Industrial Scale Synthesis of S-(-)-2-Methyl-3-Phenyl-1-Propanol via Chemical Resolution
The pharmaceutical and advanced materials industries are constantly seeking robust, scalable pathways for producing high-purity chiral building blocks, particularly those serving as precursors for liquid crystal monomers and complex alkaloid structures. Patent CN109534957B introduces a groundbreaking synthetic methodology for S-(-)-2-methyl-3-phenyl-1-propanol, a critical chiral intermediate that has historically posed significant challenges in terms of enantiomeric separation and industrial feasibility. This patent outlines a comprehensive six-step process that transitions from a simple catalytic hydrogenation of alpha-methyl cinnamaldehyde to a sophisticated chemical resolution strategy, effectively bypassing the limitations of traditional chromatographic separation methods. By leveraging diastereomeric salt formation and recrystallization techniques, this technology offers a viable route for manufacturing ton-scale quantities of this valuable alcohol, addressing the growing demand from R&D teams who require consistent stereochemical integrity for downstream drug synthesis.
Furthermore, the strategic implementation of this synthesis route represents a paradigm shift in how fine chemical manufacturers approach the production of non-natural chiral alcohols. Unlike previous academic studies that merely described the synthesis of the racemic mixture without addressing the critical separation of enantiomers, this invention provides a complete, end-to-end solution that guarantees the isolation of the single S-(-) isomer. The process is designed with industrial scalability in mind, utilizing common catalysts such as palladium on carbon or Raney nickel and readily available resolving agents like R-(+)-alpha-phenylethylamine. For procurement managers and supply chain directors, this translates to a reliable source of high-value intermediates that can be integrated into existing production lines without requiring exotic reagents or specialized, low-throughput equipment, thereby securing the supply chain for critical active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the separation and purification of chiral isomers for compounds like 2-methyl-3-phenyl-1-propanol have been fraught with inefficiencies that hinder large-scale commercialization. Traditional methods often rely heavily on chromatography, which, while effective for analytical purposes or small-batch laboratory synthesis, suffers from inherently small treatment capacities and low operational efficiency. This makes chromatography economically unviable for industrial applications where tonnage production is required to meet global market demands. Additionally, extraction methods are frequently proposed as alternatives, but these are strictly dependent on significant differences in physical properties between the optical isomers. In the case of the target compound, the two enantiomers possess nearly identical physical properties in an achiral environment, rendering standard extraction techniques completely ineffective and leaving manufacturers with no practical way to isolate the desired S-(-) enantiomer from the racemic mixture.
The Novel Approach
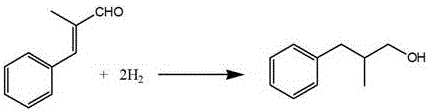
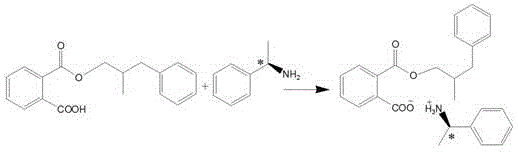
The novel approach detailed in patent CN109534957B circumvents these bottlenecks by employing a chemical resolution strategy that transforms the difficult separation of enantiomers into a manageable separation of diastereomeric salts. The process begins with the catalytic hydrogenation of alpha-methyl cinnamaldehyde to generate the racemic alcohol, followed by a derivatization step using phthalic anhydride to create a mono-ester with a free carboxylic acid group. This functional group serves as a handle for subsequent acid-base reactions with a chiral amine, specifically R-(+)-alpha-phenylethylamine, to form diastereomeric salts. These salts exhibit distinct solubility profiles in specific solvent systems, allowing for the selective crystallization and purification of the target isomer. This method not only ensures high optical purity but also utilizes standard unit operations like filtration and crystallization that are easily scalable in a chemical plant.
