Advanced Manufacturing of Hydrolyzable Silane Esters for High-Performance Marine Coatings
Advanced Manufacturing of Hydrolyzable Silane Esters for High-Performance Marine Coatings
The chemical industry continuously seeks robust methodologies for synthesizing functional monomers that balance performance with process safety. Patent CN1396168A introduces a transformative approach for the manufacture of (methyl) acrylic silane esters, a critical class of hydrolyzable monomers extensively utilized in marine anti-pollution and anti-fouling paint formulations. This technology addresses long-standing challenges in the sector by replacing hazardous reagents with a cleaner anhydride exchange protocol. By reacting a (meth)acrylic anhydride with a specific silanized compound, manufacturers can achieve high transformation efficiencies without the evolution of corrosive gases or the generation of solid waste streams. This innovation represents a significant leap forward for any reliable silane ester supplier aiming to meet the stringent environmental and safety standards of modern chemical manufacturing.
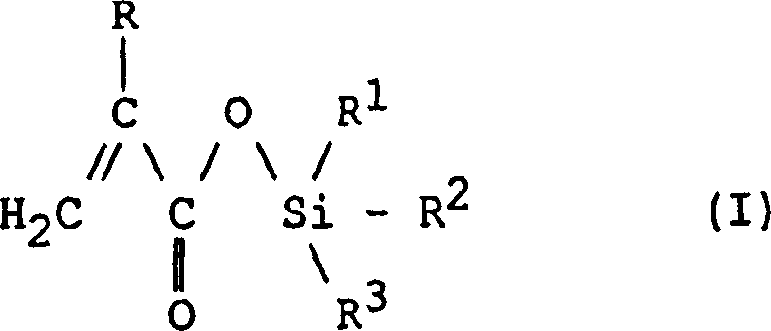
The core value proposition of this patent lies in its ability to produce high-purity intermediates suitable for direct incorporation into polymer matrices. The resulting silane esters serve as essential adhesion promoters and cross-linking agents in coatings that contact seawater, such as ship hulls and offshore structures. The structural versatility allowed by the variable R groups ensures that the material properties can be finely tuned for specific hydrolysis rates and mechanical durability. For procurement teams, understanding this underlying chemistry is vital, as it dictates the purity profile and stability of the final coating product. The method described ensures that the delicate acrylic double bond remains intact during synthesis, preserving the monomer's reactivity for subsequent polymerization steps.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of silyl acrylates has been plagued by significant operational hazards and inefficiencies. Traditional routes often rely on the reaction of acrylic acid or its esters with chlorosilanes, a process that inevitably generates stoichiometric amounts of hydrogen chloride gas. This corrosive byproduct necessitates expensive scrubbing systems and specialized corrosion-resistant equipment, drastically increasing capital expenditure. Furthermore, alternative pathways involving hydrosilylation of acetylenic compounds require precious metal catalysts and pose severe safety risks due to the potential formation of explosive hydrogen gas. Other methods utilizing silver salts or expensive silylating agents introduce heavy metal contamination issues, requiring complex downstream purification steps to meet pharmaceutical or electronic grade standards. These legacy processes are not only environmentally burdensome but also suffer from lower atom economy and higher waste disposal costs.
