Advanced C2-Symmetric Bisboron Catalysts for Scalable Chiral Amine Production
The pharmaceutical and fine chemical industries are currently witnessing a paradigm shift in asymmetric synthesis, driven by the urgent need to eliminate expensive and toxic precious metal residues from active pharmaceutical ingredients. Patent CN109046447B introduces a groundbreaking methodology for the synthesis of C2-symmetric bicyclic bisboron catalysts, which offer a robust, metal-free alternative for the asymmetric hydrogenation of aryl ketimines. This technology addresses critical pain points in modern drug manufacturing by providing a pathway to chiral secondary amines that is not only highly enantioselective but also economically superior to traditional transition metal-catalyzed processes. By leveraging the unique Lewis acidity of boron centers within a rigid bicyclic framework, this invention enables the activation of molecular hydrogen under mild conditions, thereby opening new avenues for sustainable and cost-effective production of high-value chiral intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric hydrogenation of imines has relied heavily on noble metal complexes based on rhodium, iridium, or ruthenium, which present substantial logistical and regulatory burdens for commercial manufacturers. These traditional catalysts often require stringent exclusion of air and moisture, specialized high-pressure equipment, and extensive downstream purification steps to ensure that residual metal levels comply with strict ICH Q3D guidelines for pharmaceutical products. Furthermore, the synthesis of chiral ligands for these metal complexes is frequently multi-step, low-yielding, and dependent on scarce natural chiral pools, leading to volatile pricing and supply chain instability for key starting materials. The environmental footprint of mining and refining these precious metals also conflicts with the growing industry mandate for greener chemistry, making the continued reliance on such systems increasingly untenable for large-scale operations.
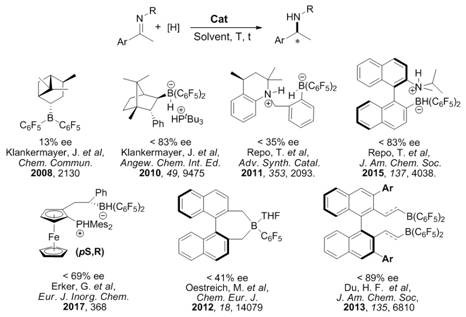
The Novel Approach
In stark contrast, the novel C2-symmetric bisboron catalyst system described in the patent data utilizes abundant non-metallic elements to achieve comparable or superior stereocontrol without the associated baggage of heavy metal chemistry. The synthetic route to the catalyst itself is remarkably streamlined, involving a direct hydroboration of readily available dienes followed by a simple coordination step with isoquinoline, which can be completed in minutes at ambient or moderately elevated temperatures. This simplicity translates directly into process robustness, as the catalyst preparation does not require inert atmosphere gloveboxes for extended periods or cryogenic conditions, thereby reducing energy consumption and operational complexity. The resulting catalysts exhibit high stability and can be tuned structurally by modifying the aryl substituents on the diene precursor, offering a versatile platform for optimizing selectivity across a broad range of imine substrates.
Mechanistic Insights into C2-Symmetric Bisboron Catalysis
The core mechanistic advantage of this technology lies in the precise spatial arrangement of the two boron centers within the bicyclic framework, which creates a well-defined chiral pocket for substrate binding and hydrogen activation. As illustrated in the reaction scheme below, the catalyst operates through a frustrated Lewis pair (FLP) type mechanism where the boron centers cooperate to heterolytically cleave the H-H bond, generating a reactive hydridoborate species that transfers hydride to the imine carbon. The C2 symmetry ensures that the approach of the substrate is highly restricted to one facial direction, resulting in the observed high enantiomeric excess values, often exceeding 90% ee in optimized conditions. This level of stereochemical control is achieved without the need for external chiral additives, as the chirality is intrinsic to the rigid backbone of the catalyst scaffold formed during the initial hydroboration step.
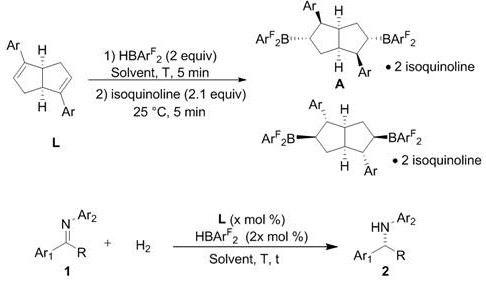
From an impurity control perspective, the metal-free nature of this catalytic system fundamentally alters the purification profile of the final drug substance, eliminating the need for specialized scavenger resins or recrystallization steps designed to remove trace metals. In traditional processes, metal residues can catalyze degradation pathways during storage or interact with biological targets, necessitating rigorous and costly analytical testing at every stage of production. With the bisboron catalyst, the primary impurities are organic byproducts that are generally easier to separate via standard chromatographic or crystallization techniques, leading to a cleaner overall mass balance and higher throughput. Additionally, the absence of redox-active metals reduces the risk of uncontrolled exotherms or side reactions such as over-reduction, enhancing the safety profile of the manufacturing process and simplifying hazard assessments for plant operations.