This transformative pathway effectively bridges the gap between laboratory discovery and commercial reality, providing a clear blueprint for the mass production of S-(-)-2-methyl-3-phenyl-1-propanol. By avoiding the need for expensive chiral columns or complex simulated moving bed chromatography systems, the new method significantly lowers the barrier to entry for manufacturing this intermediate. The use of phthalic anhydride as a temporary protecting and resolving group is particularly ingenious, as it allows for the eventual recovery of the free alcohol through a straightforward hydrolysis step. This ensures that the final product is obtained in high yield and purity, ready for immediate use in the synthesis of chiral drugs or liquid crystal materials, thus validating the industrial realizability claimed in the patent documentation.
Mechanistic Insights into Chemical Resolution and Diastereomeric Salt Formation
The core of this synthesis lies in the precise manipulation of stereochemistry through diastereomeric salt formation, a classic yet highly effective technique for chiral resolution. After the initial hydrogenation produces the racemic alcohol, the subsequent esterification with phthalic anhydride introduces a carboxylic acid moiety, converting the neutral alcohol into an acidic mono-ester. This structural modification is crucial because it enables the formation of ionic bonds with the chiral base, R-(+)-alpha-phenylethylamine. When the racemic mono-ester reacts with the single-enantiomer amine, two distinct diastereomeric salts are formed: one comprising the S-alcohol derivative and the R-amine, and the other comprising the R-alcohol derivative and the R-amine. Unlike enantiomers, diastereomers have different physical properties, including melting points and, most importantly, solubility in organic solvents.
The separation mechanism relies on the differential solubility of these diastereomeric salts in solvents such as tetrahydrofuran, ethyl acetate, or petroleum ether mixtures. By carefully controlling the temperature and solvent composition during the crystallization phase, the less soluble salt—containing the desired S-(-) configuration—precipitates out of the solution while the unwanted isomer remains in the mother liquor. This crystallization step can be repeated (recrystallization) to further enhance the optical purity, ensuring that the final isolated salt meets stringent quality specifications. Following isolation, the salt undergoes hydrolysis under acidic or basic conditions, which cleaves the ester bond and releases the free S-(-)-2-methyl-3-phenyl-1-propanol while regenerating the phthalic acid and the chiral amine. This regeneration aspect is vital for cost control, as the resolving agent can potentially be recovered and recycled, minimizing waste and reducing the overall consumption of expensive chiral auxiliaries.
How to Synthesize S-(-)-2-Methyl-3-Phenyl-1-Propanol Efficiently
The synthesis of this high-value chiral intermediate requires a disciplined approach to reaction conditions and purification protocols to ensure maximum yield and optical purity. The process initiates with the hydrogenation of alpha-methyl cinnamaldehyde, where parameters such as temperature (0°C to 150°C) and hydrogen pressure (0.1 MPa to 5 MPa) must be tightly controlled to prevent over-reduction or side reactions. Following the formation of the racemic alcohol, the esterification with phthalic anhydride is conducted in solvents like toluene or tetrahydrofuran with amine catalysts, creating the necessary precursor for resolution. The critical resolution step involves mixing the mono-ester with R-(+)-alpha-phenylethylamine, followed by a meticulous crystallization process that may involve cooling to sub-zero temperatures to maximize precipitation of the target diastereomer. Finally, the hydrolysis step liberates the target alcohol, which is then separated from the byproducts using standard extraction or distillation techniques.
- Perform catalytic hydrogenation on alpha-methyl cinnamaldehyde using Pd/C or Raney Nickel to obtain racemic 2-methyl-3-phenyl-1-propanol.
- React the racemic alcohol with phthalic anhydride to form the mono-ester, followed by salt formation with R-(+)-alpha-phenylethylamine.
