Advanced Ball Milling Synthesis for High-Purity Rocuronium Bromide Intermediates
Advanced Ball Milling Synthesis for High-Purity Rocuronium Bromide Intermediates
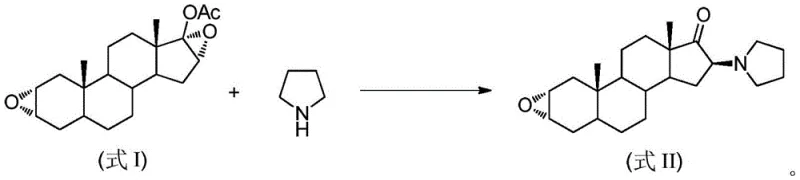
The pharmaceutical industry is constantly seeking more efficient and environmentally sustainable pathways for synthesizing complex steroidal intermediates, particularly for critical muscle relaxants like Rocuronium Bromide. A significant breakthrough in this domain is detailed in patent CN109053855B, which discloses a novel preparation method for 16β-tetrahydropyrrole androstane-2α-epoxy-17-ketone. This compound serves as a pivotal building block in the production of non-depolarizing sterol muscle relaxants, which are essential for modern anesthesia due to their rapid onset and lack of histamine release. The patented technology shifts the paradigm from traditional wet chemistry to mechanochemical synthesis, utilizing mechanical ball milling to drive the reaction. This approach not only addresses the growing regulatory pressure to reduce volatile organic compound (VOC) emissions but also streamlines the manufacturing workflow by collapsing multiple synthetic steps into a single, efficient operation. For global procurement and R&D teams, understanding this technological leap is crucial for securing a reliable supply chain of high-quality pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 16β-tetrahydropyrrole androstane-2α-epoxy-17-ketone has relied on a cumbersome two-step process that poses significant challenges for large-scale manufacturing. As illustrated in the traditional pathway, the process begins with 17-acetoxyandrostane-2α,16α-diepoxide as the raw material. The first step involves a hydrolysis reaction under alkaline conditions to remove the acetoxy group, followed by a separate ring-opening reaction with pyrrolidine. This sequential approach inherently suffers from low atom economy and requires substantial volumes of organic solvents for both reaction and purification stages. Furthermore, the alkaline conditions can lead to poor selectivity, generating difficult-to-separate impurities that compromise the overall yield and purity of the final product. The necessity for extensive post-treatment, including elutriation and complex separation technologies, results in a large volume of waste liquid, increasing both the environmental footprint and the operational costs associated with waste disposal.
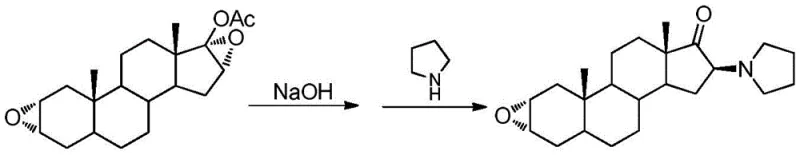
The Novel Approach
In stark contrast to the legacy methods, the innovative technique described in the patent utilizes a direct, one-pot mechanochemical strategy that fundamentally simplifies the production landscape. By employing mechanical ball milling, the reaction between 17-acetoxyandrostane-2α,16α-diepoxide and pyrrolidine is facilitated in the presence of silica gel, effectively bypassing the need for a preliminary hydrolysis step. This solid-state grinding method operates under mild conditions, typically at frequencies between 5-20Hz, and drastically reduces the reliance on organic solvents during the reaction phase. The integration of silica gel acts as a solid support that enhances the contact efficiency between reactants, promoting high conversion rates without the thermal stress often associated with solution-phase heating. Consequently, this novel approach not only shortens the production cycle but also significantly mitigates the generation of hazardous waste, aligning perfectly with green chemistry principles and offering a robust alternative for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Mechanochemical Epoxide Ring Opening
The core of this technological advancement lies in the unique mechanistic pathway enabled by mechanical force, which differs substantially from traditional thermal activation. In the ball milling environment, the kinetic energy transferred by the stainless steel balls to the reactant mixture creates localized high-energy zones that facilitate the nucleophilic attack of pyrrolidine on the epoxide ring. Unlike solution chemistry where solvent molecules might stabilize intermediates or compete for reaction sites, the solid-state environment forces intimate contact between the diepoxide substrate and the amine nucleophile. The silica gel plays a dual role here: it acts as an abrasive to increase surface area and potentially as a mild Lewis acid catalyst to activate the epoxide oxygen, thereby lowering the activation energy for the ring-opening event. This mechanism ensures that the reaction proceeds with high regioselectivity, favoring the formation of the desired 16β-tetrahydropyrrole configuration while suppressing side reactions that typically plague alkaline hydrolysis routes.
From an impurity control perspective, this mechanochemical route offers superior manageability compared to its wet chemistry counterparts. The absence of strong alkaline reagents eliminates the risk of base-catalyzed degradation of the steroid backbone, which is a common source of structural impurities in conventional synthesis. Additionally, the simplicity of the reaction matrix—consisting primarily of the substrate, amine, and silica—means that the post-reaction mixture is far less complex. This allows for straightforward purification strategies, such as simple soaking and filtration followed by recrystallization, rather than requiring chromatographic separation. The ability to achieve high purity through recrystallization from common solvents like methanol or ethanol indicates that the impurity profile is clean and predictable, a critical factor for R&D directors who must ensure that the intermediate meets stringent specifications before being advanced to the final API stage.
