Advanced Synthesis of Tetradentate Benzoquinazoline Salts for High-Performance Fluorescent Sensing Applications
The landscape of fluorescent molecular recognition has been significantly advanced by the development of novel heterocyclic architectures capable of selective ion sensing. A pivotal contribution to this field is detailed in patent CN108069912B, which discloses a robust preparation method for a tetradentate benzoquinazoline salt compound. This specific chemical entity, characterized by an anthracene core functionalized with four benzoquinazoline units, represents a breakthrough in the design of high-performance optical sensors. The innovation lies not only in the final structure but in the strategic assembly of the molecule, which combines the rigid fluorescence properties of anthracene with the coordination capabilities of quinazoline derivatives. For industries seeking a reliable electronic chemical supplier capable of delivering complex organic salts, understanding the nuances of this synthesis is paramount. The resulting compound exhibits remarkable photosensitive effects and selective recognition capabilities, particularly towards chromium ions, positioning it as a critical component for next-generation analytical devices and optoelectronic materials.
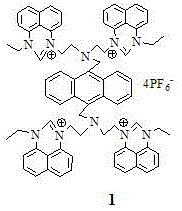
The structural integrity of this molecule is confirmed through rigorous single-crystal X-ray diffraction analysis, revealing a triclinic crystal system that ensures stability under standard conditions. The presence of the hexafluorophosphate anion is a deliberate design choice that enhances the solubility of the cationic species in polar aprotic solvents, thereby optimizing its utility in solution-phase fluorescence assays. As we delve deeper into the technical specifications, it becomes evident that this material offers a unique combination of photophysical properties and chemical stability that is rarely found in conventional fluorophores.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of multi-dentate fluorescent sensors has been plagued by inefficient reaction pathways and prohibitive costs associated with specialized precursors. Traditional methods often rely on transition metal-catalyzed cross-coupling reactions, which introduce significant challenges regarding the removal of trace metal impurities—a critical failure point for applications in high-purity electronic materials. Furthermore, conventional routes frequently suffer from poor regioselectivity, leading to complex mixtures of isomers that require extensive and wasteful chromatographic purification. These inefficiencies result in drastically reduced overall yields and extended production timelines, creating bottlenecks for cost reduction in electronic chemical manufacturing. Additionally, many existing fluorophores lack the necessary thermal and chemical stability required for industrial deployment, degrading rapidly under operational conditions or failing to maintain consistent fluorescence intensity in the presence of interfering ions.
The Novel Approach
In stark contrast, the methodology outlined in patent CN108069912B presents a streamlined, metal-free synthetic strategy that leverages readily available commodity chemicals. By utilizing anthracene as the central scaffold, the process capitalizes on the inherent symmetry of the starting material to facilitate the simultaneous installation of four functional arms. This convergent approach minimizes the number of isolation steps and maximizes atom economy. The use of simple alkylation and quaternization reactions eliminates the need for expensive palladium or copper catalysts, thereby simplifying the downstream purification process and ensuring a cleaner final product profile. This novel pathway not only accelerates the time-to-market for high-purity fluorescent material but also aligns with green chemistry principles by reducing solvent waste and energy consumption. The strategic selection of reagents ensures that the reaction conditions remain mild yet effective, allowing for precise control over the degree of substitution and minimizing the formation of unwanted by-products.
Mechanistic Insights into Anthracene-Based Quaternization and Anion Exchange
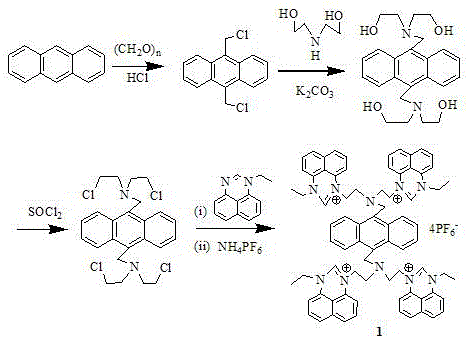
The synthesis mechanism is a masterclass in functional group interconversion, beginning with the activation of the anthracene core. The initial chloromethylation step generates highly reactive benzylic chloride intermediates, which serve as electrophilic sites for subsequent nucleophilic attacks. This is followed by the introduction of diethanolamine, which installs the necessary nitrogen-containing linkers. The critical transformation occurs during the reaction with thionyl chloride, where hydroxyl groups are converted into chloroethyl moieties. This activation is essential for the final quaternization step, where the nitrogen atoms of the N-ethylbenzoquinazoline units attack the alkyl chlorides to form stable quaternary ammonium salts. The entire sequence is driven by thermodynamic favorability and careful control of stoichiometry, ensuring that the tetradentate architecture is formed with high fidelity.
Following the formation of the tetraiodide salt, the process employs a metathesis reaction using ammonium hexafluorophosphate (NH4PF6). This anion exchange is mechanistically driven by the low solubility of the resulting hexafluorophosphate salt in the reaction medium, which precipitates the product and drives the equilibrium to completion. This step is crucial for tuning the physicochemical properties of the final sensor; the bulky, non-coordinating PF6- anion prevents interference with the metal-binding sites of the quinazoline ligands, thereby preserving the fluorescence quantum yield. Understanding this mechanistic nuance is vital for any commercial scale-up of complex organic salts, as it dictates the crystallization behavior and final purity of the material. The robust nature of these covalent bonds ensures that the sensor remains intact even in harsh chemical environments, providing reliable data for analytical applications.
