required for HIV drug synthesis. Our infrastructure is designed to handle complex refining protocols, including the salt-formation techniques described in CN113979877A, delivering consistent quality that supports your regulatory filings and production schedules.
We invite potential partners to engage with our technical procurement team to discuss how our advanced refining capabilities can support your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of implementing mother liquor recovery strategies in your own operations. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both the profitability and sustainability of your pharmaceutical manufacturing endeavors.
