Advanced Synthesis of Octahydrobenzonaphthyridine Compounds for Commercial Scale-up of Complex Pharmaceutical Intermediates
Advanced Synthesis of Octahydrobenzonaphthyridine Compounds for Commercial Scale-up of Complex Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks novel chemical entities with improved pharmacokinetic profiles, and patent CN108299429B introduces a groundbreaking class of octahydrobenzonaphthyridine compounds that address critical challenges in antitumor drug development. This patent discloses a robust synthetic methodology for producing these complex heterocyclic structures, which exhibit significantly enhanced biological activity compared to their oxidized precursors. The core innovation lies in the strategic double reduction of the benzonaphthyridine skeleton, transforming less soluble amide-ketone structures into highly bioavailable amine derivatives. For R&D directors and procurement specialists, this represents a pivotal opportunity to access high-purity pharmaceutical intermediates that offer superior druggability. The detailed chemical pathway provided in the patent ensures that manufacturers can replicate these results with high fidelity, paving the way for reliable supply chains of next-generation anticancer agents.
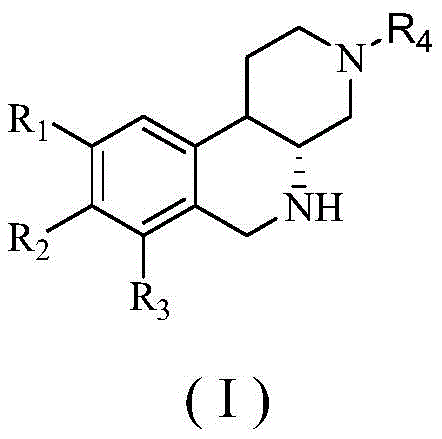
The structural versatility of these compounds, as defined by the general formula (I), allows for extensive modification at the R1, R2, R3, and R4 positions, enabling medicinal chemists to fine-tune potency and selectivity. The patent explicitly demonstrates that varying these substituents from simple alkyl groups to complex aralkyl moieties does not compromise the core synthetic efficiency. This flexibility is crucial for developing a diverse library of analogs during the lead optimization phase of drug discovery. Furthermore, the conversion from an amide to an amine functionality drastically alters the physicochemical properties, specifically enhancing aqueous solubility which is often a bottleneck in oral drug formulation. By adopting this technology, pharmaceutical companies can accelerate their pipeline development with compounds that possess both high intrinsic activity and favorable ADMET properties.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for benzonaphthyridine derivatives often suffer from harsh reaction conditions, low overall yields, and the generation of difficult-to-remove impurities that complicate downstream processing. Conventional methods frequently rely on multiple protection and deprotection steps that increase the step count and reduce the atom economy of the process. Additionally, many existing protocols utilize expensive transition metal catalysts or hazardous reagents that pose significant safety risks and environmental compliance burdens during commercial scale-up. The presence of residual heavy metals in the final active pharmaceutical ingredient (API) is a major regulatory concern, requiring costly purification steps to meet stringent international standards. These inefficiencies translate directly into higher manufacturing costs and longer lead times, creating substantial bottlenecks for supply chain managers trying to meet market demand for novel oncology therapeutics.
The Novel Approach
The novel approach detailed in patent CN108299429B overcomes these historical limitations through a streamlined, convergent synthesis strategy that maximizes yield while minimizing waste. By utilizing a Michael addition followed by a condensation reaction with formaldehyde and primary amines, the process efficiently constructs the piperidinone core with high stereocontrol. The subsequent cyclization step employs trifluoromethanesulfonic anhydride, a potent activating agent that drives the ring closure under mild conditions, thereby preserving sensitive functional groups on the aromatic rings. This method eliminates the need for toxic heavy metal catalysts, significantly simplifying the purification workflow and reducing the environmental footprint of the manufacturing process. The result is a scalable, cost-effective route that delivers high-purity intermediates suitable for direct progression into clinical trial material production.
