Advanced Manufacturing of Silanized Acrylates for High-Performance Marine Anti-Fouling Coatings
Advanced Manufacturing of Silanized Acrylates for High-Performance Marine Anti-Fouling Coatings
The development of hydrolyzable monomers for marine anti-fouling coatings represents a critical frontier in the advanced materials sector, where durability and environmental compliance are paramount. Patent CN1280295C introduces a robust and commercially viable methodology for the synthesis of silanized (meth)acrylates, specifically targeting the production of high-performance binders used in self-polishing copolymer paints. This technology addresses long-standing inefficiencies in traditional esterification processes by utilizing a direct reaction between (meth)acrylic anhydride and organosilicon compounds. For R&D directors and procurement specialists seeking a reliable silicone materials supplier, understanding the mechanistic elegance and operational simplicity of this anhydride-based route is essential for securing a competitive edge in the global coatings market.
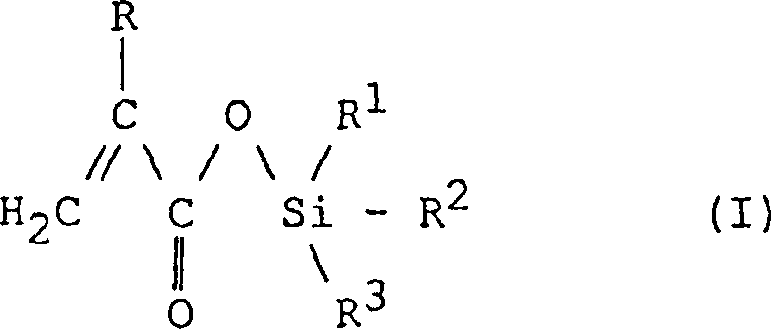
The core innovation lies in the specific structural configuration of the resulting silane ester, which allows for controlled hydrolysis in seawater, thereby releasing biocides at a steady rate while the coating surface erodes smoothly. The patent delineates a general formula where the silicon atom is bonded to three organic groups and one oxygen atom linked to the acrylic backbone. This precise molecular architecture ensures that the polymer matrix maintains its integrity until exposed to the specific pH and ionic conditions of the marine environment. By mastering the synthesis of these complex intermediates, manufacturers can offer clients superior products that meet stringent international maritime regulations regarding antifouling efficacy and ecological safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of silyl esters has been plagued by significant engineering challenges and safety hazards associated with traditional acylation agents. Conventional routes often rely on the reaction of silanols with acid chlorides, a process that inevitably generates stoichiometric amounts of corrosive hydrogen chloride gas as a byproduct. This necessitates the use of specialized glass-lined reactors and extensive gas scrubbing infrastructure to handle the toxic emissions, drastically increasing capital expenditure and operational complexity. Furthermore, alternative methods involving transesterification often suffer from equilibrium limitations, requiring the continuous removal of alcohol byproducts to drive the reaction forward, which complicates the process control and reduces overall throughput efficiency in large-scale manufacturing facilities.
The Novel Approach
In stark contrast, the methodology described in the patent utilizes (meth)acrylic anhydride as the acylating agent, fundamentally altering the reaction profile to eliminate gaseous byproducts entirely. This approach allows the reaction to proceed safely in standard stainless steel reactors equipped with simple stirring and heating mechanisms, removing the need for corrosion-resistant linings. The reaction produces a carboxylic acid as the sole byproduct, which remains in the liquid phase and can be easily separated from the target silane ester via distillation due to differences in volatility. This shift from gas-generating chemistry to a clean liquid-phase transformation significantly simplifies the downstream processing units, reducing both the energy consumption and the maintenance burden associated with handling aggressive chemical waste streams.
Mechanistic Insights into Anhydride-Mediated Silyl Esterification
The reaction mechanism involves a nucleophilic attack by the oxygen atom of the silanol or alkoxysilane on the carbonyl carbon of the acrylic anhydride. This process is facilitated by the electron-withdrawing nature of the carbonyl group, which makes the central carbon highly susceptible to nucleophilic substitution. In the presence of a basic catalyst such as 1-Methylimidazole, the kinetics of this acylation are significantly accelerated, allowing the reaction to reach completion within a practical timeframe of 3 to 8 hours at moderate temperatures ranging from 80 to 120°C. The catalyst functions by activating the anhydride or deprotonating the silanol intermediate, thereby lowering the activation energy barrier and ensuring high conversion rates exceeding 96% under optimized conditions.
