Scalable Synthesis of Tetrahydronaphthyridine Derivatives for High-Potency H3 Receptor Ligands
Scalable Synthesis of Tetrahydronaphthyridine Derivatives for High-Potency H3 Receptor Ligands
The pharmaceutical landscape for treating central nervous system disorders and respiratory conditions is continually evolving, driven by the need for highly selective receptor ligands with improved safety profiles. Patent CN101798305A discloses a novel class of tetrahydronaphthyridine derivatives that function as potent histamine H3 receptor ligands, offering significant therapeutic potential for diseases such as allergic rhinitis, asthma, and cognitive impairments. This intellectual property outlines robust synthetic methodologies that address the challenges of constructing complex heterocyclic scaffolds with high precision. For procurement and supply chain leaders, understanding the underlying chemistry is crucial for securing a reliable pharmaceutical intermediate supplier capable of delivering these high-value compounds. The disclosed processes leverage advanced catalytic techniques to ensure consistent quality and scalability, positioning these intermediates as key assets in the development of next-generation therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing fused heterocyclic systems like tetrahydronaphthyridines often suffer from poor regioselectivity and harsh reaction conditions that can compromise the integrity of sensitive functional groups. Conventional nucleophilic substitutions frequently require elevated temperatures and strong bases, leading to the formation of difficult-to-remove impurities and reduced overall yields. Furthermore, the lack of modularity in older synthetic routes makes it challenging to introduce diverse substituents at specific positions on the aromatic ring, limiting the ability to optimize pharmacokinetic properties during drug discovery. These inefficiencies result in increased production costs and extended lead times, creating bottlenecks for research and development teams aiming to bring new candidates to clinical trials efficiently.
The Novel Approach
The methodology presented in the patent overcomes these hurdles through a convergent synthetic strategy that utilizes protected piperidone precursors and enamine chemistry to construct the core scaffold with exceptional control. As illustrated in the reaction scheme below, the process begins with the condensation of a protected piperidone to form an enamine, which subsequently undergoes cyclization with acrylamide derivatives to establish the naphthyridine ring system. This route allows for the introduction of various protecting groups, such as benzyl or allyl, which safeguard the nitrogen atom during subsequent transformations. The flexibility of this approach enables the late-stage functionalization of the molecule, permitting the attachment of diverse heteroaromatic moieties essential for biological activity.
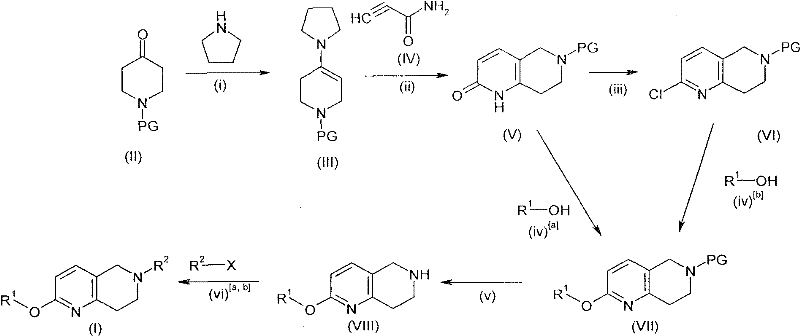
By employing this modular design, manufacturers can rapidly generate libraries of analogues for structure-activity relationship studies without redesigning the entire synthetic pathway. The use of mild conditions during the key bond-forming steps ensures that the final products meet stringent purity specifications required for pharmaceutical applications. This novel approach not only enhances the efficiency of the synthesis but also provides a versatile platform for the commercial scale-up of complex pharmaceutical intermediates, ensuring a steady supply of high-quality materials for drug development programs.