How to Synthesize C2-Symmetric Bicyclic Diboron Catalysts Efficiently
The synthesis of these high-performance catalysts is designed for operational simplicity, allowing chemical engineers to produce the active species in situ or as isolated solids with minimal training or specialized infrastructure. The process begins with the careful selection of the diene precursor, where the steric bulk of the aryl groups determines the final cavity size of the catalyst, followed by a rapid hydroboration reaction that sets the stereochemistry. Detailed standardized synthesis steps see the guide below, which outlines the precise stoichiometry and temperature profiles required to maximize yield and selectivity. This straightforward protocol ensures that the technology can be transferred seamlessly from laboratory discovery to pilot plant production without the need for extensive re-optimization.
- Under nitrogen atmosphere, sequentially add diene L, hydroboron reagent, and toluene solvent into a reaction vessel.
- Control the hydroboration reaction temperature precisely (e.g., 25°C or 80°C) for 5 minutes to dictate the catalyst structure.
- Cool to room temperature, add isoquinoline, stir, remove solvent, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this boron-based catalytic technology represents a strategic opportunity to de-risk the supply of critical chiral intermediates while simultaneously driving down the cost of goods sold. By removing the dependency on platinum group metals, manufacturers can insulate themselves from the geopolitical volatility and price fluctuations that characterize the precious metals market, ensuring more predictable budgeting and long-term contract stability. The simplified synthesis of the catalyst itself means that it can be produced in-house or sourced from a wider range of chemical suppliers, reducing single-source dependency and enhancing overall supply chain resilience against disruptions. Furthermore, the reduced regulatory burden associated with metal-free processes accelerates the timeline for regulatory filings and approvals, allowing new drugs to reach the market faster and with lower compliance costs.
- Cost Reduction in Manufacturing: The elimination of expensive precious metal salts and complex chiral ligands results in a drastic reduction in raw material costs, while the simplified purification train lowers utility and waste disposal expenses significantly. Without the need for metal scavenging agents or extensive washing protocols to meet residual metal specifications, the overall processing time per batch is shortened, increasing asset utilization and throughput capacity. This economic efficiency is compounded by the high turnover numbers of the catalyst, which allow for lower catalyst loading rates while maintaining high conversion, further driving down the variable cost per kilogram of the final product.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, such as substituted dienes and hydroboranes, are commodity chemicals with robust global supply chains, unlike specialized metal complexes that may have limited vendors. This abundance ensures that production schedules are not held hostage by the lead times of niche reagent suppliers, facilitating just-in-time manufacturing strategies and reducing inventory holding costs. The stability of the catalyst precursors also allows for longer shelf life and easier transportation, minimizing the risk of spoilage or degradation during logistics and warehousing operations.
- Scalability and Environmental Compliance: The reaction conditions described in the patent operate at near-ambient pressures and moderate temperatures, making them inherently safer and easier to scale in standard glass-lined or stainless steel reactors without requiring specialized high-pressure autoclaves. The absence of toxic heavy metals simplifies wastewater treatment and solid waste disposal, aligning with increasingly stringent environmental regulations and corporate sustainability goals. This green chemistry profile not only reduces the environmental footprint but also enhances the brand reputation of the pharmaceutical company among stakeholders who prioritize eco-friendly manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel catalytic system in industrial settings. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide accurate guidance for process development teams. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into existing production lines.
Q: How does this boron catalyst compare to traditional precious metal catalysts?
A: Unlike traditional rhodium or iridium catalysts, this C2-symmetric bisboron system eliminates heavy metal contamination risks, simplifies purification, and significantly reduces raw material costs while maintaining high enantioselectivity.
Q: Is this catalytic system suitable for large-scale industrial production?
A: Yes, the patent demonstrates gram-scale experiments with high yields (up to 96%) and simple operational conditions, indicating strong potential for commercial scale-up without complex pressure or temperature requirements.
Q: What represents the key innovation in the catalyst synthesis method?
A: The core innovation lies in controlling the hydroboration reaction temperature to selectively produce different structural isomers of the diboron catalyst, allowing for tunable stereoselectivity in the final amine products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Secondary Amines Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free catalytic technologies like the C2-symmetric bisboron system in reshaping the landscape of chiral amine synthesis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are translated into robust, GMP-compliant manufacturing processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications and enantiomeric excess values, guaranteeing that every batch meets the exacting standards required by global regulatory agencies. We are committed to helping our partners leverage this advanced chemistry to achieve superior product quality and operational efficiency.
We invite you to engage with our technical procurement team to discuss how this catalyst technology can be tailored to your specific synthetic challenges and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits of switching to this metal-free route for your key intermediates. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will strengthen your supply chain and enhance your competitive advantage in the marketplace.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