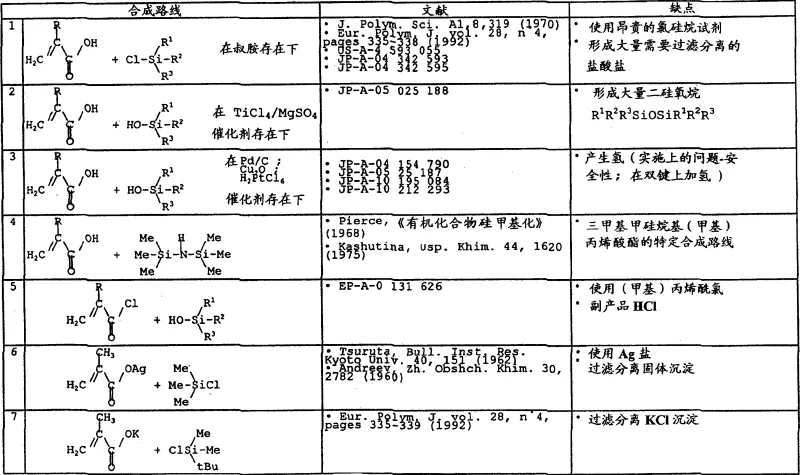
The Novel Approach
The methodology disclosed in CN1396168A circumvents these pitfalls by employing a transesterification-like reaction between a (meth)acrylic anhydride and a silanol or alkoxysilane. This liquid-phase reaction proceeds smoothly under mild thermal conditions, typically between 75°C and 120°C, without the need for exotic catalysts or extreme pressures. The absence of gaseous byproducts means the reaction can be conducted in standard stainless steel reactors equipped with simple stirring and heating capabilities. Moreover, the process avoids the formation of solid precipitates, eliminating the need for energy-intensive filtration, washing, and drying unit operations. The crude reaction mixture can be directly subjected to distillation to isolate the target silane ester, streamlining the workflow and significantly reducing the overall processing time. This shift from hazardous gas-evolving reactions to a contained liquid-phase exchange marks a pivotal improvement in process safety and operational simplicity.
Mechanistic Insights into Anhydride-Silane Exchange Catalysis
The reaction mechanism involves a nucleophilic attack by the oxygen atom of the silanol or alkoxysilane on the carbonyl carbon of the anhydride. This step is facilitated by the presence of basic catalysts such as 1-Methylimidazole, which activate the nucleophile and stabilize the transition state. The cleavage of the anhydride bond results in the formation of the desired silyl ester and a carboxylic acid or ester byproduct, depending on the starting silane. The choice of catalyst is critical; while Lewis acids like Zinc Chloride can be used, nitrogenous bases like 1-Methylimidazole are preferred for their ability to suppress side reactions such as the formation of disiloxanes. The kinetics of this exchange are favorable, allowing for high conversion rates within a reasonable timeframe of 3 to 8 hours. Understanding this mechanistic pathway allows chemists to optimize the molar ratios of reactants, typically maintaining an anhydride excess to drive the equilibrium towards the product side.
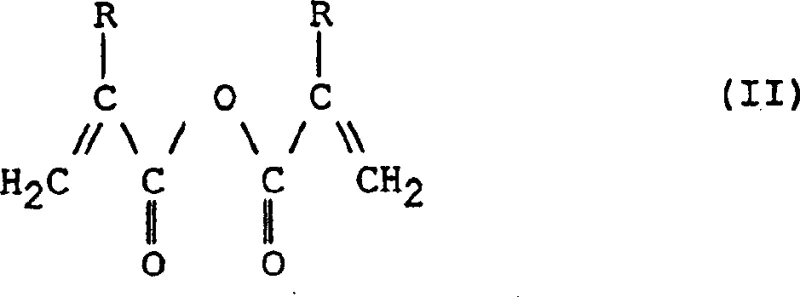
Controlling impurities is paramount in the synthesis of polymerizable monomers to prevent premature curing or discoloration. The patent emphasizes the necessity of conducting the reaction in the presence of radical inhibitors, such as quinhydrones, MEHQ, or sterically hindered phenols like BHT. These additives scavenge free radicals that might otherwise initiate the polymerization of the acrylic double bond at elevated temperatures. Additionally, the process is preferably carried out in the presence of air, which acts as a further safeguard against radical propagation. This dual inhibition strategy ensures that the monomer remains stable throughout the synthesis and subsequent distillation. The resulting product exhibits a clean impurity profile, with minimal formation of silyl oxides or polymeric tars, which is crucial for applications requiring optical clarity or consistent curing behavior in marine coatings.
How to Synthesize (Methyl) Acrylic Silane Ester Efficiently
The synthesis protocol outlined in the patent provides a robust framework for laboratory and pilot-scale production. It begins with the precise charging of methacrylic anhydride and the chosen trialkylalkoxysilane into a reactor equipped with a distillation column. The addition of catalytic amounts of 1-Methylimidazole and radical inhibitors initiates the reaction upon heating. The detailed standardized synthesis steps see the guide below, which outlines the specific temperature ramps and distillation cuts required to achieve pharmaceutical-grade purity. Adhering to these parameters ensures maximum yield while minimizing the degradation of the sensitive acrylic functionality.