How to Synthesize 16β-Tetrahydropyrrole Androstane-2α-Epoxy-17-Ketone Efficiently
Implementing this synthesis route requires precise control over mechanical parameters and stoichiometric ratios to maximize efficiency and yield. The process is designed to be operationally simple, making it highly attractive for scale-up, yet it demands attention to detail regarding the milling frequency and the ratio of solid additives. The patent data suggests that optimizing the mass ratio of the diepoxide to silica gel is critical for maintaining the correct viscosity and grinding efficiency within the mill. Furthermore, the choice of recrystallization solvent post-reaction can fine-tune the final crystal habit and purity. For process chemists looking to adopt this methodology, the following guide outlines the standardized steps derived from the patent examples to ensure reproducible results.
- Load 17-acetoxyandrostane-2α,16α-diepoxide, pyrrolidine, stainless steel balls, and silica gel into a ball milling tank.
- Set the ball mill operating frequency between 5-20Hz and perform mechanical grinding for 10 to 60 minutes.
- Transfer the mixture, soak with a small amount of organic solvent, filter, concentrate, and recrystallize to obtain the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this mechanochemical synthesis represents a strategic opportunity to optimize both cost structures and supply reliability. The elimination of the hydrolysis step and the drastic reduction in solvent usage translate directly into lower raw material costs and reduced expenditure on solvent recovery or disposal systems. By simplifying the process from two distinct chemical transformations into a single mechanical operation, manufacturers can significantly reduce the labor hours and equipment occupancy time required per batch. This efficiency gain allows for higher throughput without the need for capital-intensive expansion of reactor farms, thereby enhancing the overall capacity utilization of the production facility. Moreover, the reduced dependency on large volumes of hazardous solvents mitigates supply chain risks associated with solvent availability and price volatility, ensuring a more stable and predictable production schedule.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the minimization of input materials and waste management costs. By removing the need for alkaline hydrolysis reagents and significantly cutting down on organic solvents, the variable cost per kilogram of the intermediate is substantially lowered. The simplified post-treatment protocol, which avoids complex separation technologies like column chromatography, further reduces operational expenses related to consumables and energy. Additionally, the high yield reported in the patent examples implies better material utilization, meaning less starting material is wasted, which contributes to a more favorable cost of goods sold (COGS) profile for the final product.
- Enhanced Supply Chain Reliability: A streamlined manufacturing process inherently reduces the number of potential failure points in the supply chain. With fewer unit operations and less reliance on sensitive reaction conditions like strict temperature control or anhydrous environments, the risk of batch failures is minimized. The robustness of the ball milling technique means that production can be maintained consistently even under varying external conditions, ensuring that delivery timelines are met reliably. Furthermore, the use of readily available reagents like pyrrolidine and silica gel, combined with the reduced need for specialized solvents, simplifies inventory management and reduces the lead time for sourcing raw materials, thereby strengthening the overall resilience of the supply network.
- Scalability and Environmental Compliance: Scaling mechanochemical processes is increasingly feasible with modern continuous ball milling technologies, allowing for seamless transition from pilot to commercial scale. The inherent safety of operating without large volumes of flammable solvents reduces the regulatory burden and insurance costs associated with hazardous chemical storage and handling. From an environmental standpoint, the drastic reduction in waste liquid generation aligns with increasingly strict global environmental regulations, future-proofing the supply chain against potential regulatory crackdowns on chemical discharge. This sustainability advantage not only lowers compliance costs but also enhances the corporate social responsibility profile of the supply chain, which is a growing priority for multinational pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis method. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a clear picture of what partners can expect when integrating this intermediate into their supply chain. Understanding these nuances is vital for making informed decisions about vendor selection and process adoption.
Q: What are the primary advantages of the ball milling method over traditional solution chemistry for this intermediate?
A: The ball milling method significantly reduces solvent usage, eliminates the need for a separate hydrolysis step, and simplifies post-treatment, leading to higher overall yield and reduced environmental waste compared to the conventional two-step alkaline process.
Q: What are the optimal reaction parameters for maximizing yield in this mechanochemical process?
A: According to patent data, optimal results are achieved with a molar ratio of diepoxide to pyrrolidine between 1:1 and 1:8, a mass ratio of diepoxide to silica gel of 1:2 to 1:10, and a milling frequency of 8-16 Hz for approximately 20 minutes.
Q: How does this synthesis route impact the purity profile of the final API intermediate?
A: The solid-state reaction offers excellent selectivity, minimizing side reactions common in alkaline hydrolysis. Subsequent recrystallization from solvents like methanol/water ensures high purity specifications suitable for downstream pharmaceutical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 16β-Tetrahydropyrrole Androstane-2α-Epoxy-17-Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving anesthetics like Rocuronium Bromide. Our technical team has thoroughly analyzed the advancements presented in patent CN109053855B and is fully equipped to leverage this mechanochemical technology for commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and uninterrupted supply of materials. Our facilities are outfitted with state-of-the-art ball milling equipment and rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required for pharmaceutical applications.
We invite global partners to collaborate with us to optimize their supply chains through this innovative technology. By choosing NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can drive value and efficiency for your organization.