How to Synthesize Tetradentate Benzoquinazoline Salt Efficiently
The execution of this synthesis requires precise adherence to reaction parameters to achieve the reported yields and purity levels. The process is divided into distinct stages, each requiring specific attention to temperature, stoichiometry, and workup procedures. From the initial generation of HCl gas for chloromethylation to the final recrystallization from acetonitrile, every step contributes to the overall quality of the high-purity fluorescent material. Operators must ensure that anhydrous conditions are maintained during the thionyl chloride reaction to prevent hydrolysis, and that the quaternization reflux is sustained for sufficient duration to drive the reaction to completion. The detailed标准化 synthesis steps见下方的指南 provide a comprehensive roadmap for laboratory and pilot-scale production, ensuring reproducibility and safety throughout the manufacturing process.
- Chloromethylation of anthracene using paraformaldehyde and HCl gas to form 9,10-dichloromethylanthracene.
- Nucleophilic substitution with diethanolamine to introduce hydroxyethyl arms, followed by chlorination with thionyl chloride.
- Quaternization with N-ethylbenzoquinazoline and subsequent anion exchange with ammonium hexafluorophosphate to yield the final salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain managers, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere technical performance. The reliance on bulk commodity chemicals such as anthracene, paraformaldehyde, and diethanolamine insulates the production process from the volatility associated with specialized reagent markets. This foundational stability translates directly into cost reduction in electronic chemical manufacturing, as the raw material costs are predictable and significantly lower than those of organometallic precursors. Furthermore, the elimination of transition metal catalysts removes the necessity for expensive scavenging resins and complex filtration protocols, streamlining the operational workflow and reducing the total cost of ownership. The simplicity of the unit operations—primarily involving reflux, filtration, and washing—allows for easy integration into existing multipurpose reactor setups without requiring capital-intensive equipment upgrades.
- Cost Reduction in Manufacturing: The synthetic pathway is inherently economical due to the avoidance of precious metal catalysts and the use of high-yield transformations. By eliminating the need for chromatographic purification in favor of simple precipitation and washing steps, the process drastically reduces solvent consumption and labor hours. This efficiency gain allows for a substantial reduction in the variable cost per kilogram, making the final fluorescent sensor competitively priced for widespread deployment in analytical kits and sensing devices. The qualitative improvement in process efficiency ensures that margins are preserved even when scaling to industrial volumes.
- Enhanced Supply Chain Reliability: Sourcing risk is minimized because all key starting materials are widely available from multiple global suppliers. Unlike proprietary intermediates that may be subject to single-source bottlenecks, reagents like thionyl chloride and N-ethylbenzoquinazoline derivatives can be procured with short lead times. This diversity in the supply base ensures continuity of supply, reducing lead time for high-purity fluorescent materials and protecting downstream production schedules from disruption. The robustness of the intermediates also allows for safe storage and transport, further enhancing logistical flexibility.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reaction conditions that do not require extreme pressures or cryogenic temperatures. This makes the technology transfer from laboratory to plant straightforward and low-risk. Additionally, the waste streams generated are primarily aqueous saline solutions and common organic solvents, which are easier to treat and dispose of compared to heavy metal-contaminated waste. This alignment with environmental compliance standards reduces the regulatory burden and associated disposal costs, supporting sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this tetradentate benzoquinazoline salt. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, offering clarity on the material's capabilities and limitations. Understanding these details is essential for integrating this compound into your specific R&D or production workflows effectively.
Q: What is the primary application of this tetradentate benzoquinazoline salt?
A: This compound is primarily designed for fluorescent molecular recognition systems, specifically showing high selectivity for chromium ions (Cr3+) in solution, making it valuable for environmental analysis and chemical sensing.
Q: Why is the hexafluorophosphate (PF6-) counterion used instead of iodide?
A: The anion exchange from iodide to hexafluorophosphate significantly enhances the stability and solubility profile of the salt in organic solvents like acetonitrile, which is critical for its performance in fluorescence spectroscopy applications.
Q: Is this synthesis route suitable for large-scale production?
A: Yes, the process utilizes commercially available starting materials like anthracene and standard reagents such as thionyl chloride and diethanolamine, avoiding exotic catalysts, which facilitates scalable manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetradentate Benzoquinazoline Salt Supplier
The successful implementation of this advanced fluorescent sensor technology requires a manufacturing partner with deep expertise in complex organic synthesis and strict quality control. NINGBO INNO PHARMCHEM stands ready to support your initiatives, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific requirements of quaternization and anion exchange reactions, ensuring that every batch meets stringent purity specifications. With our rigorous QC labs and state-of-the-art analytical instrumentation, we guarantee the consistency and reliability of the final product, enabling you to focus on your core application development without worrying about supply variability.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis to your specific volume and purity requirements. By requesting a Customized Cost-Saving Analysis, you can uncover opportunities to optimize your supply chain further. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that will demonstrate the viability of this partnership for your long-term strategic goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