- Charge a reactor with (meth)acrylic anhydride, a trialkylalkoxysilane or silanol, and a radical inhibitor such as TOPANOL A or BHT.
- Add a catalytic amount of 1-Methylimidazole or a Lewis acid catalyst and heat the mixture to 75-120°C while bubbling air to maintain an oxidative environment.
- Monitor conversion via gas chromatography until completion, then purify the crude mixture by vacuum distillation to isolate the pure silane ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers tangible benefits beyond mere technical feasibility. The elimination of hazardous gas handling and solid waste treatment translates directly into reduced operational expenditures and lower regulatory compliance burdens. By simplifying the purification train to a single distillation step, manufacturers can achieve faster batch turnover times and higher throughput. This efficiency is critical for maintaining a steady supply of high-purity intermediates in a volatile market. Furthermore, the use of commercially available anhydrides and silanes reduces dependency on specialized, high-cost reagents, thereby stabilizing raw material costs and mitigating supply chain risks associated with niche chemical sourcing.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive corrosion-resistant reactors required for HCl-generating routes and removes the capital cost associated with gas scrubbing systems. By avoiding solid filtration and drying steps, the method significantly reduces energy consumption and labor costs associated with unit operations. The high conversion rates reported in the examples mean less raw material is wasted, improving the overall atom economy of the process. These factors combine to deliver substantial cost savings in marine coating manufacturing, allowing for more competitive pricing strategies without compromising margin.
- Enhanced Supply Chain Reliability: The reactants involved, such as methacrylic anhydride and various alkoxysilanes, are commodity chemicals with well-established global supply chains. This contrasts with proprietary or hazardous reagents that may face shipping restrictions or availability fluctuations. The robustness of the reaction conditions allows for flexible production scheduling, as the process is tolerant to minor variations in feedstock quality. Consequently, suppliers can offer more reliable lead times for high-purity silane esters, ensuring that downstream coating manufacturers can maintain their production schedules without interruption.
- Scalability and Environmental Compliance: Scaling this process from bench to commercial production is straightforward due to the absence of exothermic gas evolution and the use of standard distillation equipment. The liquid-phase nature of the reaction facilitates easy heat transfer and mixing in large vessels. From an environmental perspective, the lack of HCl emissions and heavy metal catalysts simplifies waste management and aligns with increasingly strict green chemistry regulations. This compliance advantage reduces the risk of regulatory fines and enhances the sustainability profile of the final product, a key selling point for eco-conscious end-users in the marine industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this method into their existing manufacturing portfolios.
Q: What are the primary safety advantages of this silane ester synthesis method?
A: Unlike traditional methods using chlorosilanes which evolve corrosive HCl gas, or hydrosilylation which generates explosive hydrogen, this anhydride exchange route produces no hazardous gaseous byproducts, allowing for operation in standard stainless steel reactors without specialized scrubbing systems.
Q: How is polymerization prevented during the high-temperature synthesis?
A: The process mandates the use of radical inhibitors such as quinhydrones, MEHQ, phenothiazine, or sterically hindered phenols like BHT at concentrations of 0.05-0.5wt%, combined with air bubbling to suppress premature curing of the acrylic double bond.
Q: Can this method be scaled for industrial production of marine antifouling agents?
A: Yes, the method eliminates solid filtration steps and uses liquid-phase reactants that are easily handled in bulk. The purification relies on standard distillation techniques suitable for large-scale continuous or batch processing, ensuring high throughput for coating manufacturers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (Methyl) Acrylic Silane Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance monomers play in the development of next-generation marine coatings. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of silane ester meets the exacting standards required for hydrolyzable coating applications. Our commitment to quality assurance means that you can rely on us for materials that deliver consistent performance in the field.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific application needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain efficiency and product quality.
