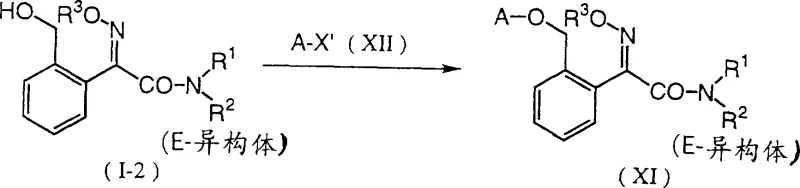Advanced Synthesis of 2-(2-Hydroxymethylphenyl)acetamide Derivatives for High-Efficiency Fungicide Production
Advanced Synthesis of 2-(2-Hydroxymethylphenyl)acetamide Derivatives for High-Efficiency Fungicide Production
The global demand for high-efficacy agricultural fungicides continues to drive innovation in intermediate synthesis, particularly for alkoxyiminoacetamide derivatives which serve as critical precursors. Patent CN1175244A discloses a groundbreaking preparation method for 2-(2-hydroxymethylphenyl)acetamide derivatives that addresses long-standing challenges in stereochemical control and process efficiency. This technology enables the production of the biologically active E-isomer with exceptional purity by integrating a deprotection step with a simultaneous isomerization reaction. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, understanding this mechanistic breakthrough is essential for securing a competitive supply chain. The patent outlines a robust pathway starting from protected benzyl halides, proceeding through Grignard acylation, and culminating in a clever acid-catalyzed transformation that eliminates the need for tedious isomer separation.
Traditional synthetic routes for these valuable intermediates often suffer from poor stereocontrol, requiring additional purification steps to enrich the desired E-isomer, which drastically increases manufacturing costs and waste generation. Conventional methods typically involve the conversion of a monochloromethyl group to a hydroxymethyl group at a later stage, or they fail to control the geometry of the oxime double bond effectively during the initial coupling. These legacy processes frequently result in mixtures of E and Z isomers that necessitate expensive chromatographic separation or recrystallization protocols, thereby limiting their viability for commercial scale-up of complex agrochemical intermediates. Furthermore, the harsh conditions sometimes required for late-stage functional group interconversion can degrade sensitive moieties, leading to lower overall yields and impurity profiles that are difficult to manage in a GMP-compliant environment.
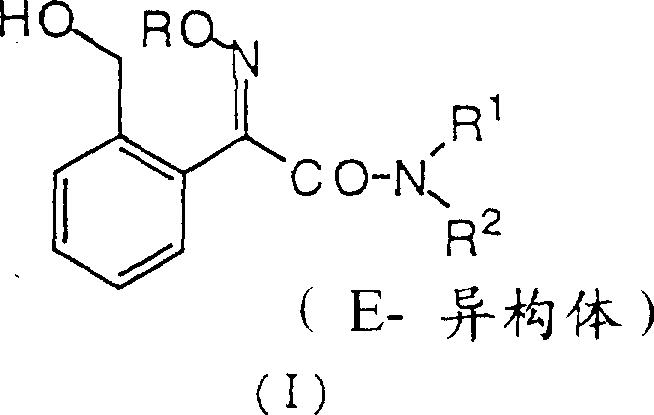
In stark contrast, the novel approach detailed in the patent leverages a strategic protection-deprotection sequence that inherently favors the formation of the thermodynamically stable E-isomer. By employing a hydroxyl protecting group (P) such as a tetrahydropyranyl or ethoxyethyl moiety during the early synthetic stages, the molecule remains stable during the formation of the Grignard reagent and subsequent acylation. The true innovation lies in the final step, where acidic treatment removes the protecting group and concurrently drives the isomerization of the oxime functionality to the E-configuration. This tandem reaction not only simplifies the workflow by removing a discrete isomerization unit operation but also significantly enhances the purity of the final product, directly contributing to cost reduction in agricultural fungicide manufacturing.
Mechanistic Insights into Grignard Acylation and Acid-Catalyzed Isomerization
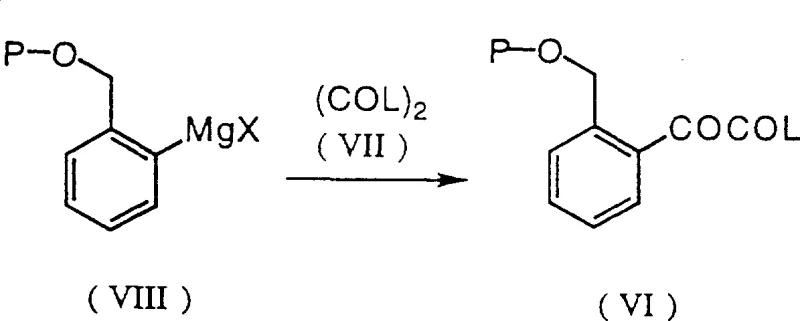
The core of this synthesis relies on the precise execution of a Grignard reaction followed by an electrophilic capture with an oxalic acid derivative. As illustrated in the reaction schemes, the process begins with the formation of an organomagnesium species from a protected 2-bromobenzyl derivative. This step requires careful temperature control, typically between -70°C and -50°C, to prevent Wurtz-type coupling side reactions while ensuring complete metal insertion. The resulting Grignard reagent is then reacted with a dialkyl oxalate or oxalyl halide to generate an alpha-keto ester or acid halide intermediate. This electrophilic carbonyl addition is highly exothermic and demands rigorous thermal management to maintain the integrity of the keto-ester linkage, which is pivotal for the subsequent amidation step.