From an impurity control perspective, the formation of disiloxanes (silyl oxides) is a potential side reaction that can compromise the quality of the final monomer. However, the patent highlights that the use of specific catalysts and the maintenance of an inert or air-bubbled atmosphere effectively suppress the condensation of silanol groups into siloxanes. Additionally, the inclusion of polymerization inhibitors like MEHQ or BHT is critical to prevent the premature radical polymerization of the methacrylate double bond during the thermal process. This dual strategy of kinetic acceleration and radical suppression ensures that the crude reaction mixture contains predominantly the desired monomer and the separable acid byproduct, facilitating a straightforward purification protocol that yields a product with exceptional chemical purity suitable for sensitive coating applications.
How to Synthesize Tributylsilyl Methacrylate Efficiently
The synthesis of tributylsilyl methacrylate serves as a prime example of the versatility and efficiency of this anhydride-based protocol. The process begins by charging a reactor with high-purity methacrylic anhydride and tributylmethoxysilane in a molar ratio of approximately 1.1:1, ensuring a slight excess of the anhydride to drive the equilibrium towards the product. Polymerization inhibitors such as TOPANOL A and BHT are added to the mixture to stabilize the reactive double bonds against thermal initiation, while a catalytic amount of 1-Methylimidazole is introduced to enhance the reaction rate. The detailed standardized synthesis steps for optimizing yield and purity are outlined in the guide below.
- Charge a reactor with methacrylic anhydride and the corresponding trialkylalkoxysilane or trialkylsilanol, maintaining a molar ratio between 0.9: 1 and 1.2:1.
- Add a polymerization inhibitor such as MEHQ or BHT and optionally a catalyst like 1-Methylimidazole to accelerate kinetics.
- Heat the mixture to 80-120°C under air bubbling for 3-8 hours until conversion exceeds 96%, followed by vacuum distillation to isolate the pure ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this anhydride-based synthesis route offers profound strategic benefits that extend beyond mere chemical yield. The elimination of corrosive gas byproducts translates directly into reduced capital investment for plant infrastructure, as facilities no longer require expensive acid-gas scrubbing towers or glass-lined reactors. This simplification of the hardware requirements allows for faster deployment of production capacity and lowers the barrier to entry for manufacturing these high-value intermediates. Furthermore, the reliance on readily available raw materials like methacrylic anhydride and common alkoxysilanes ensures a stable and resilient supply chain that is less susceptible to the bottlenecks often associated with specialized acyl chlorides.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by removing the need for complex waste treatment systems associated with hydrogen chloride neutralization. Since the byproduct is a liquid carboxylic acid that can potentially be recycled or sold as a commodity chemical, the overall material efficiency of the plant is markedly improved. Additionally, the ability to operate at atmospheric pressure without the need for high-vacuum systems during the reaction phase reduces energy consumption and equipment maintenance costs, leading to a lower cost of goods sold for the final silane ester product.
- Enhanced Supply Chain Reliability: By utilizing a synthetic route that avoids hazardous gas evolution, the manufacturing process becomes inherently safer and more robust against regulatory shutdowns. The use of standard stainless steel equipment means that production can be easily scaled or transferred between different manufacturing sites without requiring specialized retrofitting. This flexibility ensures consistent delivery schedules for downstream coating manufacturers, mitigating the risks of supply disruptions that can occur with more fragile or hazardous chemical processes.
- Scalability and Environmental Compliance: The absence of solid filtration steps and the simplicity of the purification via distillation make this process ideally suited for large-scale commercial production. The environmental footprint is minimized due to the lack of acidic gas emissions and the reduction in solvent usage for washing steps. This aligns perfectly with modern green chemistry principles and helps end-users in the marine coatings industry meet increasingly strict environmental regulations regarding volatile organic compounds and hazardous waste generation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of silanized acrylates using this patented technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating these monomers into new coating formulations.
Q: What are the primary advantages of using acid anhydrides over acid chlorides for silane ester synthesis?
A: Using acid anhydrides eliminates the generation of corrosive hydrogen chloride (HCl) gas, removing the need for expensive gas scrubbing systems and allowing the use of standard stainless steel reactors instead of glass-lined vessels.
Q: How is the purity of the final silanized methacrylate ensured in this process?
A: The process yields a crude mixture containing the target ester and a heavier carboxylic acid byproduct; high purity is achieved through simple fractional distillation or thin-film evaporation which separates the volatile ester from higher boiling impurities.
Q: Can this synthesis method be scaled for industrial production of marine coating additives?
A: Yes, the method operates at atmospheric pressure with moderate temperatures (75-120°C) and avoids solid filtration steps, making it highly scalable and suitable for continuous or large-batch commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Silanized Methacrylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity silane monomers play in the performance of next-generation marine antifouling systems. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global coating manufacturers. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of silanized methacrylate meets the exacting standards required for hydrolyzable binder applications, delivering consistent quality that protects your brand reputation.
We invite you to collaborate with us to leverage this advanced anhydride-based technology for your specific project needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and reduce your overall material costs.