Mechanistic Insights into Palladium-Catalyzed Cross-Coupling
A critical component of the synthetic utility described in the patent is the application of palladium-catalyzed cross-coupling reactions to install the final heteroaromatic substituents. This transformation typically involves the reaction of a chloro- or bromo-tetrahydronaphthyridine intermediate with a heterocyclic amine or phenol in the presence of a palladium source such as Pd2(dba)3 and a bidentate phosphine ligand like BINAP. The mechanism proceeds through an oxidative addition of the palladium catalyst to the carbon-halogen bond, followed by coordination and insertion of the nucleophile, and finally reductive elimination to release the coupled product. This catalytic cycle is highly efficient and operates under relatively mild conditions, often facilitated by microwave irradiation in the laboratory setting to accelerate reaction kinetics.
From an impurity control perspective, the choice of ligand and base plays a pivotal role in minimizing the formation of homocoupling byproducts and dehalogenated species. The patent specifies the use of sodium tert-butoxide or cesium carbonate as bases, which effectively deprotonate the nucleophile without promoting degradation of the sensitive tetrahydronaphthyridine core. Additionally, the use of protecting groups on the piperidine nitrogen prevents competitive coordination with the palladium catalyst, ensuring that the reaction occurs exclusively at the desired site. This level of mechanistic understanding allows process chemists to fine-tune reaction parameters, such as temperature and stoichiometry, to maximize yield and purity, thereby reducing the burden on downstream purification processes.
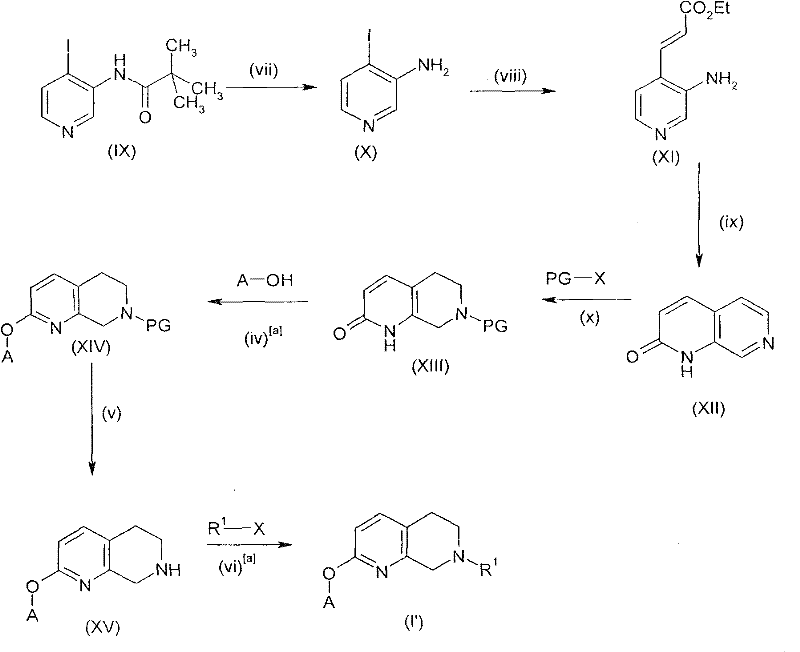
Furthermore, the patent describes an alternative synthetic pathway, shown in the scheme above, which utilizes a pyridine-based precursor to access specific regioisomers of the target molecule. This route involves the hydrolysis of an amide followed by cyclization and reduction steps, demonstrating the versatility of the chemical platform. By having multiple synthetic entry points, manufacturers can select the most cost-effective and scalable route based on the availability of raw materials and the specific substitution pattern required. This strategic flexibility is invaluable for supply chain management, as it mitigates the risk of disruptions associated with single-source starting materials.
How to Synthesize Tetrahydronaphthyridine Derivatives Efficiently
The synthesis of these high-value intermediates requires precise control over reaction conditions and purification techniques to ensure compliance with regulatory standards. The patent provides detailed experimental procedures that serve as a blueprint for manufacturing, outlining specific reagents, solvents, and workup protocols. Implementing these methods involves a sequence of condensation, cyclization, and coupling steps, each optimized to balance reaction rate with product quality. For R&D teams looking to replicate or scale these processes, adherence to the specified parameters is essential to achieve the reported yields and purity levels. The detailed standardized synthesis steps are outlined in the guide below.