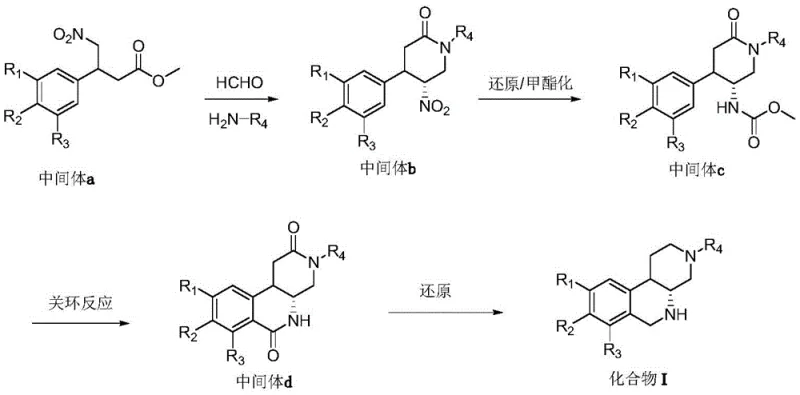
Mechanistic Insights into Trifluoromethanesulfonic Anhydride-Mediated Cyclization
The heart of this synthetic innovation is the intramolecular cyclization step that forms the fused tetracyclic system, a transformation that requires precise control over reaction kinetics and thermodynamics. The mechanism involves the activation of the carbamate carbonyl group by trifluoromethanesulfonic anhydride in the presence of DMAP, generating a highly reactive electrophilic species. This activated intermediate undergoes a nucleophilic attack by the electron-rich aromatic ring, facilitated by the ortho-positioning of the methoxy or alkoxy substituents which enhance the nucleophilicity of the benzene ring. The reaction proceeds through a Wheland intermediate before rearomatization yields the stable hexahydro-3,5-diazabenzo[c]naphthalene-2,6-dione scaffold. Understanding this mechanistic nuance is vital for process chemists, as it highlights the importance of maintaining anhydrous conditions and precise stoichiometry to prevent hydrolysis of the activated species.
Following the cyclization, the final reduction step utilizes lithium aluminum hydride to simultaneously reduce both the lactam and ketone functionalities to their corresponding amine and methylene groups. This double reduction is critical for unlocking the enhanced biological activity described in the patent, as it converts the rigid, planar amide structure into a more flexible, three-dimensional amine conformation. The use of LiAlH4 in anhydrous THF at elevated temperatures ensures complete conversion, although careful quenching is required to manage the exothermic nature of the reaction. From an impurity control perspective, this step is robust because the reduction potential of LiAlH4 is sufficiently high to drive the reaction to completion, minimizing the risk of partially reduced byproducts that could act as genotoxic impurities in the final drug substance.
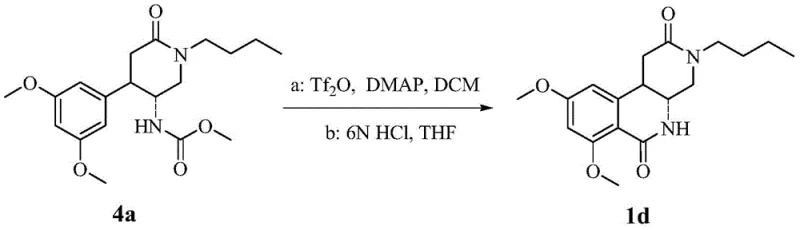
How to Synthesize Octahydrobenzonaphthyridine Efficiently
The synthesis of these high-value intermediates requires strict adherence to the optimized reaction parameters outlined in the patent to ensure consistent quality and yield. The process begins with the preparation of the nitro-piperidinone scaffold, followed by selective reduction and protection, before proceeding to the critical cyclization and final reduction stages. Each step has been engineered to balance reaction rate with selectivity, ensuring that the final product meets the rigorous purity specifications demanded by the pharmaceutical industry. Operators must pay close attention to temperature control and reagent addition rates, particularly during the exothermic reduction phases, to maintain safety and product integrity. The detailed standardized synthesis steps provided below serve as a comprehensive guide for technical teams aiming to implement this technology in a GMP-compliant manufacturing environment.
- Preparation of Nitro-Piperidinone Intermediate: React substituted methyl cinnamate derivatives with nitromethane via Michael addition, followed by condensation with primary amines and formaldehyde.