- Purify the diastereomeric salt via crystallization, then hydrolyze to release the pure S-(-)-enantiomer and recover byproducts.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route offers substantial strategic advantages over conventional sourcing methods. The primary benefit lies in the drastic simplification of the purification process, which eliminates the need for low-throughput chromatographic separations that often act as bottlenecks in production schedules. By shifting to a crystallization-based resolution method, manufacturers can utilize standard reactor vessels and filtration equipment, significantly increasing the batch size and overall throughput. This scalability directly translates to improved supply continuity, as the production capacity is no longer constrained by the limited loading capacity of chiral columns. Furthermore, the reliance on commodity chemicals such as phthalic anhydride and alpha-methyl cinnamaldehyde ensures that raw material availability remains stable, mitigating the risks associated with sourcing specialized or scarce reagents.
- Cost Reduction in Manufacturing: The elimination of expensive chromatography resins and the associated solvent consumption for column operations leads to a significant reduction in variable manufacturing costs. Additionally, the potential for recycling the chiral resolving agent, R-(+)-alpha-phenylethylamine, after the hydrolysis step further drives down the cost of goods sold. The process utilizes robust, non-precious metal catalysts like Raney nickel or standard palladium on carbon for the initial hydrogenation, avoiding the high costs associated with exotic homogeneous catalysts. These factors combine to create a highly cost-effective production model that allows for competitive pricing in the global market for chiral intermediates.
- Enhanced Supply Chain Reliability: The use of widely available industrial raw materials ensures that the supply chain is resilient against disruptions caused by the scarcity of niche chemicals. Since the process does not depend on single-source suppliers for specialized chiral catalysts or columns, procurement teams can diversify their vendor base for inputs, enhancing overall security of supply. The robustness of the chemical resolution method also means that production can be easily scaled up or down based on market demand without requiring major capital investment in new equipment. This flexibility allows suppliers to respond rapidly to fluctuations in demand from the pharmaceutical and liquid crystal sectors, ensuring consistent delivery performance.
- Scalability and Environmental Compliance: The transition from chromatography to crystallization significantly reduces the volume of organic solvent waste generated per kilogram of product, aligning with modern green chemistry principles and environmental regulations. Crystallization processes generally have a lower environmental footprint compared to continuous elution methods, simplifying waste treatment and disposal protocols. Moreover, the ability to run larger batches in standard reactors improves energy efficiency per unit of product, contributing to a more sustainable manufacturing profile. This environmental compliance is increasingly important for multinational corporations aiming to reduce their carbon footprint and meet strict sustainability goals in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of S-(-)-2-methyl-3-phenyl-1-propanol based on the disclosed patent technology. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on the feasibility and advantages of this specific route. Understanding these details is essential for technical teams evaluating the integration of this intermediate into their own synthesis pipelines or for procurement specialists assessing supplier capabilities.
Q: Why is chemical resolution preferred over chromatography for this chiral alcohol?
A: Chromatography has limited treatment capacity and low efficiency, making it unsuitable for industrial-scale production. Chemical resolution via diastereomeric salt formation allows for high-volume processing and easier purification through crystallization.
Q: What are the key raw materials required for this synthesis?
A: The process utilizes commercially available and industrially supplied raw materials including alpha-methyl cinnamaldehyde, hydrogen, phthalic anhydride, and R-(+)-alpha-phenylethylamine, ensuring robust supply chain continuity.
Q: How is the chiral purity achieved in this patent method?
A: Chiral purity is achieved through the formation of diastereomeric salts using R-(+)-alpha-phenylethylamine. The specific solubility differences between the salts allow for the selective crystallization and isolation of the desired S-(-) isomer.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-(-)-2-Methyl-3-Phenyl-1-Propanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity chiral intermediates play in the development of next-generation pharmaceuticals and advanced materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global clients. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of S-(-)-2-methyl-3-phenyl-1-propanol meets the highest standards of optical and chemical purity required for sensitive downstream applications. Our infrastructure is designed to support the complex resolution and hydrolysis steps outlined in patent CN109534957B, providing a secure and reliable source for this valuable building block.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced synthesis capabilities can support your long-term strategic goals in the fine chemical and pharmaceutical sectors.