- Condensation of protected piperidone with pyrrolidine to form an enamine intermediate under dehydrating conditions.
- Reaction of the enamine with acrylamide followed by cyclization and chlorination to generate the chloro-tetrahydronaphthyridine core.
- Final functionalization via Mitsunobu etherification or Pd-catalyzed arylation to install the side chain and heteroaromatic groups.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting the synthetic routes disclosed in this patent offers substantial strategic benefits for procurement and supply chain operations within the pharmaceutical sector. The modular nature of the synthesis allows for the use of commercially available starting materials, reducing dependency on custom-synthesized precursors that often carry long lead times and high price tags. By streamlining the production process, manufacturers can achieve significant cost reduction in pharmaceutical intermediate manufacturing, passing these savings on to their clients while maintaining healthy margins. The ability to switch between different synthetic pathways based on raw material availability further enhances supply chain resilience, ensuring continuity of supply even in volatile market conditions.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts in certain steps, or the use of catalytic amounts of palladium that can be recovered, significantly lowers the material cost per kilogram. Furthermore, the high selectivity of the reactions reduces the need for extensive chromatographic purification, which is often the most costly part of intermediate production. This efficiency translates directly into a more competitive pricing structure for the final active pharmaceutical ingredient, enabling broader patient access to life-saving medications.
- Enhanced Supply Chain Reliability: The reliance on robust, well-established chemical transformations such as enamine formation and nucleophilic substitution ensures that the process is less prone to failure compared to exotic or unproven methodologies. This reliability is critical for maintaining consistent delivery schedules, which is a top priority for supply chain heads managing global drug launches. The scalability of the process from gram to multi-ton scales means that suppliers can easily ramp up production to meet surging demand without compromising on quality or safety standards.
- Scalability and Environmental Compliance: The synthetic routes are designed with green chemistry principles in mind, utilizing solvents and reagents that are easier to handle and dispose of in an environmentally responsible manner. The reduction in waste generation and the potential for solvent recycling contribute to a lower environmental footprint, aligning with the sustainability goals of modern pharmaceutical companies. This compliance with environmental regulations simplifies the permitting process for manufacturing facilities and reduces the risk of regulatory penalties, ensuring a smooth and uninterrupted production flow.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these tetrahydronaphthyridine derivatives. The answers are derived from the detailed technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these aspects helps stakeholders make informed decisions about integrating these intermediates into their development pipelines.
Q: What are the key advantages of the Pd-catalyzed coupling method described in CN101798305A?
A: The method utilizes mild conditions with Pd2(dba)3 and BINAP, allowing for high regioselectivity and compatibility with sensitive functional groups, which minimizes impurity formation compared to traditional nucleophilic substitution.
Q: How does the protecting group strategy impact the overall yield?
A: The use of benzyl or allyl protecting groups on the nitrogen atom prevents unwanted side reactions during the cyclization steps, ensuring a cleaner reaction profile and simplifying downstream purification processes.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the patent describes conditions that can be transitioned from microwave-assisted laboratory synthesis to conventional thermal reflux, facilitating scalability from kilogram to multi-ton production levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydronaphthyridine Supplier
At NINGBO INNO PHARMCHEM, we specialize in translating complex patent methodologies into commercial reality, offering our partners a seamless bridge from discovery to market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of tetrahydronaphthyridine intermediate meets the highest industry standards. Our commitment to quality and consistency makes us the preferred partner for global pharmaceutical companies seeking to optimize their supply chains.
We invite you to collaborate with us to explore the full potential of these histamine H3 receptor ligands for your drug development projects. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to support your regulatory filings and accelerate your time to market. Let us help you secure a stable and cost-effective supply of these critical pharmaceutical intermediates.