- Cyclization to Diazabenzo[c]naphthalene-dione: Protect the amine as a carbamate, then utilize trifluoromethanesulfonic anhydride and DMAP to induce intramolecular cyclization.
- Final Reduction to Octahydrobenzonaphthyridine: Reduce the cyclic dione intermediate using lithium aluminum hydride in anhydrous THF to yield the final amine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits in terms of cost stability and supply reliability. The starting materials, primarily substituted cinnamic acid derivatives and common primary amines, are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency. This raw material accessibility ensures that production schedules can be maintained even during periods of market volatility, providing a secure foundation for long-term supply agreements. Furthermore, the elimination of expensive noble metal catalysts removes a significant variable cost component, allowing for more predictable budgeting and potentially lower overall acquisition costs for the finished intermediate. The robustness of the chemistry also means that batch-to-batch variability is minimized, reducing the likelihood of production delays caused by failed quality control tests.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by replacing precious metal catalysts with inexpensive organic reagents like trifluoromethanesulfonic anhydride and zinc powder. This substitution not only lowers the direct material cost but also reduces the expense associated with metal scavenging and waste disposal, leading to substantial overall savings in the cost of goods sold. Additionally, the high yields reported in the patent examples indicate efficient atom economy, meaning less raw material is wasted per kilogram of product produced. These factors combine to create a highly competitive cost structure that can withstand pricing pressure in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: By utilizing widely available building blocks and avoiding specialized reagents with long lead times, this manufacturing route significantly enhances supply chain resilience. The synthetic steps are performed in common solvents such as dichloromethane, THF, and ethanol, which are staples in any chemical manufacturing facility, further simplifying logistics and inventory management. This operational simplicity allows for rapid scale-up from pilot plant to commercial production without the need for specialized equipment or extensive retraining of personnel. Consequently, suppliers can respond more agilely to fluctuations in demand, ensuring continuous availability of critical intermediates for drug manufacturers.
- Scalability and Environmental Compliance: The synthetic pathway is designed with scalability in mind, featuring reactions that can be safely conducted in standard stainless steel reactors without requiring extreme pressures or temperatures. The absence of heavy metals simplifies the environmental compliance profile, making it easier to obtain necessary permits and adhere to increasingly strict environmental regulations regarding effluent discharge. Waste streams are primarily organic and can be managed through standard incineration or solvent recovery systems, reducing the environmental burden and associated disposal costs. This alignment with green chemistry principles not only protects the environment but also enhances the corporate social responsibility profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these octahydrobenzonaphthyridine compounds. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for making informed decisions about integrating this technology into your existing drug development or manufacturing workflows. We encourage technical teams to review these points carefully to fully appreciate the capabilities and limitations of the described synthetic method.
Q: What is the primary advantage of the octahydrobenzonaphthyridine structure over its precursor?
A: The double reduction of the precursor ketone/amide structure to the octahydrobenzonaphthyridine amine significantly improves water solubility and enhances antitumor activity by up to 100 times compared to the unreduced compound.
Q: How does the cyclization step ensure high purity in the final intermediate?
A: The use of trifluoromethanesulfonic anhydride with DMAP facilitates a clean intramolecular acylation, minimizing side reactions and allowing for effective purification via standard silica gel chromatography.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the route utilizes commercially available starting materials like substituted cinnamates and avoids expensive transition metal catalysts, making it highly scalable and cost-effective for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Octahydrobenzonaphthyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the octahydrobenzonaphthyridine scaffold in modern oncology drug design and are committed to supporting its development through expert contract manufacturing services. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from laboratory discovery to full-scale industrial supply. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the highest international standards. Our dedication to quality and consistency makes us the ideal partner for pharmaceutical companies seeking a dependable source of complex heterocyclic intermediates.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis route to your specific volume and purity requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall manufacturing expenses. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver high-quality octahydrobenzonaphthyridine compounds that will accelerate your drug development timeline. Contact us today to initiate a partnership that combines cutting-edge chemistry with reliable commercial execution.