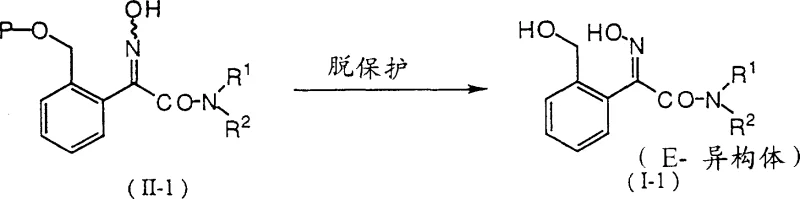
Following the acylation, the intermediate is converted into the amide using a primary or secondary amine, establishing the nitrogen framework required for the final fungicide structure. The subsequent oximation introduces the critical imino ether functionality. However, the most chemically intriguing aspect is the final deprotection mechanism. When the protected intermediate is treated with an acid catalyst—such as p-toluenesulfonic acid or hydrochloric acid—the protecting group is cleaved to reveal the free hydroxymethyl group. Simultaneously, the acidic environment facilitates the equilibration of the oxime double bond, driving the equilibrium heavily towards the E-isomer. This phenomenon suggests that the E-isomer is thermodynamically favored under these specific acidic conditions, allowing manufacturers to obtain high-purity material without the need for complex downstream processing, thereby ensuring high-purity agrochemical intermediates suitable for direct formulation.
How to Synthesize 2-(2-Hydroxymethylphenyl)acetamide Derivatives Efficiently
The synthesis of these high-value intermediates requires a disciplined approach to reaction conditions and reagent quality to ensure consistent batch-to-batch reproducibility. The process outlined in the patent provides a clear roadmap for transitioning from laboratory scale to pilot plant operations, emphasizing the importance of maintaining anhydrous conditions during the Grignard formation and controlling the acidity during the final deprotection. Operators must strictly adhere to the specified temperature ranges and stoichiometric ratios to minimize byproduct formation. For a detailed breakdown of the operational parameters, reagent grades, and workup procedures necessary to implement this technology, please refer to the standardized synthesis guide below.
- Prepare the protected benzyl halide intermediate by reacting 2-bromobenzyl alcohol with a protecting group agent like ethyl vinyl ether or dihydropyran under acidic catalysis.
- Form the Grignard reagent from the protected halide and react it with an oxalic acid derivative (e.g., diethyl oxalate) at low temperatures (-60°C to -78°C) to form the alpha-keto ester.
- Convert the ester to the amide using an amine, followed by oximation with O-alkylhydroxylamine, and finally perform acid-catalyzed deprotection which simultaneously drives E-isomer formation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers substantial strategic benefits beyond mere technical elegance. By consolidating the deprotection and isomerization steps, the process inherently reduces the total number of unit operations required, which translates directly into lower capital expenditure and reduced operational overhead. The elimination of a dedicated isomer separation step means less solvent consumption, reduced waste disposal costs, and shorter cycle times, all of which contribute to a more lean and efficient manufacturing model. This streamlined workflow enhances the reliability of supply by minimizing the risk of bottlenecks associated with complex purification stages, ensuring a steady flow of materials for downstream fungicide production.
- Cost Reduction in Manufacturing: The integration of deprotection and isomerization into a single pot operation significantly lowers processing costs by reducing energy consumption and labor hours. Since the process avoids the use of expensive transition metal catalysts or specialized chiral auxiliaries, the raw material costs remain competitive. Furthermore, the high selectivity for the E-isomer reduces the loss of valuable material during purification, maximizing the yield per kilogram of starting material and driving down the overall cost of goods sold.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as magnesium, oxalates, and common mineral acids ensures that the supply chain is robust and less susceptible to disruptions caused by the scarcity of exotic reagents. The stability of the protected intermediates allows for flexible production scheduling, as key precursors can be stockpiled without significant degradation. This flexibility empowers supply chain planners to respond rapidly to fluctuations in market demand for agricultural fungicides without compromising on quality or lead times.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reactor configurations and well-understood chemical transformations that are easily transferred from pilot to commercial scale. The reduction in solvent usage and waste generation aligns with modern green chemistry principles, facilitating easier compliance with increasingly stringent environmental regulations. This sustainability profile not only mitigates regulatory risk but also enhances the corporate social responsibility standing of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these intermediates. The answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is critical for partners looking to integrate this technology into their existing production portfolios or for those evaluating the technical feasibility of new product lines.
Q: Why is the E-isomer preferred in alkoxyiminoacetamide fungicides?
A: The E-isomer typically exhibits significantly higher fungicidal activity compared to the Z-isomer. The patented process uniquely combines deprotection with isomerization to maximize the yield of the biologically active E-form without requiring a separate purification step.
Q: What are the advantages of using a protected hydroxymethyl group in this synthesis?
A: Using a protected group (such as tetrahydropyranyl or ethoxyethyl) prevents side reactions during the vigorous Grignard formation and acylation steps. It ensures the integrity of the hydroxymethyl functionality until the final stage, improving overall purity and yield.
Q: Can this process be scaled for commercial agrochemical production?
A: Yes, the process utilizes standard industrial reagents like magnesium, oxalates, and common acids for deprotection. The elimination of complex separation steps for isomers makes it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2-Hydroxymethylphenyl)acetamide Derivative Supplier
The technological advancements described in patent CN1175244A represent a significant leap forward in the efficient production of agrochemical intermediates, offering a pathway to higher purity and lower costs. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such complex synthetic routes into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of 2-(2-hydroxymethylphenyl)acetamide derivative meets the exacting standards required for high-performance fungicide formulations.
We invite you to collaborate with us to leverage this innovative synthesis method for your specific applications. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized processes can enhance your bottom line. Please contact our technical procurement team today to request specific COA data and discuss route feasibility assessments for your next project, ensuring a partnership built on quality, reliability, and mutual growth.